Chapter 2 Essential Chemistry for Biology Power Point

Chapter 2 Essential Chemistry for Biology Power. Point® Lectures created by Edward J. Zalisko for Campbell Essential Biology, Sixth Edition, and Campbell Essential Biology with Physiology, Fifth Edition – Eric J. Simon, Jean L. Dickey, Kelly A. Hogan, and Jane B. Reece © 2016 Pearson Education, Inc.

Figure 2. 0 -1 Why Chemistry Matters © 2016 Pearson Education, Inc.

Figure 2. 0 -1 a © 2016 Pearson Education, Inc.

Figure 2. 0 -1 b © 2016 Pearson Education, Inc.

Figure 2. 0 -1 c © 2016 Pearson Education, Inc.

Figure 2. 0 -2 Chapter Thread: Radioactivity © 2016 Pearson Education, Inc.

Biology and Society: Radiation and Health • Radioactivity, the emission of high-energy particles, can • penetrate living tissues and • kill cells by damaging DNA. • Radiation can also be medically beneficial, helping to treat cancer. © 2016 Pearson Education, Inc.

Biology and Society: Radiation and Health • Radiation is most dangerous when exposure is • uncontrolled and • covers most or all of the body, such as when a person is exposed to radioactive fallout from a nuclear detonation or accident. © 2016 Pearson Education, Inc.

Biology and Society: Radiation and Health • Controlled medical radiation therapy exposes only a small part of the body to a precise dosage of radiation. • This provides a large and deadly dose to cancerous cells but mostly spares surrounding healthy tissues. © 2016 Pearson Education, Inc.

Biology and Society: Radiation and Health • What makes something radioactive? • Many questions about life can be reduced to questions about chemicals and their interactions. • Knowledge of chemistry is therefore essential to understanding biology. © 2016 Pearson Education, Inc.

Some Basic Chemistry • Take any biological system apart, and you eventually end up at the chemical level. • Chemical reactions are always occurring in the human body. © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • Matter is anything that occupies space and has mass. • Matter is found on Earth in three physical states: 1. solid, 2. liquid, and 3. gas. © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • Mass is a measure of the amount of material in an object. • An element is a substance that cannot be broken down into other substances by chemical reactions. • All matter is composed of chemical elements. © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • There are 92 naturally occurring elements. Examples are • carbon, • oxygen, and • gold. • Each element has a symbol derived from its English, Latin, or German name. © 2016 Pearson Education, Inc.
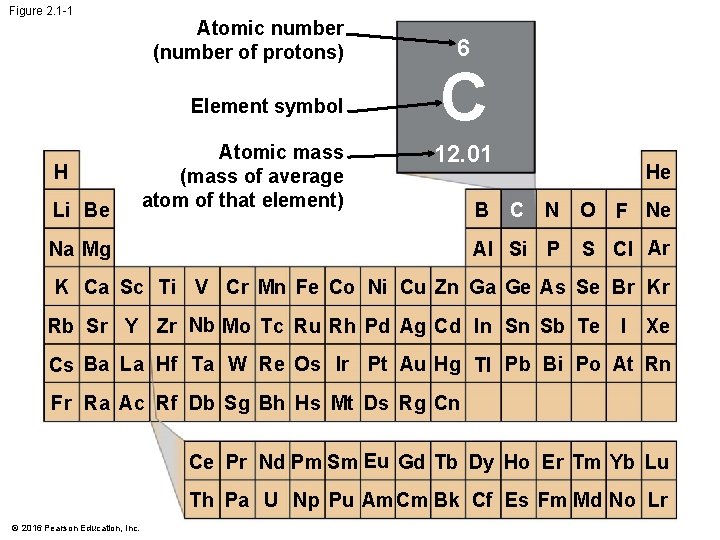
Figure 2. 1 -1 Atomic number (number of protons) Element symbol H Li Be Atomic mass (mass of average atom of that element) 6 C 12. 01 Na Mg B He C N AI Si P O F Ne S CI Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg Cn Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr © 2016 Pearson Education, Inc.

Figure 2. 1 -2 Mercury (Hg) © 2016 Pearson Education, Inc.

Figure 2. 1 -3 Copper (Cu) © 2016 Pearson Education, Inc.

Figure 2. 1 -4 Lead (Pb) © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • Of the naturally occurring elements, 25 are essential to people. • Four of these elements make up about 96% of the weight of the body. These four elements are 1. oxygen (O), 2. carbon (C), 3. hydrogen (H), and 4. nitrogen (N). © 2016 Pearson Education, Inc.

Figure 2. 2 Carbon (C): 18. 5% Oxygen (O): 65. 0% Calcium (Ca): 1. 5% Phosphorus (P): 1. 0% Potassium (K): 0. 4% Sulfur (S): 0. 3% Sodium (Na): 0. 2% Chlorine (Cl): 0. 2% Hydrogen (H): 9. 5% Nitrogen (N): 3. 3% © 2016 Pearson Education, Inc. Magnesium (Mg): 0. 1% Trace elements: less than 0. 01% Boron (B) Chromium (Cr) Cobalt (Co) Copper (Cu) Fluorine (F) Iodine (I) Iron (Fe) Manganese (Mn) Molybdenum (Mo) Selenium (Se) Silicon (Si) Tin (Sn) Vanadium (V) Zinc (Zn)

Matter: Elements and Compounds • Much of the remaining 4% is accounted for by 7 elements. These include the following. 1. Calcium, important for building strong bones and teeth, is found abundantly in dairy products, sardines, and green, leafy vegetables. 2. Phosphorus, a component of DNA, can be obtained by eating eggs, beans, and nuts. © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • Trace elements are • required in only very small amounts and • essential for life. • An iodine deficiency causes goiter. • Fluorine • is added to dental products and drinking water and • helps to maintain healthy bones and teeth. © 2016 Pearson Education, Inc.
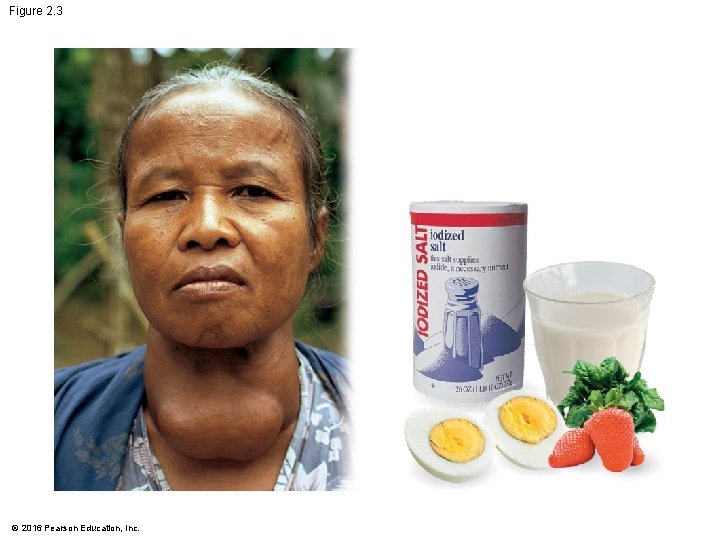
Figure 2. 3 © 2016 Pearson Education, Inc.

Figure 2. 3 -1 © 2016 Pearson Education, Inc.

Figure 2. 3 -2 © 2016 Pearson Education, Inc.

Matter: Elements and Compounds • Elements can combine to form compounds, substances that contain two or more elements in a fixed ratio. • Common compounds include • Na. Cl (table salt) and • H 2 O (water). © 2016 Pearson Education, Inc.

Atoms • Each element consists of one kind of atom. • An atom is the smallest unit of matter that still retains the properties of an element. © 2016 Pearson Education, Inc.

The Structure of Atoms • Atoms are composed of subatomic particles. • A proton is positively charged. • An electron is negatively charged. • A neutron is electrically neutral. • When an atom has an equal number of protons and electrons, • its net electrical charge is zero and • the atom is neutral. © 2016 Pearson Education, Inc.

The Structure of Atoms • Most atoms have protons and neutrons packed tightly into the nucleus. • The nucleus is the atom’s central core. • Electrons move around the nucleus. © 2016 Pearson Education, Inc.
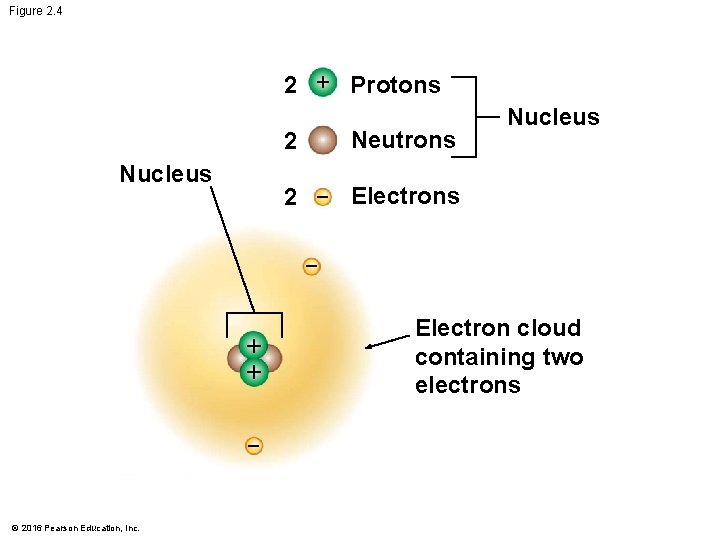
Figure 2. 4 2 + Protons Neutrons 2 Nucleus 2 Electrons + + © 2016 Pearson Education, Inc. Electron cloud containing two electrons

The Structure of Atoms • All atoms of a particular element have the same unique number of protons. • This number is the element’s atomic number. • Thus, an atom of helium, with 2 protons, has an atomic number of 2, and no other element has 2 protons. • An atom’s mass number is the sum of the number of protons and neutrons in its nucleus. © 2016 Pearson Education, Inc.

The Structure of Atoms • An atom’s atomic mass • is close to its mass number, the sum of its protons and neutrons, • but may differ slightly because it represents an average of all the naturally occurring forms of that element. © 2016 Pearson Education, Inc.

Isotopes • Isotopes are alternate mass forms of an element. • Isotopes • have the same number of protons and electrons but • differ in their number of neutrons. © 2016 Pearson Education, Inc.

Table 2. 1 © 2016 Pearson Education, Inc.

Isotopes • A radioactive isotope is one in which the nucleus decays spontaneously. • Radiation from decaying isotopes can • damage cellular molecules and • pose serious health risks. • Natural sources of radiation can also pose a threat. • Radon, a radioactive gas, is a carcinogen that can cause lung cancer. • Radon testing is a standard procedure before a house is bought or sold. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • Cells use radioactive isotopes the same way they use nonradioactive isotopes of the same element. • Once the cell takes up a radioactive isotope, the location and concentration of the isotope can be detected because of the radiation it emits. • This makes radioactive isotopes useful as tracers for monitoring living organisms. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • A medical diagnostic tool called a PET scan works by detecting small amounts of radiation emitted by radioactive materials that were purposefully introduced into the body. © 2016 Pearson Education, Inc.
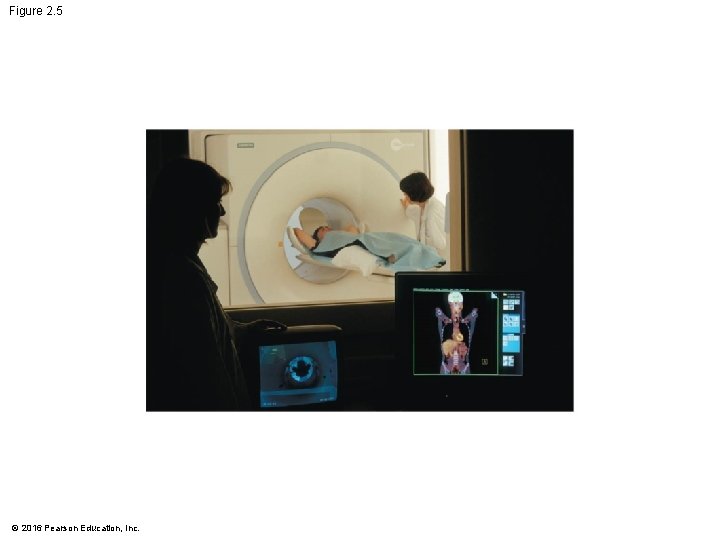
Figure 2. 5 © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • In 2012, researchers from a U. S. pharmaceutical company announced the results of a study that used PET scans to investigate Alzheimer’s disease. • The observation that the brains of people with Alzheimer’s are often filled with clumps of a protein called amyloid led the researchers to question whether these clumps could be detected by a PET scan. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • The researchers formed the hypothesis that a molecule called florbetapir, which contains the radioactive isotope fluorine-18, could be detected by PET scans after it binds to amyloid deposits in living patients. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • The researchers’ prediction was that using florbetapir during PET scans could help with diagnosis and thereby improve disease management. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • Their experiment involved using the radioactive isotope and PET scans to test 229 patients who had been diagnosed with mental decline. © 2016 Pearson Education, Inc.

The Process of Science: Can Radioactive Tracers Identify Brain Diseases? • The results indicated that 113 patients showed amyloid deposits in their PET scans. As a result, • doctors changed the diagnosis in 55% of the patients and • the PET scan data led to changes in treatment (such as different drugs) in 87% of cases. © 2016 Pearson Education, Inc.

Chemical Bonding and Molecules • Only electrons are directly involved in chemical reactions. • The number of electrons in an atom determines the chemical properties of that atom. • Chemical reactions enable atoms to transfer or share electrons. • These interactions usually result in atoms staying close together, held by attractions called chemical bonds. © 2016 Pearson Education, Inc.

Ionic Bonds • When an atom loses or gains electrons, it becomes electrically charged. • Ions are atoms or molecules that are electrically charged as a result of gaining or losing electrons. • Ionic bonds are formed between oppositely charged ions. • Compounds, such as table salt, that are held together by ionic bonds, are called ionic compounds. © 2016 Pearson Education, Inc.
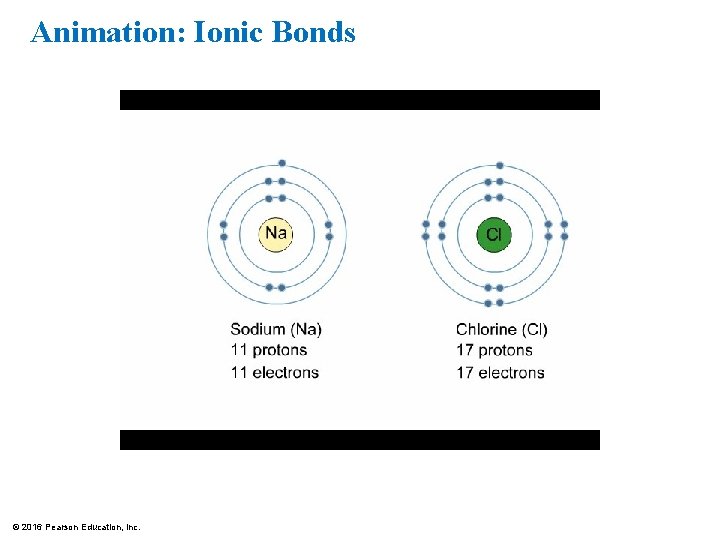
Animation: Ionic Bonds © 2016 Pearson Education, Inc.
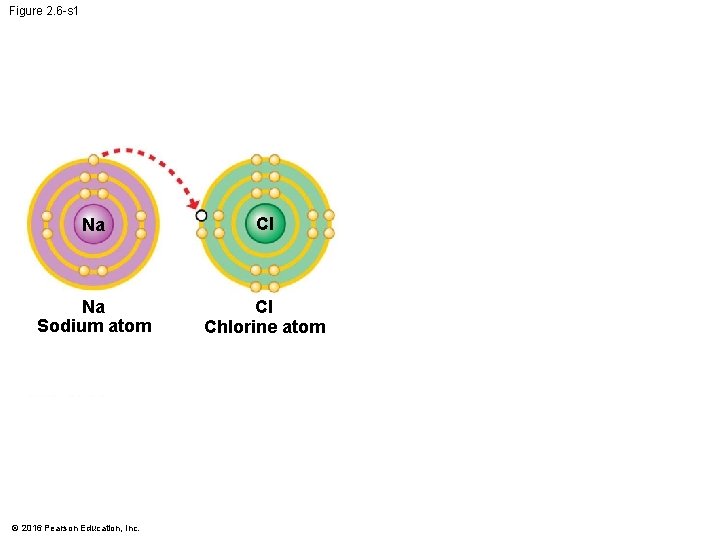
Figure 2. 6 -s 1 Na Cl Na Sodium atom Cl Chlorine atom © 2016 Pearson Education, Inc.
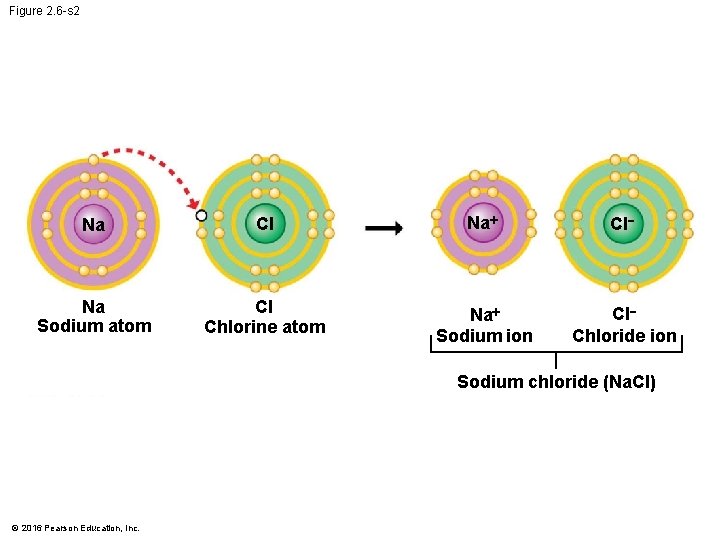
Figure 2. 6 -s 2 Na Cl Na+ Cl Na Sodium atom Cl Chlorine atom Na+ Sodium ion Cl Chloride ion Sodium chloride (Na. Cl) © 2016 Pearson Education, Inc.

Covalent Bonds • A covalent bond forms when two atoms share one or more pairs of electrons. • Covalent bonds • are the strongest of the various bonds and • hold atoms together in a molecule. © 2016 Pearson Education, Inc.

Animation: Covalent Bonds © 2016 Pearson Education, Inc.
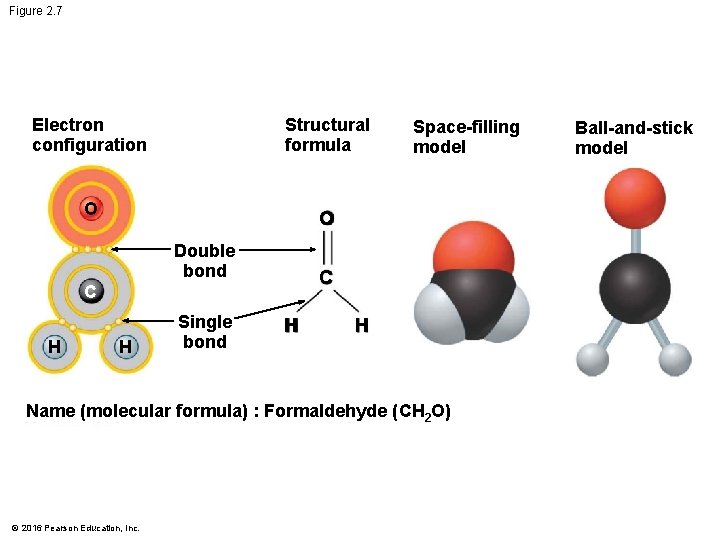
Figure 2. 7 Electron configuration Structural formula O O Double bond C C H H Single bond Space-filling model H H Name (molecular formula) : Formaldehyde (CH 2 O) © 2016 Pearson Education, Inc. Ball-and-stick model

Hydrogen Bonds • A molecule of water (H 2 O) consists of two hydrogen atoms joined to one oxygen atom by single covalent bonds, but the electrons are not shared equally between the oxygen and hydrogen atoms. • This causes water to be a polar molecule, one with an uneven distribution of charge. © 2016 Pearson Education, Inc.

Animation: Water Structure © 2016 Pearson Education, Inc.

Hydrogen Bonds • The polarity of water results in weak electrical attractions between neighboring water molecules. • These weak attractions are called hydrogen bonds. © 2016 Pearson Education, Inc.
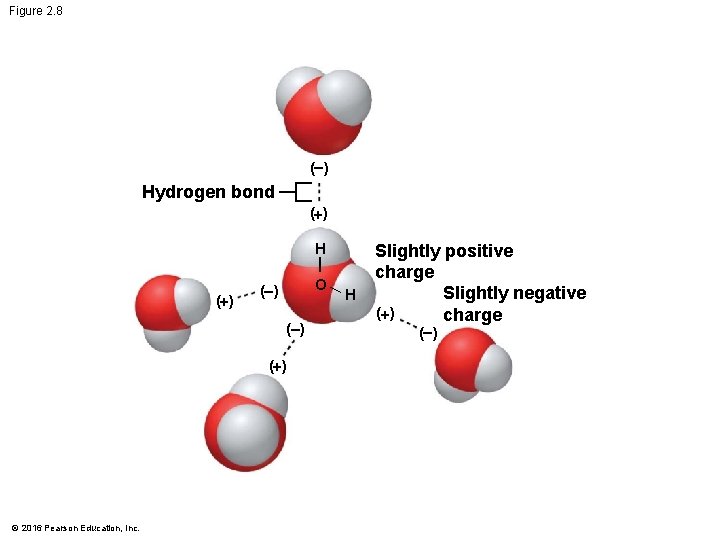
Figure 2. 8 ( ) Hydrogen bond (+) H (+) O ( ) (+) © 2016 Pearson Education, Inc. H Slightly positive charge Slightly negative (+) charge ( )

Chemical Reactions • Cells constantly rearrange molecules by • breaking existing chemical bonds and • forming new ones. • Such changes in the chemical composition of matter are called chemical reactions. © 2016 Pearson Education, Inc.

Chemical Reactions • Chemical reactions include • reactants, the starting materials, and • products, the end materials. • Chemical reactions • can rearrange matter • but cannot create or destroy matter. © 2016 Pearson Education, Inc.

Water and Life • Life on Earth began in water and evolved there for 3 billion years before spreading onto land. • Modern life is still tied to water. • Your cells are composed of 70– 95% water. • The abundance of water is a major reason that Earth is habitable. © 2016 Pearson Education, Inc.

Figure 2. 9 © 2016 Pearson Education, Inc.

Structure/Function: Water • The polarity of water molecules and the hydrogen bonding that results explain most of water’s lifesupporting properties. • We will explore four of those properties: 1. the cohesive nature of water, 2. the ability of water to moderate temperature, 3. the biological significance of ice floating, and 4. the versatility of water as a solvent. © 2016 Pearson Education, Inc.

The Cohesion of Water • Water molecules stick together as a result of hydrogen bonding. • This tendency of molecules of the same kind to stick together is called cohesion and is much stronger for water than for most other liquids. • Trees depend on cohesion to help transport water from their roots to their leaves. © 2016 Pearson Education, Inc.
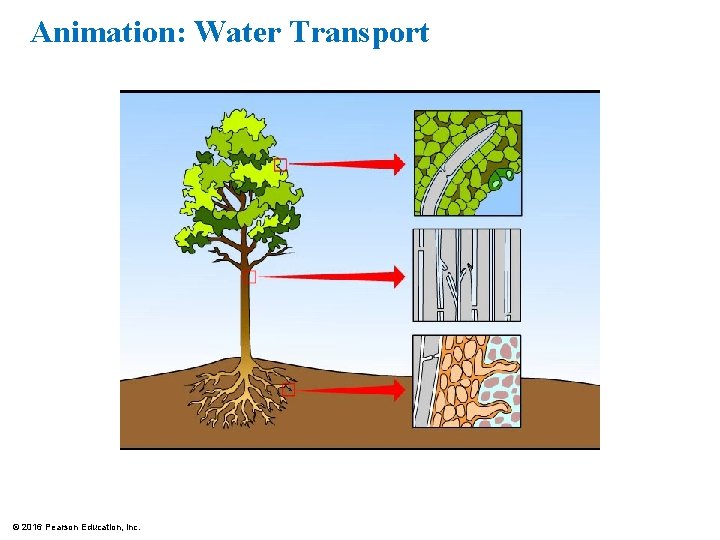
Animation: Water Transport © 2016 Pearson Education, Inc.

Figure 2. 10 Evaporation from the leaves Colorized SEM Flo w of w ate r Microscopic waterconducting tubes © 2016 Pearson Education, Inc. Cohesion due to hydrogen bonds between water molecules

Figure 2. 10 -1 Evaporation from the leaves © 2016 Pearson Education, Inc.
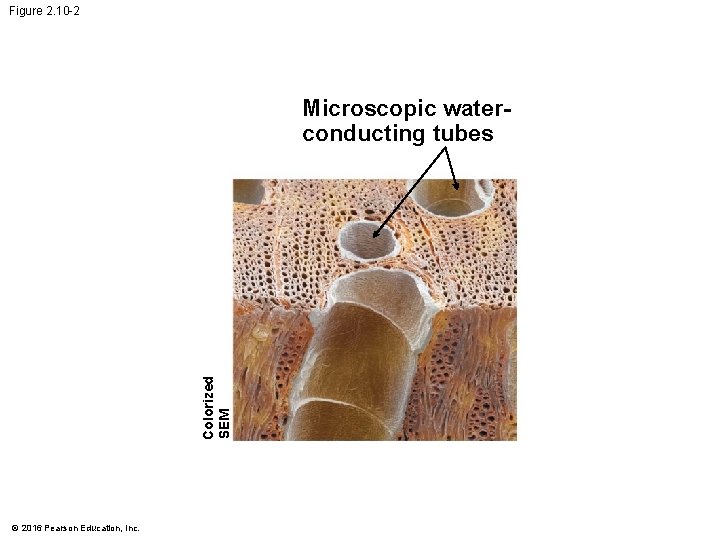
Figure 2. 10 -2 Colorized SEM Microscopic waterconducting tubes © 2016 Pearson Education, Inc.

The Cohesion of Water • Surface tension is a measure of how difficult it is to stretch or break the surface of a liquid. • Hydrogen bonds give water unusually high surface tension and make water behave as though it were coated with an invisible film. © 2016 Pearson Education, Inc.

Figure 2. 11 © 2016 Pearson Education, Inc.

How Water Moderates Temperature • Because of hydrogen bonding, water has a stronger resistance to temperature change than most other substances. • When water is heated, the heat energy • first disrupts hydrogen bonds and • then makes water molecules jostle around faster. © 2016 Pearson Education, Inc.

How Water Moderates Temperature • Because heat is first used to break hydrogen bonds rather than raise the temperature, water absorbs and stores a large amount of heat while warming up only a few degrees. • Conversely, when water cools, hydrogen bonds form, a process that releases heat. © 2016 Pearson Education, Inc.

How Water Moderates Temperature • Water can moderate temperatures. • Earth’s giant water supply enables temperatures on the planet to stay within limits that permit life by • storing a huge amount of heat from the sun during warm periods and • giving off heat that warms the air during cold periods. • Evaporative cooling occurs when a substance evaporates and the surface of the liquid remaining behind cools down. © 2016 Pearson Education, Inc.

Figure 2. 12 © 2016 Pearson Education, Inc.

The Biological Significance of Ice Floating • When water molecules get cold enough, they move apart, forming ice. • A chunk of ice floats because it is less dense than the liquid water in which it is floating. © 2016 Pearson Education, Inc.
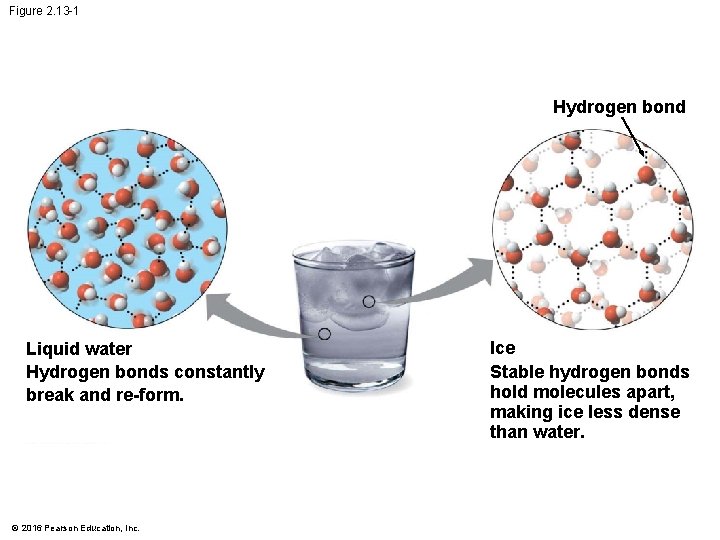
Figure 2. 13 -1 Hydrogen bond Liquid water Hydrogen bonds constantly break and re-form. © 2016 Pearson Education, Inc. Ice Stable hydrogen bonds hold molecules apart, making ice less dense than water.

Figure 2. 13 -2 © 2016 Pearson Education, Inc.

The Biological Significance of Ice Floating • When a deep body of water cools and a layer of ice forms on top, the floating ice acts as an insulating “blanket” over the liquid water, allowing life to persist under the frozen surface. • If ice did not float, ponds, lakes, and even the oceans would freeze solid. © 2016 Pearson Education, Inc.

Water as the Solvent of Life • A solution is a liquid consisting of a homogeneous mixture of two or more substances. • The dissolving agent is the solvent. • The dissolved substance is the solute. • When water is the solvent, the result is an aqueous solution. © 2016 Pearson Education, Inc.

Water as the Solvent of Life • Water can dissolve an enormous variety of solutes necessary for life, providing a medium for chemical reactions. • When water dissolves salt ions, each ion becomes surrounded by oppositely charged regions of water molecules. © 2016 Pearson Education, Inc.
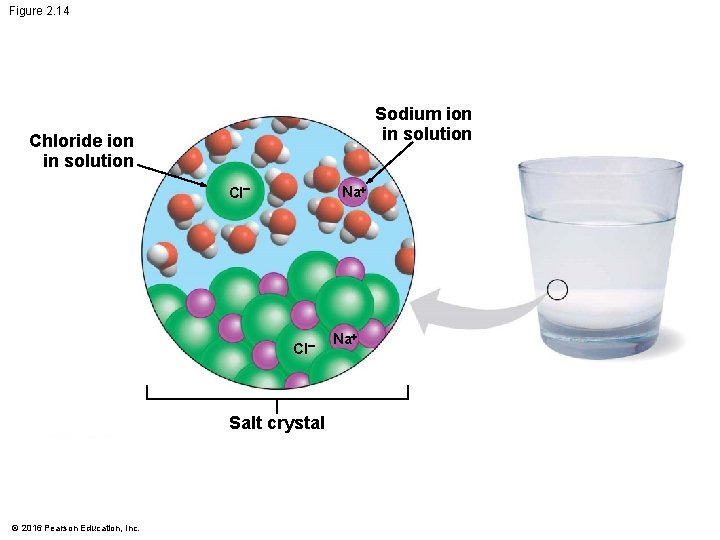
Figure 2. 14 Sodium ion in solution Chloride ion in solution Cl Na+ Cl Salt crystal © 2016 Pearson Education, Inc. Na+

Acids, Bases, and p. H • A chemical compound that releases H+ to a solution is an acid. • A compound that accepts H+ and removes them from solution is a base. • To describe the acidity of a solution, chemists use the p. H scale, a measure of the hydrogen ion (H+) concentration in a solution. • The scale ranges from 0 (most acidic) to 14 (most basic) • Each p. H unit represents a tenfold change in the concentration of H+. © 2016 Pearson Education, Inc.
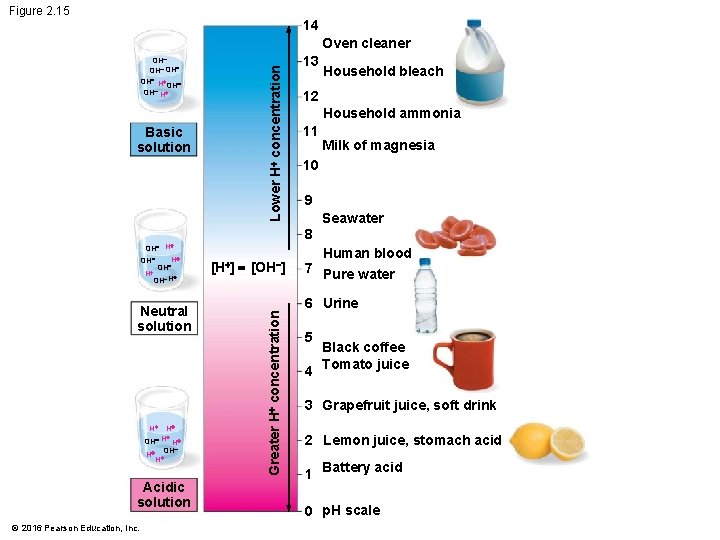
Figure 2. 15 14 Basic solution Lower H+ concentration Oven cleaner OH OH H+ Neutral solution H+ H+ + OH H H+ +OH H Acidic solution © 2016 Pearson Education, Inc. [H+] = [OH ] 12 Household ammonia 11 Milk of magnesia 10 9 Seawater Human blood 7 Pure water 6 Urine Greater H+ concentration H+ OH H Household bleach 8 OH H+ OH 13 5 Black coffee 4 Tomato juice 3 Grapefruit juice, soft drink 2 Lemon juice, stomach acid 1 Battery acid 0 p. H scale
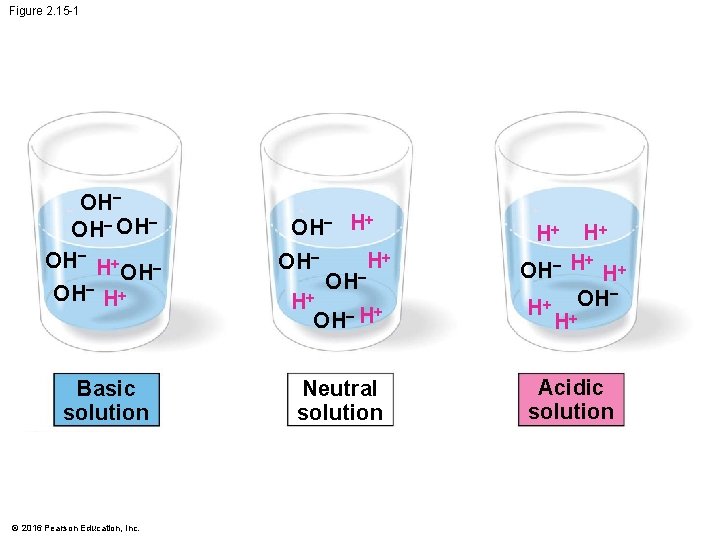
Figure 2. 15 -1 OH OH H+ Basic solution © 2016 Pearson Education, Inc. + OH H OH H+ + H OH H+ + H OH H+ OH + H Neutral solution Acidic solution OH H+ H+

Acids, Bases, and p. H • Buffers are substances that minimize changes in p. H. Buffers • accept H+ when they are in excess and • donate H+ when they are depleted. • When CO 2 dissolves in seawater, it reacts with water to form an acid, which • lowers ocean p. H and • can greatly change marine environments. © 2016 Pearson Education, Inc.
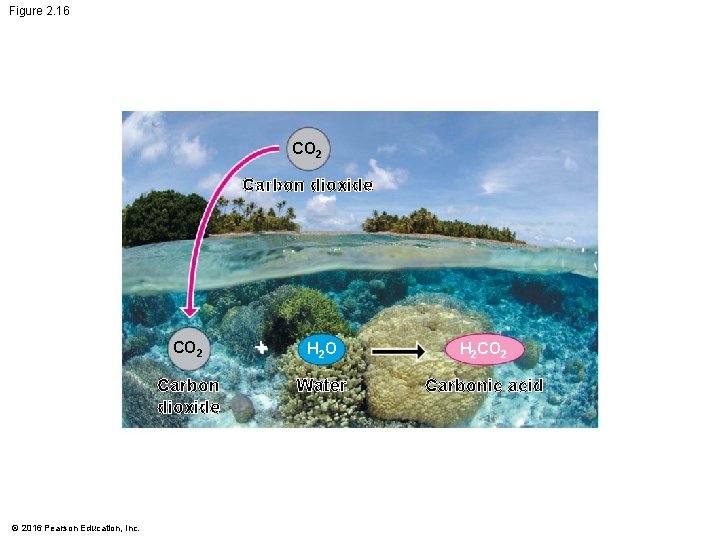
Figure 2. 16 CO 2 Carbon dioxide © 2016 Pearson Education, Inc. + H 2 O H 2 CO 2 Water Carbonic acid

Evolution Connection: Radioactivity as an Evolutionary Clock • The natural process of radioactive decay can be used to obtain important data about the evolutionary history of life on Earth. • Fossils are reliable chronological records of life because we can determine their ages through radiometric dating, which is based on the decay of radioactive isotopes. © 2016 Pearson Education, Inc.
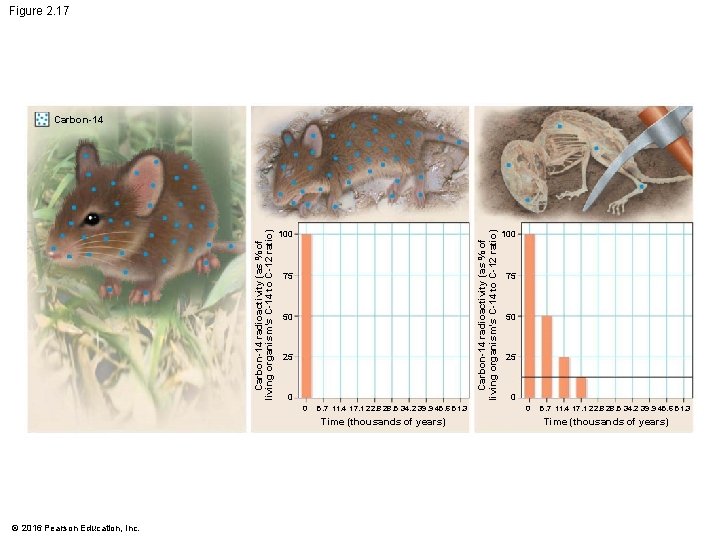
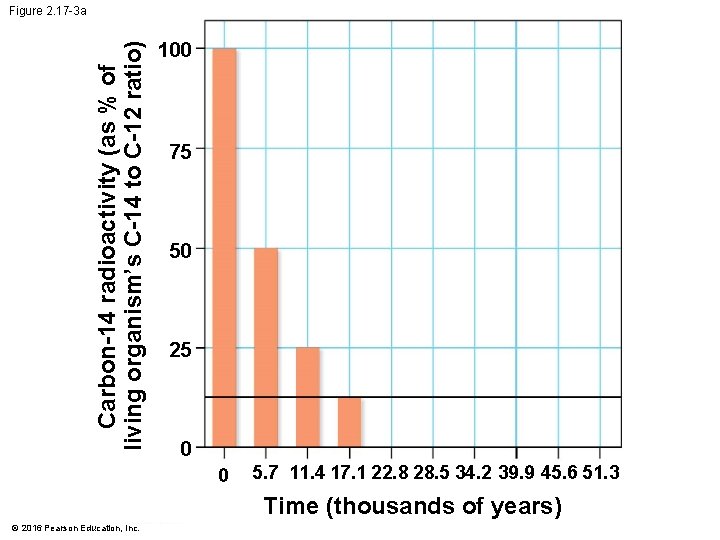
Figure 2. 17 100 Carbon-14 radioactivity (as % of living organism’s C-14 to C-12 ratio) Carbon-14 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years) © 2016 Pearson Education, Inc. 100 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years)

Figure 2. 17 -1 Carbon-14 © 2016 Pearson Education, Inc.
Carbon-14 radioactivity (as % of living organism’s C-14 to C-12 ratio) Figure 2. 17 -2 100 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years) © 2016 Pearson Education, Inc.
Carbon-14 radioactivity (as % of living organism’s C-14 to C-12 ratio) Figure 2. 17 -2 a 100 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years) © 2016 Pearson Education, Inc.
Carbon-14 radioactivity (as % of living organism’s C-14 to C-12 ratio) Figure 2. 17 -3 100 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years) © 2016 Pearson Education, Inc.
Carbon-14 radioactivity (as % of living organism’s C-14 to C-12 ratio) Figure 2. 17 -3 a 100 75 50 25 0 0 5. 7 11. 4 17. 1 22. 8 28. 5 34. 2 39. 9 45. 6 51. 3 Time (thousands of years) © 2016 Pearson Education, Inc.

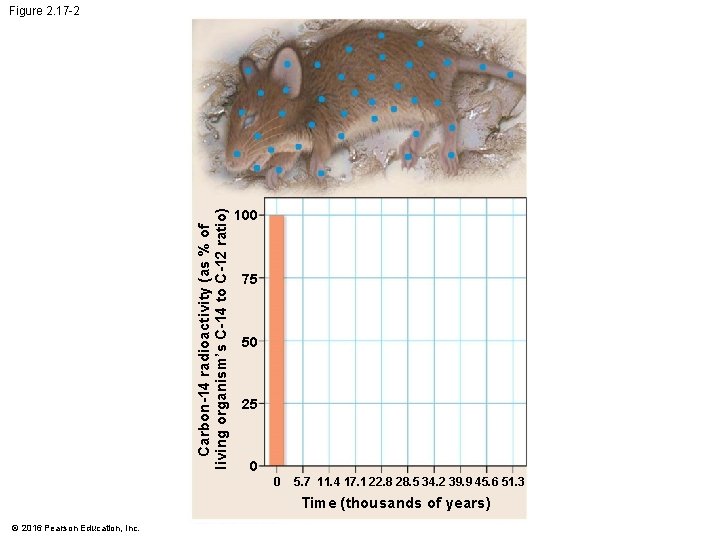
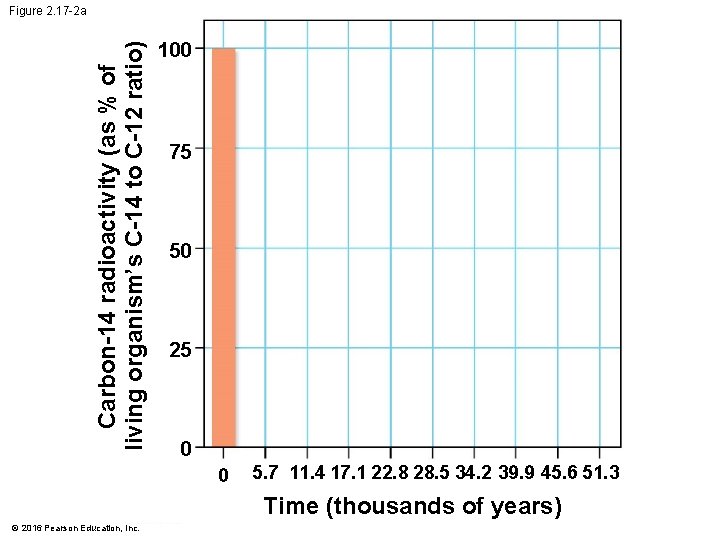
Evolution Connection: Radioactivity as an Evolutionary Clock • Carbon-14 • is present in trace amounts in the environment and • is a radioactive isotope with a half-life of 5, 700 years. © 2016 Pearson Education, Inc.

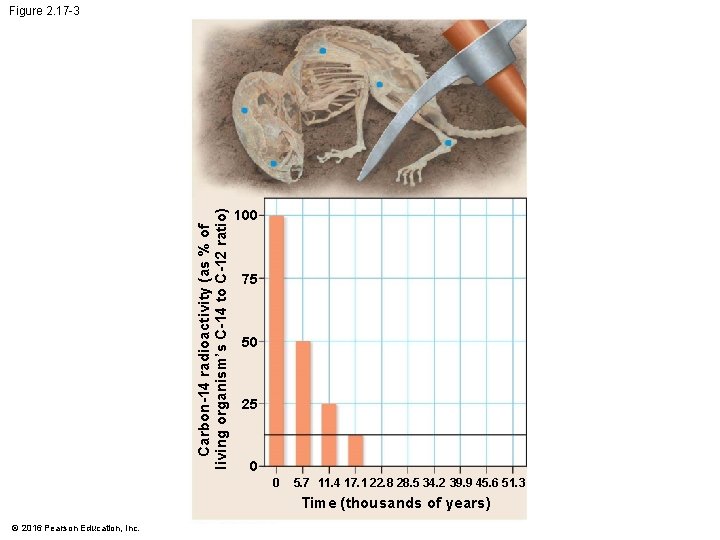
Evolution Connection: Radioactivity as an Evolutionary Clock • A living organism assimilates the different isotopes of an element in proportions that reflect their relative abundances in the environment. • A fossil’s age can be estimated by measuring the ratio of the two isotopes to learn how many half-life reductions have occurred since it died. © 2016 Pearson Education, Inc.

Evolution Connection: Radioactivity as an Evolutionary Clock • Using such techniques, scientists can • estimate the ages of fossils from around the world and • place them in an ordered sequence called the fossil record. © 2016 Pearson Education, Inc.
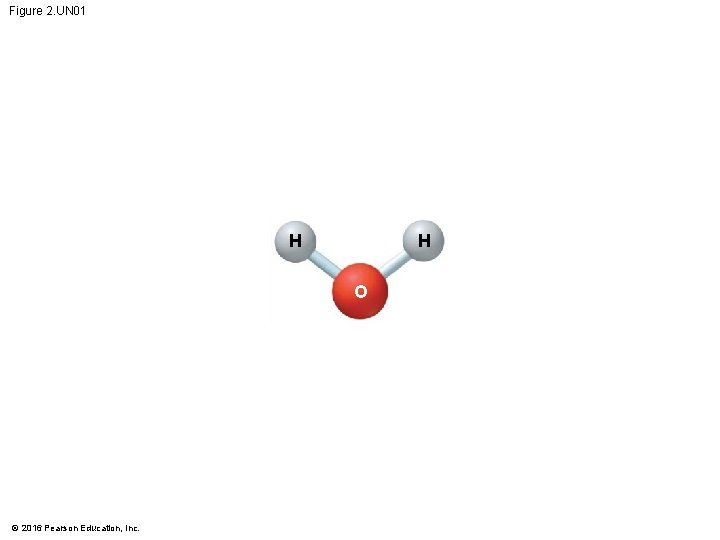
Figure 2. UN 01 H H O © 2016 Pearson Education, Inc.
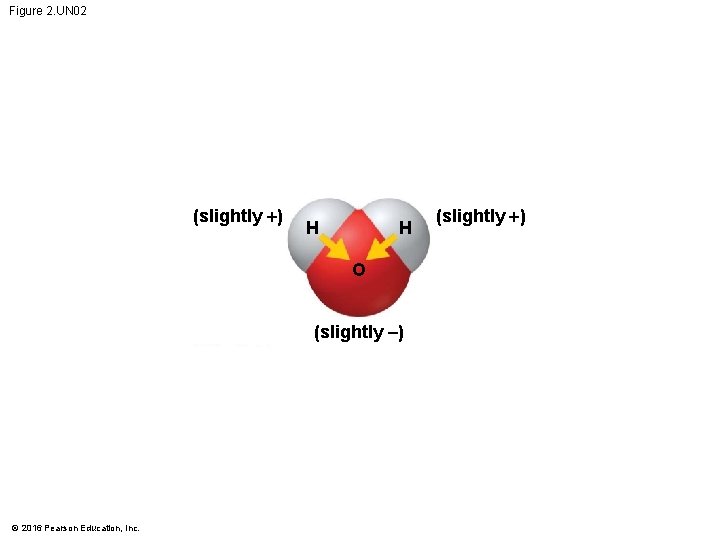
Figure 2. UN 02 (slightly +) H H O (slightly ) © 2016 Pearson Education, Inc. (slightly +)
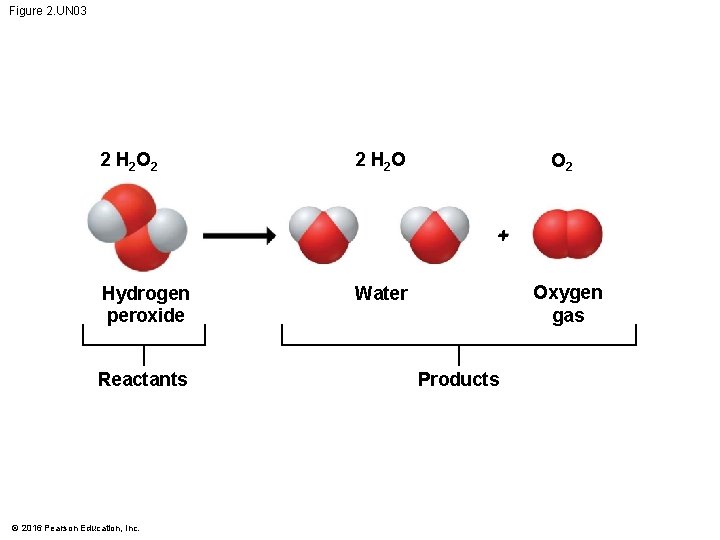
Figure 2. UN 03 2 H 2 O 2 2 H 2 O O 2 + Hydrogen peroxide Reactants © 2016 Pearson Education, Inc. Oxygen gas Water Products
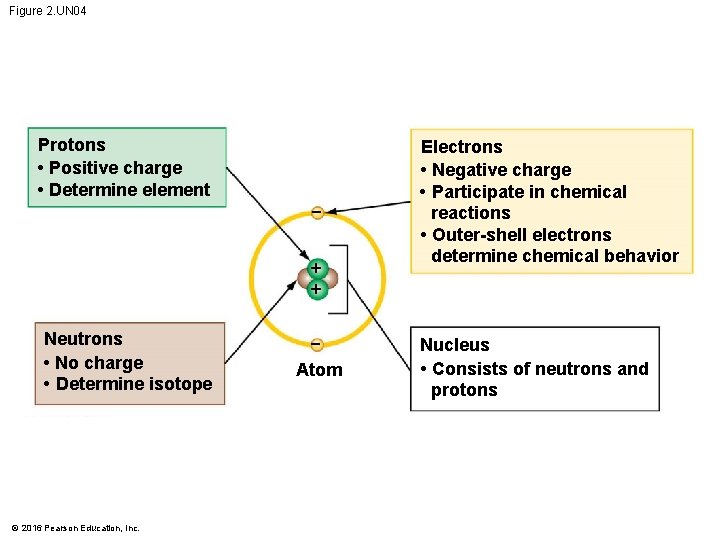
Figure 2. UN 04 Protons • Positive charge • Determine element + + Neutrons • No charge • Determine isotope © 2016 Pearson Education, Inc. Atom Electrons • Negative charge • Participate in chemical reactions • Outer-shell electrons determine chemical behavior Nucleus • Consists of neutrons and protons
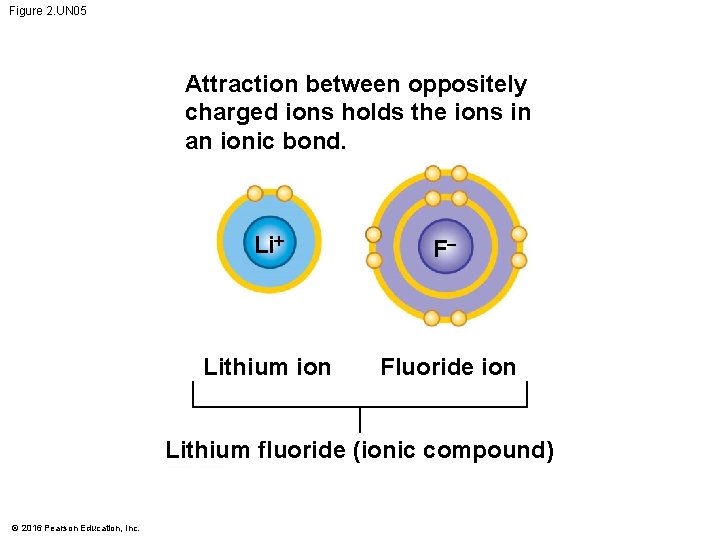
Figure 2. UN 05 Attraction between oppositely charged ions holds the ions in an ionic bond. Li+ F Lithium ion Fluoride ion Lithium fluoride (ionic compound) © 2016 Pearson Education, Inc.
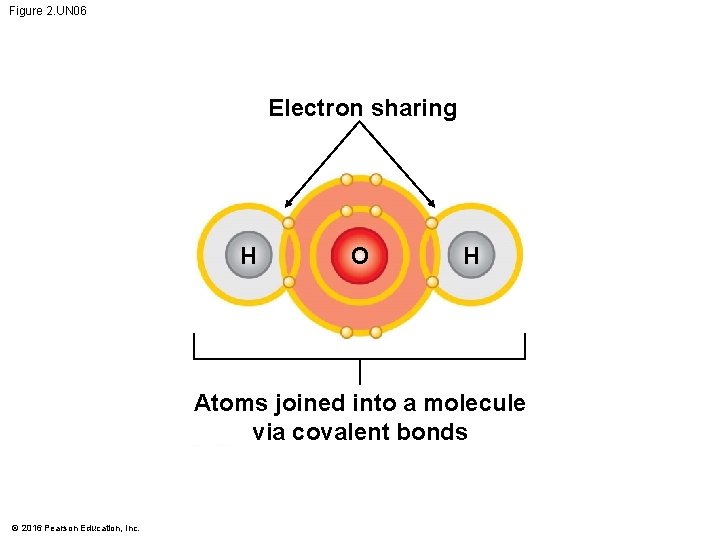
Figure 2. UN 06 Electron sharing H O H Atoms joined into a molecule via covalent bonds © 2016 Pearson Education, Inc.
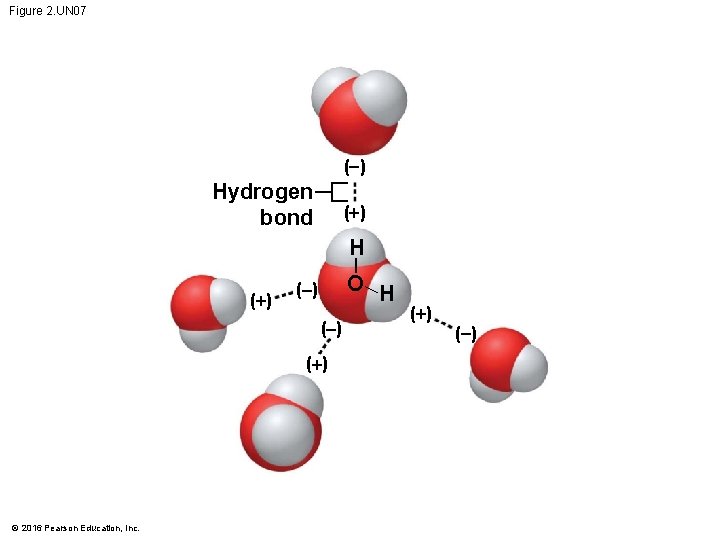
Figure 2. UN 07 ( ) Hydrogen bond (+) H (+) O H ( ) (+) © 2016 Pearson Education, Inc. (+) ( )
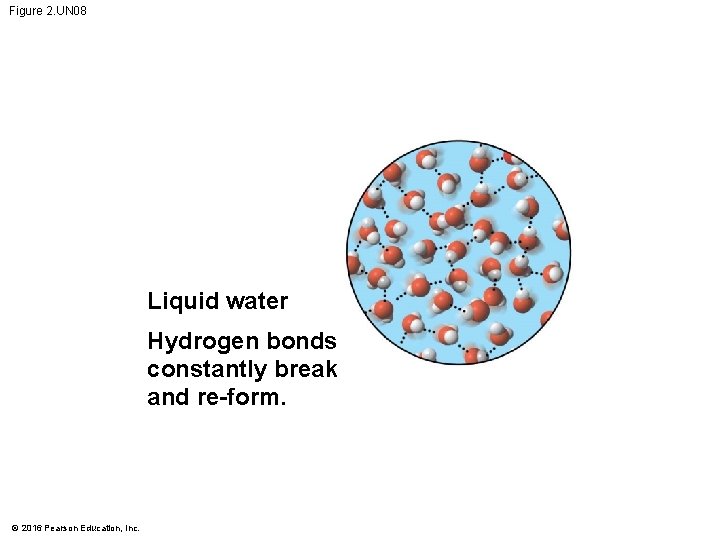
Figure 2. UN 08 Liquid water Hydrogen bonds constantly break and re-form. © 2016 Pearson Education, Inc.
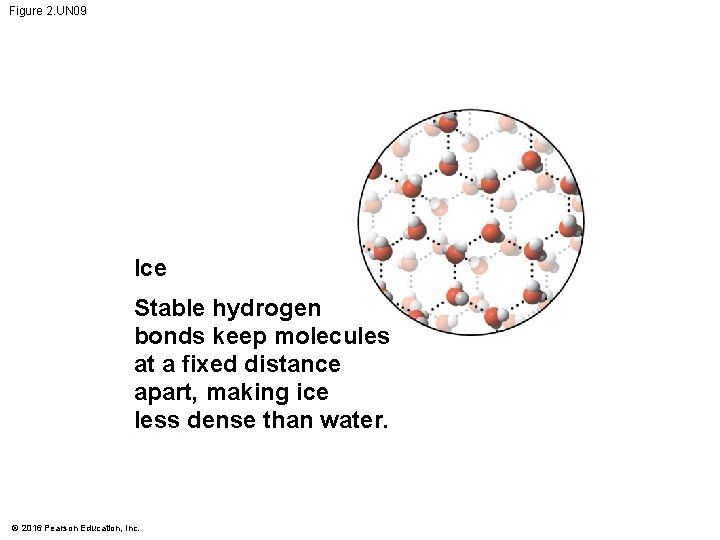
Figure 2. UN 09 Ice Stable hydrogen bonds keep molecules at a fixed distance apart, making ice less dense than water. © 2016 Pearson Education, Inc.
![Figure 2. UN 10 Basic Neutral [H+] = [OH ] Acidic p. H scale Figure 2. UN 10 Basic Neutral [H+] = [OH ] Acidic p. H scale](http://slidetodoc.com/presentation_image_h2/722ea862a234468881976f0e21820e93/image-103.jpg)
Figure 2. UN 10 Basic Neutral [H+] = [OH ] Acidic p. H scale © 2016 Pearson Education, Inc. p. H 14 Lower H+ concentration p. H 7 H+ concentration equal to OH concentration Greater H+ concentration p. H 0
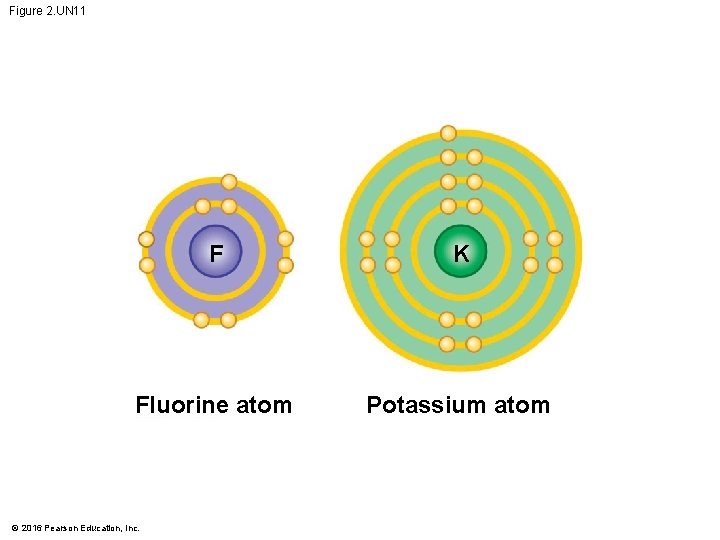
Figure 2. UN 11 F K Fluorine atom Potassium atom © 2016 Pearson Education, Inc.
- Slides: 104