Chapter 2 Matter Can be physically separated Substances

* Chapter 2
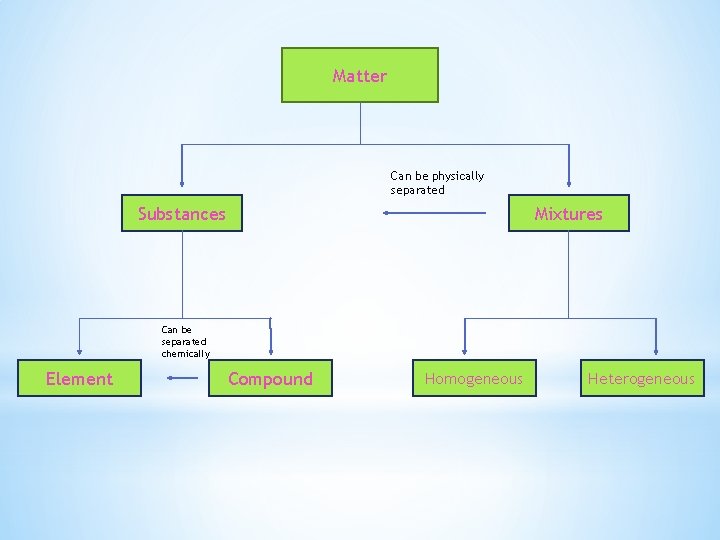
Matter Can be physically separated Substances Mixtures Can be separated chemically Element Compound Homogeneous Heterogeneous

* *Matter: Anything that has mass and takes up space *Two ways to describe matter: * Extensive: a property that depends on the amount of matter in a sample. (mass, volume) * Intensive: property that depends on the type of matter in a sample, not the amount of matter. (hardness, melting point, color, boiling point)

* *Solid: definite shape and volume *Liquid: indefinite shape, flows, fixed volume *Gas: indefinite shape and volume, compresses easily * Vapor: gaseous state of a substance that’s liquid at room temp.

* *Substance (or pure substance): matter that has a uniform and definite composition and can’t be separated without changing chemical composition. * Every sample of a substance has identical intensive properties * Substances can be either an element or a compound Element Compound Simplest form of matter that has a unique set of properties Substance that contains two or more elements chemically combined in a fixed proportion -can be broken down chemically

* *Mixture: physical blend of two or more components * Heterogeneous: composition is not uniform * Homogeneous (aka solution): composition is uniform throughout

* *Milk *Chicken Noodle Soup *Muddy Water *Air *Hot Chocolate *Red Bull *Apple *Bronze

*Complete the “Whatsa Matter” Worksheet with a partner! *

*Physical Property: quality or condition of a substance that can be observed or measured without changing the substance’s composition. (color, melting point, boiling point) *Physical change: some properties of a material change, but the composition of the material does not change. * Can be reversible or irreversible * Words: boil, freeze, melt, condense, break, split, grind, crush, cut http: //ishare. rediff. com/video/entertainment/diet-cokementos/676123 *

*Chemical Property: the ability of a specific substance to undergo a specific chemical change. (Ability to rust) *Chemical Change (aka chemical reaction): A change that produces matter with a different composition than the original matter. * The composition of matter always changes * Words: burn, rot, rust, decompose, ferment, explode, corrode *

*Clues (although they can be tricky) * Transfer of energy * Change in color * Production of gas * Formation of a precipitate (solid that forms and settles out of a liquid) How can you know for sure it’s a physical or chemical change? Test the sample’s composition *

*Law of Conservation of Mass: in any physical or chemical change, mass is conserved. *The mass of the products is always equal to the mass of the reactants. *

* *Sugar dissolving in tea • Chemical Change • Physical Change

* *Logs burning • Chemical Change • Physical Change

* *Breaking water up by separating it into hydrogen and oxygen • Chemical Change • Physical Change

* *Metal rusting • Chemical Change • Physical Change http: //tess. schoolwaxtv. com/learn-lab-files-physical-chemical-changes

* e g n a h C l a c i s y h P C hem ica l. C ha ng e Hom ogen Pur e Su bst ance Matter HETEROGENEOUS ous
- Slides: 17