Chapter 2 Properties of Matter Chemistry 1 Pure



















- Slides: 21

Chapter 2 Properties of Matter Chemistry 1


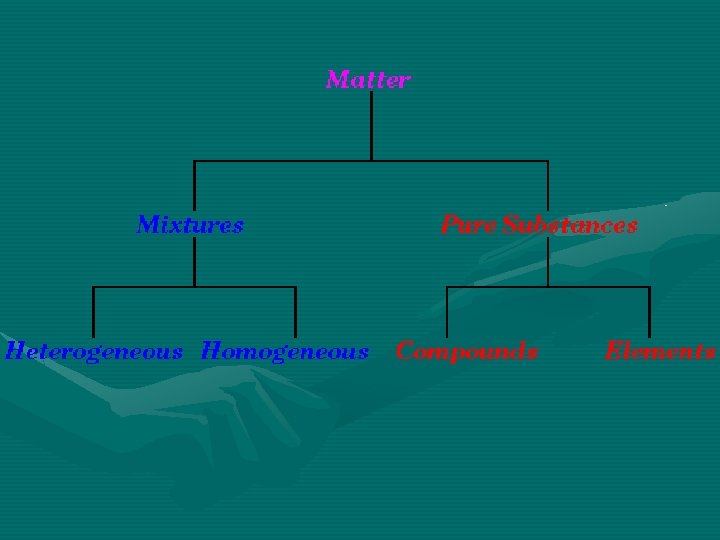
Pure Substances 2. 1 • • • Matter that always has exactly the same composition Every sample the same b/c substance has a fixed, uniform composition 2 categories 1. Element – Ex: Iron, Copper, Carbon 2. Compound – Ex: Water, Sugar, Table Salt

Elements 2. 1 • Substance that can’t be broken down into a simpler substance • Only contains 1 type of atom • Atom – smallest particle of an element • Room temp (68 o. F) = most elements are solid • Elements: – 1 or 2 letters (1 st capitalized, 2 nd lowercase) – Based on Latin names

Compounds 2. 1 • Substance made of 2 or more elements in a fixed proportion • Can be broken down into simpler substances • Compound properties different than elements’ properties that make up compound

Mixtures 2. 1 • • • Properties can vary b/c composition is not fixed Tend to retain properties of individual substance HETEROGENEOUS MIXTURE – “different kind” – Parts are noticeable different – Ex: mixed nuts • HOMOGENEOUS MIXTURE – Substances are so evenly distributed that it is difficult to distinguish one substance from another – Ex: stainless steel = iron + chromium + nickel


• Solution – homogenous mixture 1. 2. 3. – • Suspensions – heterogeneous mixture – 1. 2. 3. – • Liquids do not separate into layers over time If poured thru a filter, substances are not trapped Can see thru b/c light passes without being scattered in all directions Very Small particles “shake well before use” Separate into layers Trapped by a filter Can scatter light in all directions = cloudy Large particles Colloid – 1. 2. 3. – Contains intermediate particles Do not separate into layers Cannot use a filter to separate Scattering of light Examples: milk and fog

Physical Properties 2. 2

Physical Properties 2. 2 • Characteristic of a material that can be observed or measured without changing composition • Viscosity, conductivity, malleability, hardness, melting point, boiling point, density • Viscosity – its resistance to flow – The higher the viscosity, the slower the substance flows – Usually decreases when heated • Conductivity – Ability for heat flow – High conductivity = good conductor metals • If good conductor of heat, normally good conductor of electricity

• Malleability – Ability of a solid to be hammered without shattering – Most metals are malleable – Solids that shatter = brittle • Hardness – See what materials scratch the other – Diamond = hardness known material – What do you use to sharpen a knife? • Melting/Boiling Point – Melting Point – Substance changes from a solid to a liquid • Water = 0 o. C – Boiling Point – Substance boils • Water = 100 o. C

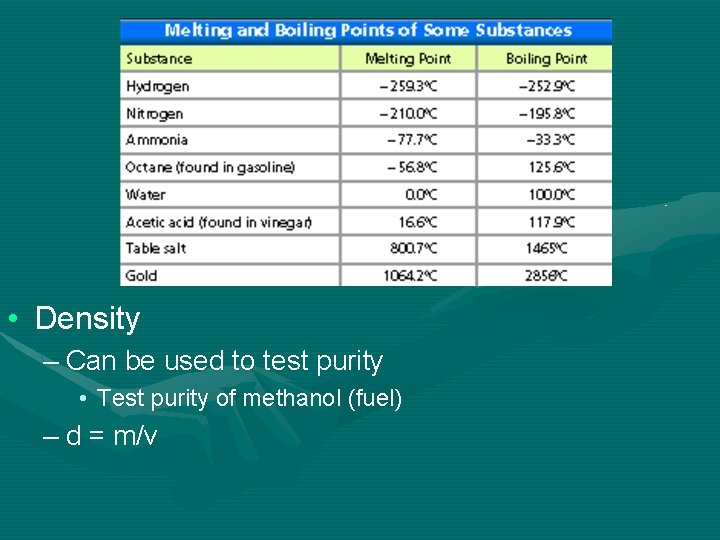
• Density – Can be used to test purity • Test purity of methanol (fuel) – d = m/v

Using Physical Properties 2. 2 • Identifying Material 1. Decide which properties to test 2. Run tests on sample of unknown 3. Compare with previous data reported – Choose Materials – Wouldn’t want a shoelace from wood (must be flexible and durable)

Using Properties to Separate Mixtures 2. 2 • Filtration – process that separates materials based on the size of their particles – Small particles can pass thru a strainer – Ex: brewing coffee • Distillation – process that separates the substance in a solution based on their boiling points – Ex: fresh water for a submarine • Subs can only hold water for about 10 day convert seawater to fresh water


Recognizing Physical Changes 2. 2 • Occurs when some of the properties of a material change but the substance remains the same – Ex: phase changes – Cutting – Crumpling – Some can be reversed, some can not

Chemical Properties 2. 3

Observing Chemical Properties 2. 3 • Ability to produce a change in the composition of matter • Can only be observed when sample is changing to a different substance • Ex: Candles Burning – Candle = made of paraffin = made of C & H • When candle burns, C combines with air to from Carbon Dioxide and H combines with the air to form water.

Chemical Properties 2. 3 • Flammability – Materials ability to burn in the presence of oxygen • Ex: paper and gasoline • Reactivity – How readily a substance combines chemically with other substance • Ex: N – extremely low reactivity • Ex: O – highly reactive element (rust)

Recognizing Chemical Changes 2. 3 • Substance reacts and forms one or more new substances • Change in Color – – Jewelry darkens Matches turn black Roofs Rust • Production of Gas – – Mix vinegar with baking soda = bubbles of carbon dioxide Baking powder + acid in a cake recipe = bubbles of carbon dioxide • Formation of a Precipitate – Formation of any solid that separates from a liquid mixture – Curdling of milk

Is a Change Chemical or Physical? • Can not be POSITIVE a chemical change has occurred when you see bubbles, color change, or precipitate – Ex: water boiling = physical change – Iron heated = gray red = physical change • Ask: are different substances present after the change? – Chemical Change = Composition of matter changes – Physical Change = Composition of matter is the same