Chapter 2 Matter and Change Anything in black

Chapter 2 Matter and Change Anything in black letters = write it in your notes (‘knowts’)
2. 1 – Properties of Matter - Anything that has mass Mass - Measure of the amount of matter in an object Matter Mass We need a better definition for mass…

Mass - Measure of an object’s inertia. Inertia - Resistance to change in motion. Which of the above has more inertia? Volume - amount of space occupied by an object

mass Density = volume UNITS mass grams volume m. L density g/m. L or cm 3 or g/cm 3

The density of an object normally decreases as its temperature increases. Hot air rises!! The density of water is approx. 1 g/m. L

States of Matter Solid Particles are packed closely together; definite shape and volume
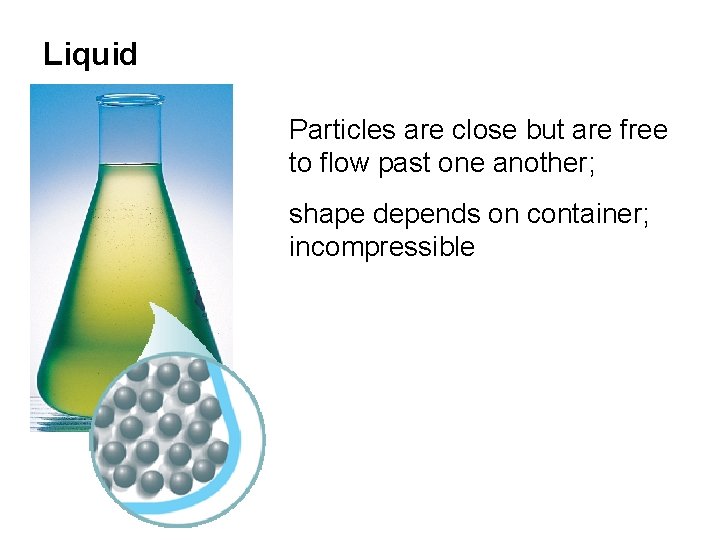
Liquid Particles are close but are free to flow past one another; shape depends on container; incompressible

Gas Particles are far apart and move freely; shape and volume depend on container; can be compressed

Physical Change A change in appearance but NOT in the chemical identity of a substance. The melting point of gallium metal is 30˚C. The figure shows how the heat from a person’s hand can melt a sample of gallium.
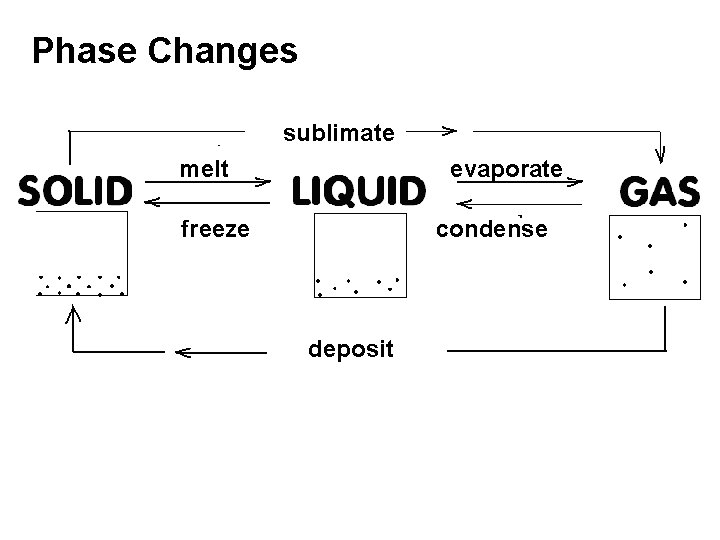
Phase Changes sublimate melt evaporate freeze condense deposit
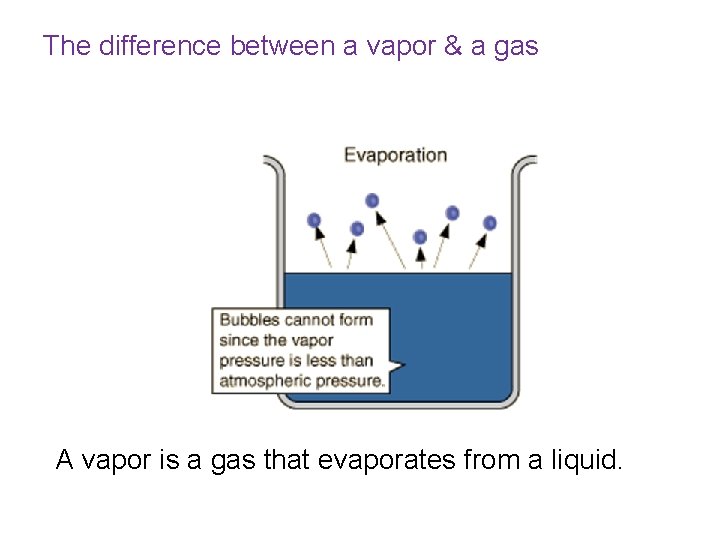
The difference between a vapor & a gas A vapor is a gas that evaporates from a liquid.
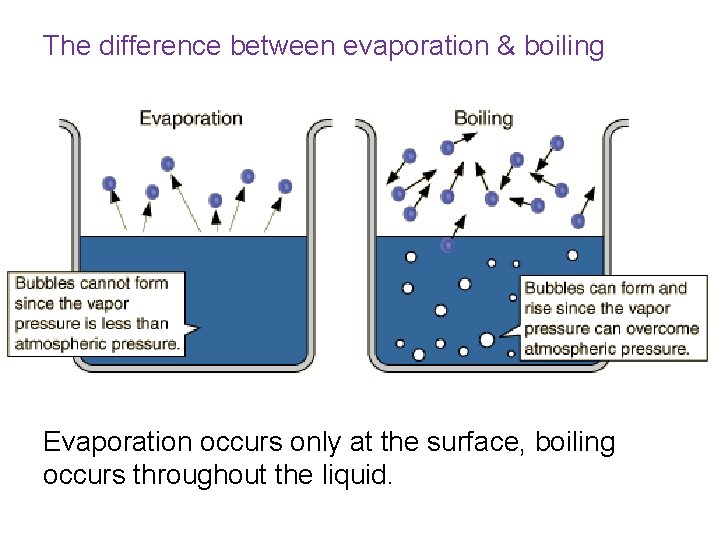
The difference between evaporation & boiling Evaporation occurs only at the surface, boiling occurs throughout the liquid.

The white cloud is NOT steam. It is actually hot water vapor condensing to form a mist of water droplets

2. 2 – Mixtures Mixture - physical blend of 2 or more things Homogeneous – same composition throughout, uniformly mixed; solutions Heterogeneous – variable composition, nonuniformly mixed; contains more than 1 phase.

Olive oil Homogeneous Vinegar Homogeneous Oil & Vinegar Heterogeneous (2 phases)
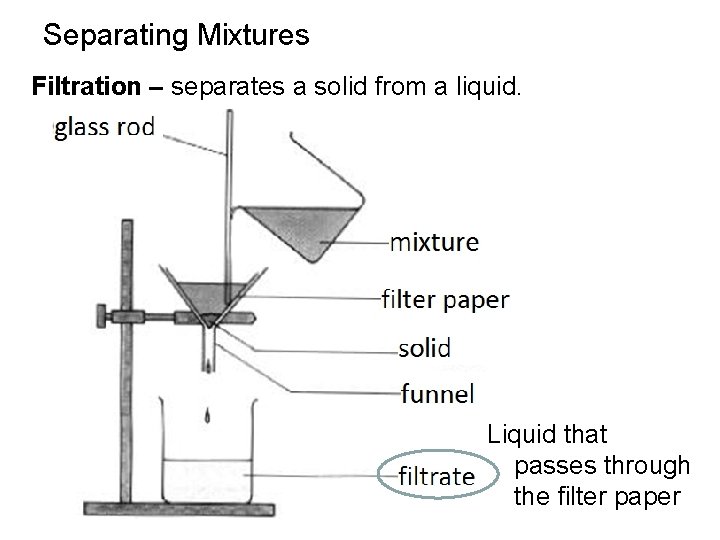
Separating Mixtures Filtration – separates a solid from a liquid. Liquid that passes through the filter paper
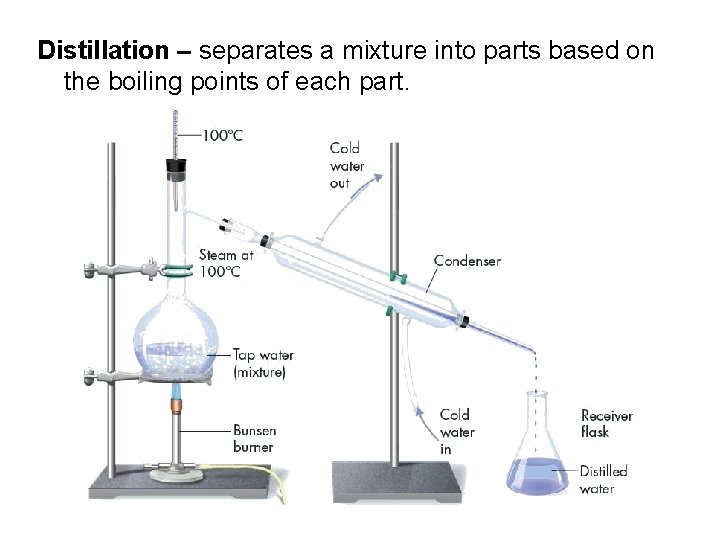
Distillation – separates a mixture into parts based on the boiling points of each part.
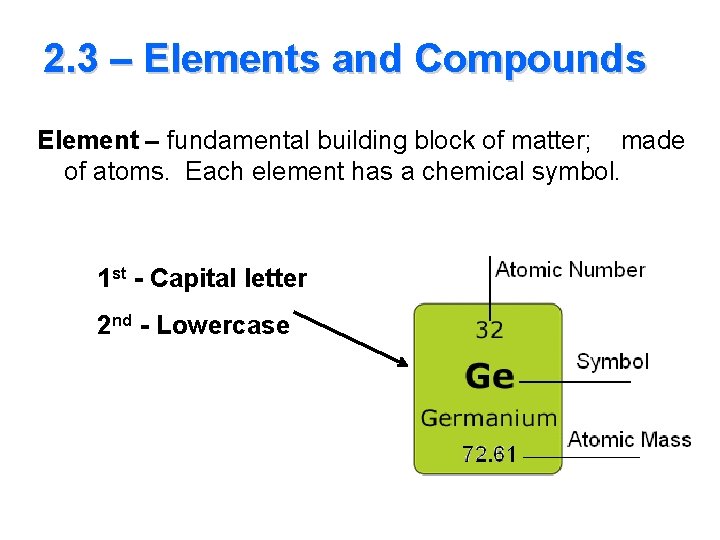
2. 3 – Elements and Compounds Element – fundamental building block of matter; made of atoms. Each element has a chemical symbol. 1 st - Capital letter 2 nd - Lowercase
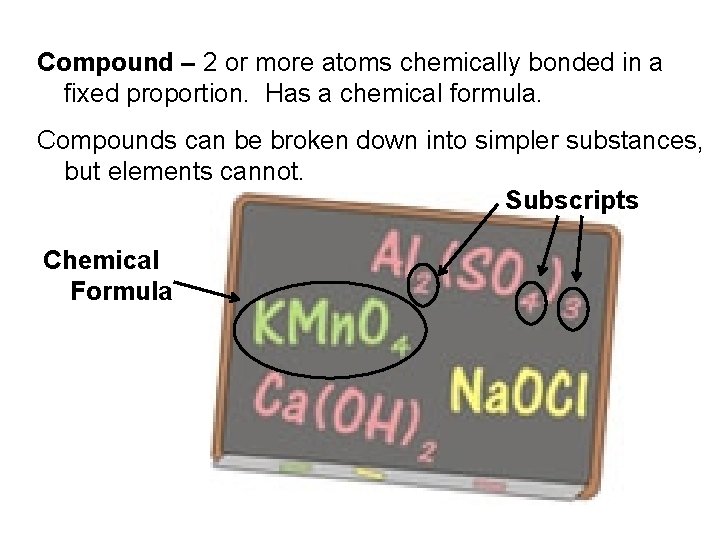
Compound – 2 or more atoms chemically bonded in a fixed proportion. Has a chemical formula. Compounds can be broken down into simpler substances, but elements cannot. Subscripts Chemical Formula

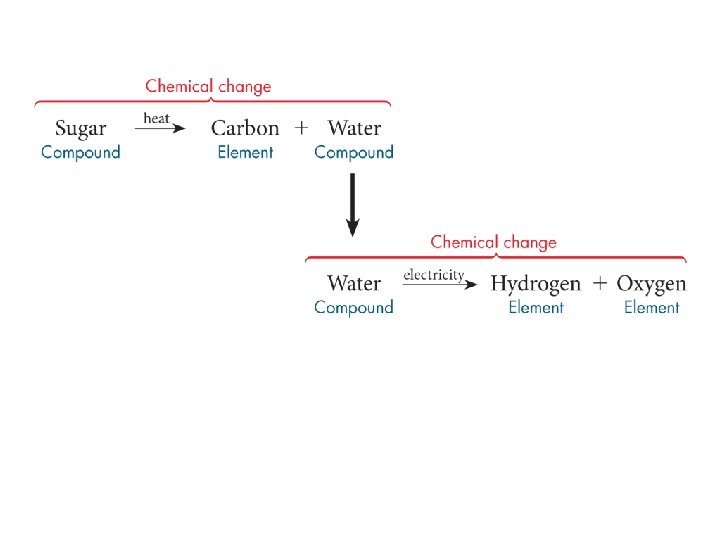
Chemical Change change in chemical identity; requires a chemical reaction. The layer of sugar is heated until it breaks down into solid carbon and water vapor.

The properties of compounds are different from the elements that form them. + Sodium (Na) Metal so reactive it reacts with water Chlorine (Cl 2) Toxic gas used in WWI for chemical warfare Salt (Na. Cl) Relatively harmless 2 poisonous substances react to form a completely benign compound that is necessary for us to live.
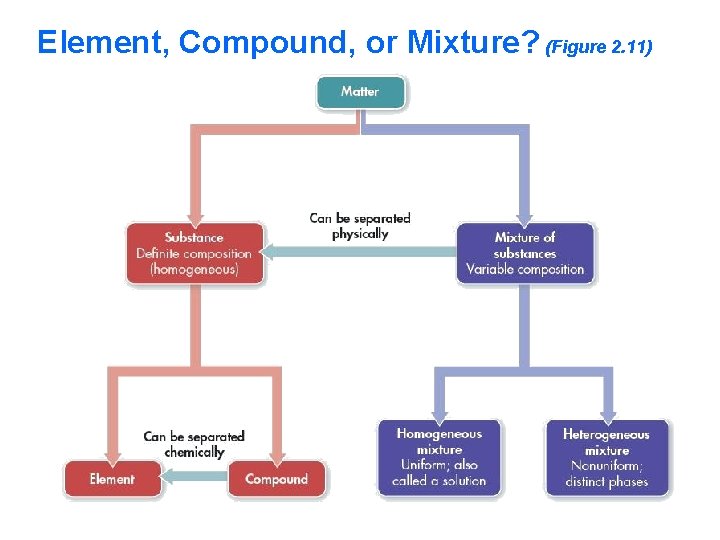
Element, Compound, or Mixture? (Figure 2. 11)
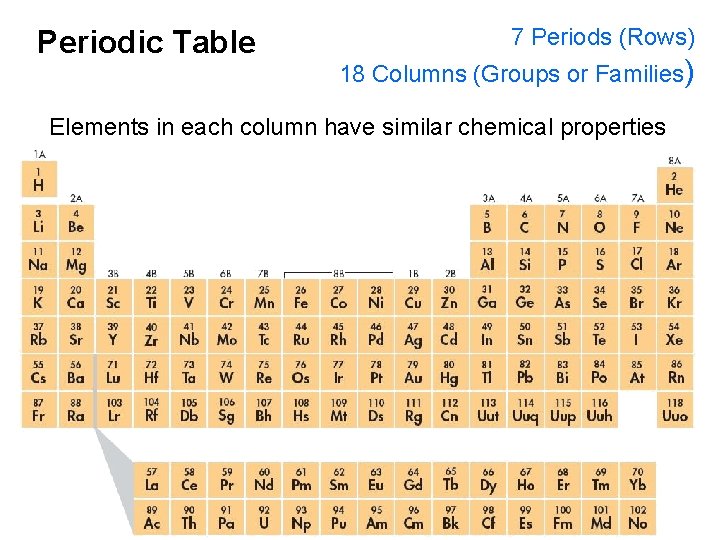
Periodic Table 7 Periods (Rows) 18 Columns (Groups or Families) Elements in each column have similar chemical properties

2. 4 – Chemical Reactions Chemical changes are also known as chemical reactions. Reactant(s) Product(s)

Possible clues that a reaction has happened 1. Transfer of Energy

Possible clues that a reaction has happened 2. Color Change

Possible clues that a reaction has happened 3. Gas Production

Possible clues that a reaction has happened 4. Precipitate Formation

Law of Conservation of Mass In any chemical reaction, reactant mass = product mass
- Slides: 30