Chapter 2 Measurements in Chemistry Questions to be

Chapter 2 Measurements in Chemistry

Questions to be Answered • What does a measurement involve? • How do you make measurements properly? • How do we convert between measurements of one unit to measurements of a new unit?

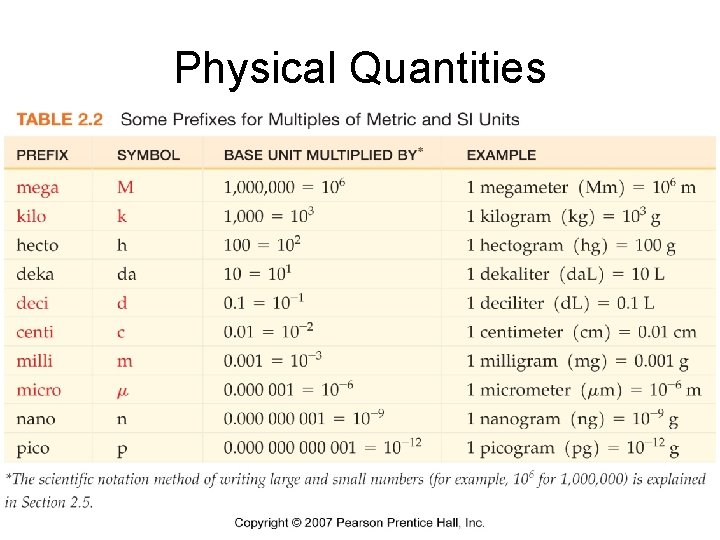
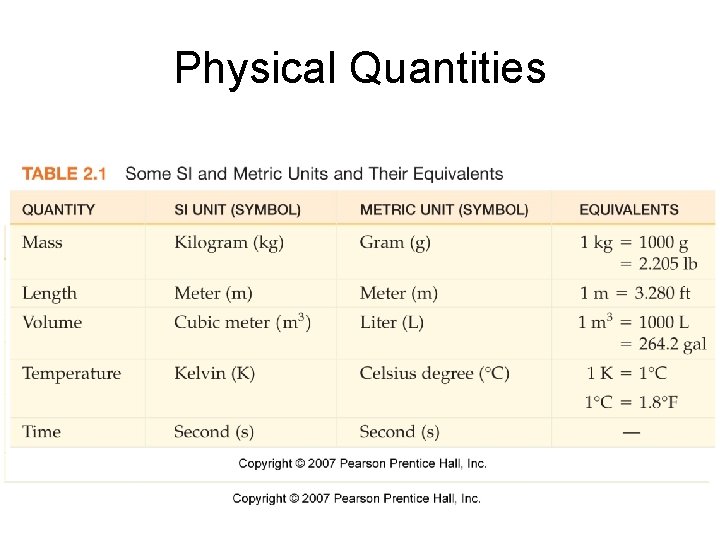
Physical Quantities • Physical quantities – measured physical properties – Number – Unit

Proper Measurements • Number – Reflect the certainty to which the measurement was made • Unit – Represent the type of measurement made • Mass • Volume • Length
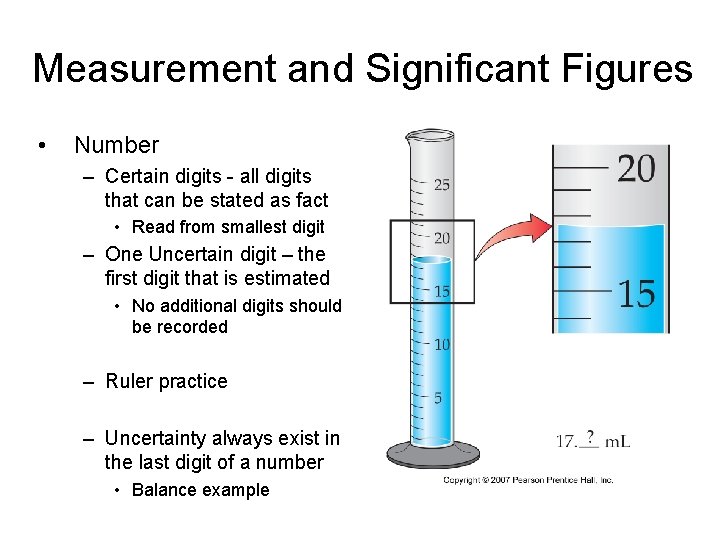
Measurement and Significant Figures • Number – Certain digits - all digits that can be stated as fact • Read from smallest digit – One Uncertain digit – the first digit that is estimated • No additional digits should be recorded – Ruler practice – Uncertainty always exist in the last digit of a number • Balance example

Measurement and Significant Figures • The total number of digits used to express such a measurement is the number of significant figures.

Measurement and Significant Figures Instrument used directly impacts the certainty of the measurement and hence the # of significant figures that can be reported.

Scientific Notation • Scientific notation - convenient way to write a very small or a very large number. – All digits listed in the number portion are significant • Rules for conversion – Move decimal so that it follows first non-zero digit – Write all sig figs in number followed by (x 10) – Raise the ten to the appropriate power • Decimal moved left (+) the number of places moved • Decimal moved right (-) the number of places moved
Physical Quantities
Physical Quantities

Measuring Mass • Mass is a measure of the amount of matter in an object. Mass does not depend on location. • Weight is a measure of the gravitational force acting on an object. Weight depends on location. • Chemist measure grams or milligrams

Measuring Length and Volume • Length has the SI unit of meter (m) • Volume = length x width x height – Units – m 3 – Chemist tend to use milliliters (m. L) or Liters (L)

Measurement and Significant Figures • When reading a measured value – All nonzero digits are significant. – Zero’s • RULE 1. Zeros in the middle of a number are they are always significant. • RULE 2. Zeros at the beginning of a number are not significant • RULE 3. Zeros at the end of a number and after the decimal point are significant. • RULE 4. Zeros at the end of a number and before an implied decimal point may or may not be significant. We cannot tell whether they are part of the measurement or whether they act only to locate the unwritten but implied decimal point. – If a decimal point is shown the zero’s are significant

Problem • Which measurement is expressed to 4 significant figures? – A. – B. – C. – D. – E. 0. 00423 kg 24. 049 cm 1300 K 82, 306 m 62. 40 g

Performing Problems with Measurements • Why are significant figures important? • How do we convert from one unit to another?

Rounding Off Numbers • Often when doing arithmetic on a calculator, the answer is displayed with several digits. – Example - 13. 6 / 28 • How many do you keep?

Rounding Off Numbers • RULE 1. Multiplication or Division – the answer cannot have more significant figures than the original number with the fewest.

Rounding Off Numbers • RULE 2. Addition or Subtraction – the answer cannot have more digits after the decimal point than the original number with the fewest.

Rounding Off Numbers • Once you decide how many digits to retain, the rules for rounding off numbers are straightforward: – If the first number dropped is: • 4 or less – let it rest • 5 or more – let it score

Problem • The appropriate number of significant figures in the result of 15. 234 - 15. 208 is – A. – B. – C. – D. – E. 1 2 3 4 5

Problem • Select the answer that expresses the result of this calculation with the correct number of significant figures. – – – A. B. C. D. E. 13. 3568 13. 357 13. 36 13. 4 13

Problem • The result of (3. 8621 × 1. 5630) - 5. 98 is properly written as – A. – B. – C. – D. – E. 0. 06 0. 0565 0. 056462

Converting a Quantity from One Unit to Another • Factor-Label Method: (Starting quantity) x (Conversion factor) = Equivalent quantity

Converting a Quantity from One Unit to Another • What is a conversion unit – Ratios, fractions, or two measured quantities that are equivalent – Equal 1 • The important item in these numbers are UNITS

Example • How many kilometers is 26. 22 miles? – STEP 1: Identify the information given. – STEP 2: Identify the information needed to answer. – STEP 3: Find the relationship(s) between the known information and unknown answer, and plan a series of steps, including conversion factors, for getting from one to the other. – STEP 4: Solve the problem. – BALLPARK CHECK: Make a rough estimate to be sure the value and the units of your calculated answer are reasonable.

Problem • The distance between carbon atoms in ethylene is 134 picometers. Which of the following expresses that distance in meters? – A. – B. – C. – D. – E. 1. 34 × 10 -13 m 1. 34 × 10 -12 m 1. 34 × 10 -10 m 1. 34 × 10 -7 m 1. 34 × 10 -6 m

Problem • A dose of medication was prescribed to be 35 microliters. Which of the following expresses that volume in centiliters? – A. – B. – C. – D. – E. 3. 5 × 105 c. L 3. 5 × 104 c. L 3. 5 × 10 -4 c. L 3. 5 × 10 -3 c. L

Problem • The average distance between the Earth and the Moon is 240, 000 miles. Express this distance in kilometers. – A. – B. – C. – D. – E. 6. 1 × 105 km 5. 3 × 105 km 3. 9 × l 05 km 1. 5 × 105 km 9. 4 × 104 km

Problem • The speed needed to escape the pull of Earth's gravity is 11. 3 km/s. What is this speed in mi/h? – A. – B. – C. – D. – E. 65, 500 mi/h 25, 300 mi/h 18, 200 mi/h 1, 090 mi/h 5. 02 × 10 -3 mi/h
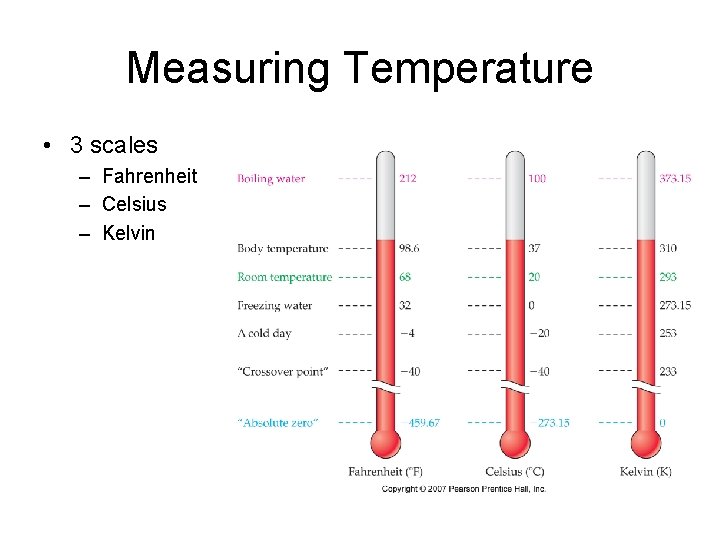
Measuring Temperature • 3 scales – Fahrenheit – Celsius – Kelvin

Measuring Temperatures • Converting Between Temperature Scales – – o. F = (1. 8 x o. C) + 32 K = o. C + 273. 15

Problem • Isopropyl alcohol, commonly known as rubbing alcohol, boils at 82. 4°C. What is the boiling point in kelvins? – A. – B. – C. – D. – E. 387. 6 K 355. 6 K 323. 6 K 190. 8 K -190. 8 K

Problem • Acetic acid boils at 244. 2°F. What is its boiling point in degrees Celsius? – A. – B. – C. – D. – E. 382. 0°C 167. 7°C 153. 4°C 117. 9°C 103. 7°C

Units of Energy and Heat • Energy: The capacity to do work or supply heat. – SI units - Joule (J) – calorie is another unit often used to measure energy. • One calorie (cal) - the amount of heat necessary to raise the temperature of 1 g of water by 1°C. – Calorie – food calorie • Energy equivalencies – 4. 184 J = 1 cal – 1000 cal = 1 Cal – 4. 184 k. J = 1 Cal

Units of Energy and Heat • A Snickers® candy bar contains 280 Calories, of which the fat content accounts for 120 Calories. What is the energy of the fat content, in k. J? – – – A. B. C. D. E. 5. 0 × 10 -1 k. J 29 k. J 5. 0 × 102 k. J 1. 2 × 103 k. J 5. 0 × 105 k. J

Problem • Natural gas, or methane, is an important fuel. Combustion of one mole of methane releases 802. 3 kilojoules of energy. How much energy does that represent in kilocalories? – – – A. B. C. D. E. 1. 918 × 10 -1 kcal 1. 918 × 102 kcal 3. 360 × 103 kcal 1. 918 × 105 kcal 3. 360 × 106 kcal

Units of Heat and Energy • Not all substances are created equal. – One calorie raises the temperature of 1 g of water by 1°C but raises the temperature of 1 g of iron by 10°C. • The amount of heat needed to raise the temperature of 1 g of a substance by 1°C is called the specific heat of the substance (c). • Specific heat is measured in units of cal/g C or J/go. C

Units of Heat and Energy • q = mcΔT – q = heat change – m = mass – c = specific heat

Units of Heat and Energy • Calculate q when 28. 6 g of water is heated from 22. 0°C to 78. 3°C. (cwater = 4. 184 J/go. C) – A. – B. – C. – D. – E. 0. 385 k. J 1. 61 k. J 6. 74 k. J 9. 37 k. J 1. 61 × 103 k. J

Problem • Ethylene glycol, used as a coolant in automotive engines, has a specific heat capacity of 2. 42 J/(go. C). Calculate q when 3. 65 kg of ethylene glycol is cooled from 132°C to 85°C. – – – A. B. C. D. E. -1900 k. J -420 k. J -99 k. J -0. 42 k. J -4. 2 × 10 -6 k. J

Density • Density relates the mass of an object to its volume. – Units • grams per cubic centimeter (g/cm 3) for solids • grams per milliliter (g/m. L) for liquids. Density = Mass (g) Volume (m. L or cm 3)

Density • If the gasoline in a full 20. 0 gallon tank weighs 116 lb, what is the density of gasoline in g/m. L • How many grams does 1. 2 L of water weigh, if at room temperature water has a density of 0. 9970 g/cm 3

Optional Homework • Text - 2. 44, 2. 45, 2. 46, 2. 47, 2. 48, 2. 50, 2. 52, 2. 54, 2. 56, 2. 58, 2. 62, 2. 64, 2. 66, 2. 68, 2. 70, 2. 72, 2. 74, 2. 78, 2. 88, 2. 90, 2. 96, 2. 106 • Chapter 2 Homework - found online
- Slides: 43