CHAPTER 2 Essential Chemistry for Biology Power Point

CHAPTER 2 Essential Chemistry for Biology Power. Point® Lectures for Essential Biology, Third Edition – Neil Campbell, Jane Reece, and Eric Simon Essential Biology with Physiology, Second Edition – Neil Campbell, Jane Reece, and Eric Simon Lectures by Chris C. Romero Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings





Biology and Society: Aids: A Matter of Chemistry • In 1981, the Centers for Disease Control (CDC) – Began tracking high numbers of otherwise healthy young men who were coming down with extremely rare diseases. • Examinations of these patients showed that they all had a failing immune system. – This condition was caused by the human immunodeficiency virus (HIV). Copyright © 2007 Pearson Education, Inc. publishing as Pearson Benjamin Cummings

• HIV binds to certain molecules found on cells of the immune system called “helper” T cells. – The virus kills these cells and wipes out one part of the body’s defenses. HIV Reproductive Cycle Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
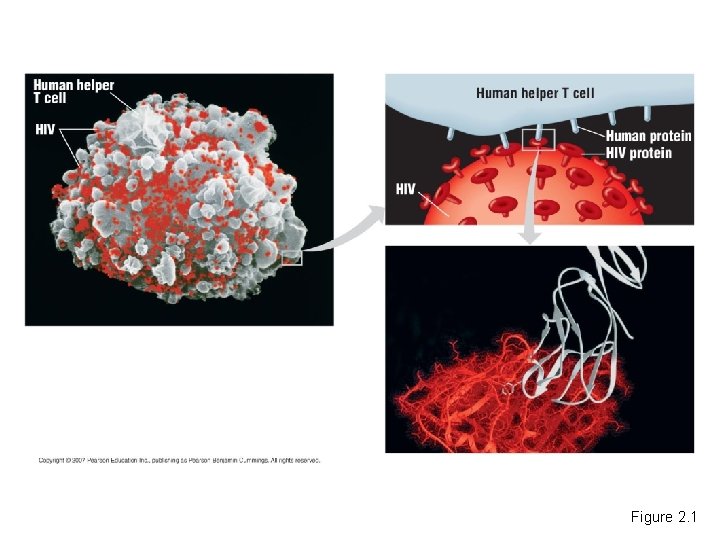
Figure 2. 1

Some Basic Chemistry • Take any biological system apart, and you eventually end up at the chemical level. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Matter: Elements and Compounds • Matter is anything that occupies space and has mass. • Matter is found on the Earth in three physical states: – Solid – Liquid – Gas Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Matter is composed of chemical elements. – Elements are substances that cannot be broken down into other substances. – There are 92 naturally occurring elements on Earth. • All the elements are listed in the periodic table. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
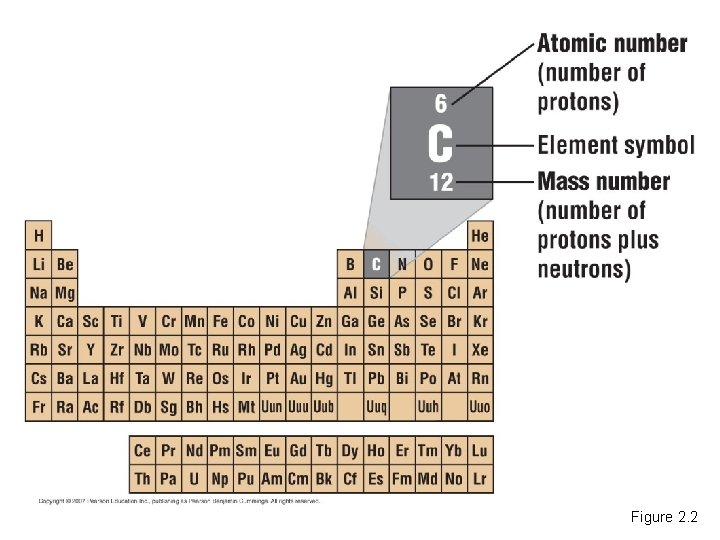
Figure 2. 2

• Twenty-five elements are essential to life. – Four of these make up about 96% of the weight of the human body. – Trace elements occur in smaller amounts. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
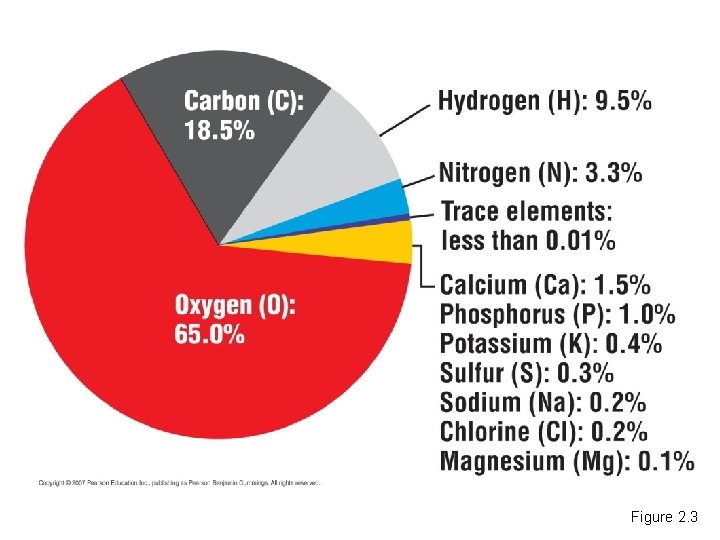
Figure 2. 3

• Trace elements are essential for life. – An iodine deficiency causes goiter. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
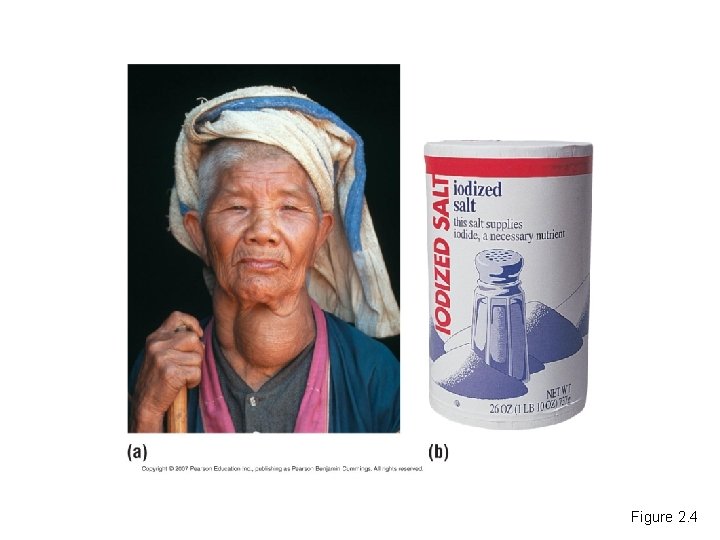
Figure 2. 4

• Elements can combine to form compounds. – These are substances that contain two or more elements in a fixed ratio. – Example: Na. Cl (salt) Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Atoms • Each element consists of one kind of atom. – An atom is the smallest unit of matter that still retains the properties of an element. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
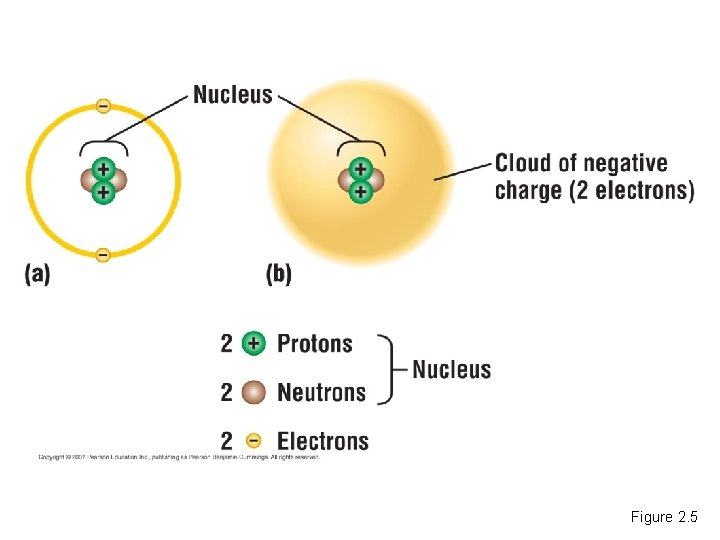
Figure 2. 5

The Structure of Atoms • Atoms are composed of subatomic particles. – A proton is positively charged. – An electron is negatively charged. – A neutron is electrically neutral. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Most atoms have protons and neutrons packed tightly into the nucleus. – The nucleus is the atom’s central core. – The electrons orbit the nucleus. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Elements differ in the number of subatomic particles in their atoms. – The number of protons, the atomic number, determines which element it is. – An atom’s mass number is the sum of the number of protons and neutrons. – Mass is a measure of the amount of matter in an object. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Isotopes • Isotopes are alternate mass forms of an element. – They have the same number of protons and electrons. – But they have a different number of neutrons. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Table 2. 1

• In radioactive isotopes, – The nucleus decays, giving off particles and energy. • Radioactive isotopes have many uses in research and medicine. – Example: PET scans Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Uncontrolled exposure to radioactive isotopes can harm living organisms by damaging DNA. – Example: the 1999 Chernobyl nuclear accident Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

The Process of Science: Can Alzheimer’s Disease Be Detected Early? • Alzheimer’s disease is a devastating illness. – It gradually destroys a person’s memory and ability to think. Copyright © 2007 Pearson Education, Inc. publishing as Pearson Benjamin Cummings

• Doctors are now using PET scans – In an attempt to diagnose Alzheimer’s disease before the onset of symptoms. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
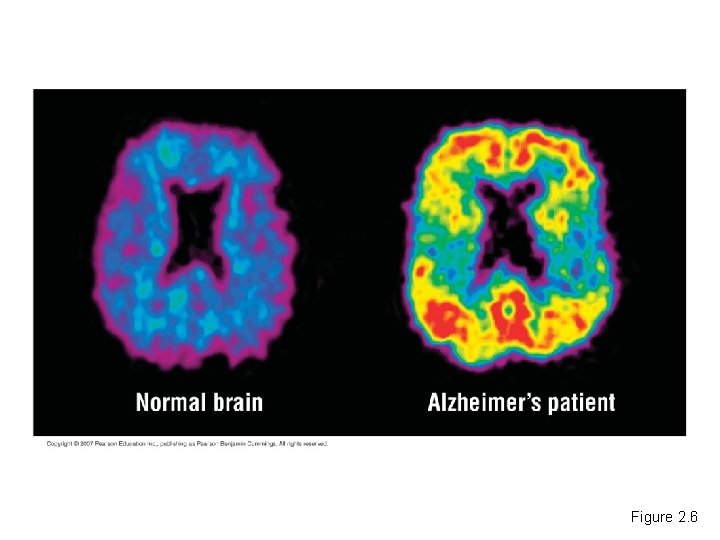
Figure 2. 6

Electron Arrangement and the Chemical Properties of Atoms • Electrons determine how an atom behaves when it encounters other atoms. • Electrons orbit the nucleus of an atom in specific electron shells. – The number of electrons in the outermost shell determines the chemical properties of an atom. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
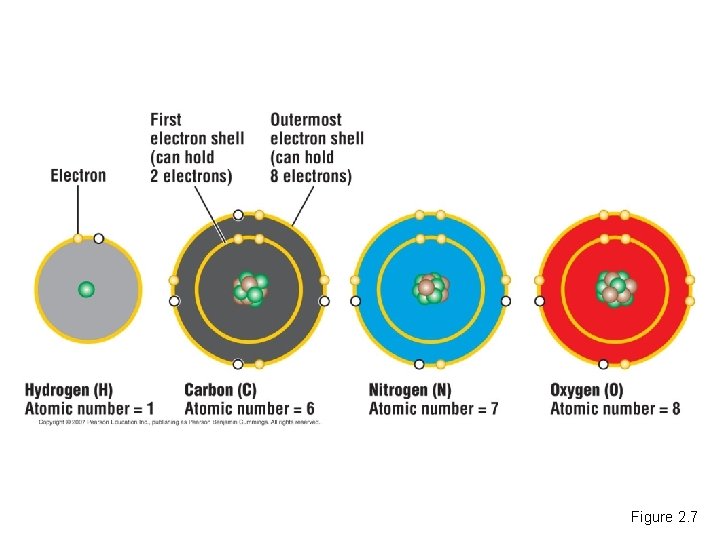
Figure 2. 7

Chemical Bonding and Molecules • Chemical reactions enable atoms to give up or acquire electrons in order to complete their outer shells. – These interactions usually result in atoms staying close together. – The atoms are held together by chemical bonds. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Ionic Bonds • When an atom loses or gains electrons, it becomes electrically charged. – Charged atoms are called ions. – Ionic bonds are formed between oppositely charged ions. Ionic Bonds Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
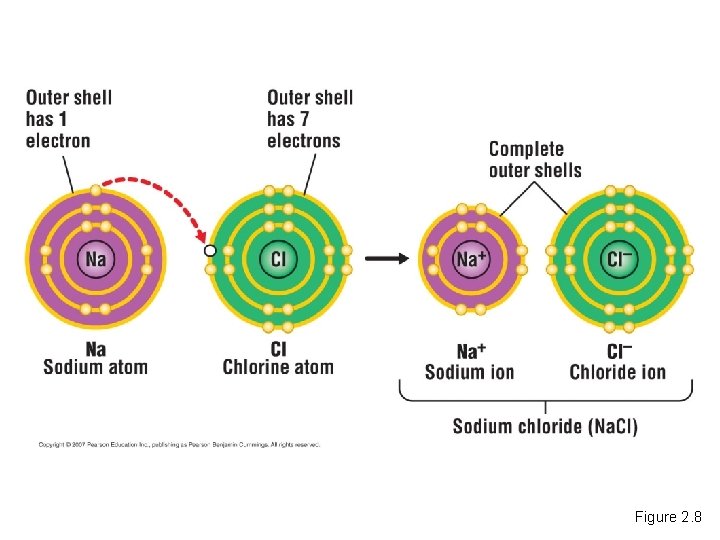
Figure 2. 8

Covalent Bonds • A covalent bond forms when two atoms share one or more pairs of outer-shell electrons. Covalent Bonds Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
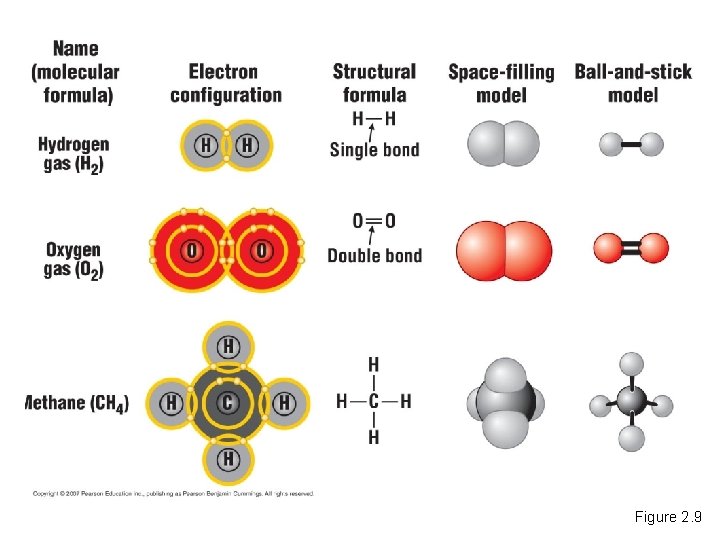
Figure 2. 9

Hydrogen Bonds • Water is a compound in which the electrons in its covalent bonds are shared unequally. – This causes it to be a polar molecule, one with opposite charges on opposite ends. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
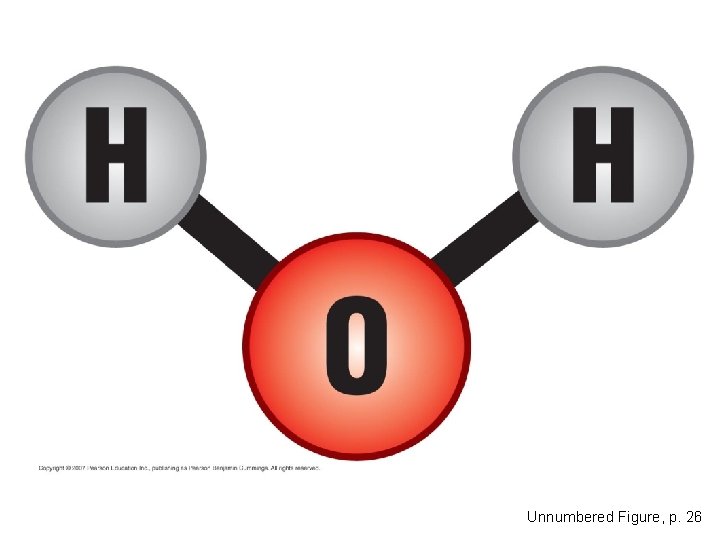
Unnumbered Figure, p. 26

• The polarity of water results in weak electrical attractions between neighboring water molecules. – These interactions are called hydrogen bonds. Water Structure Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
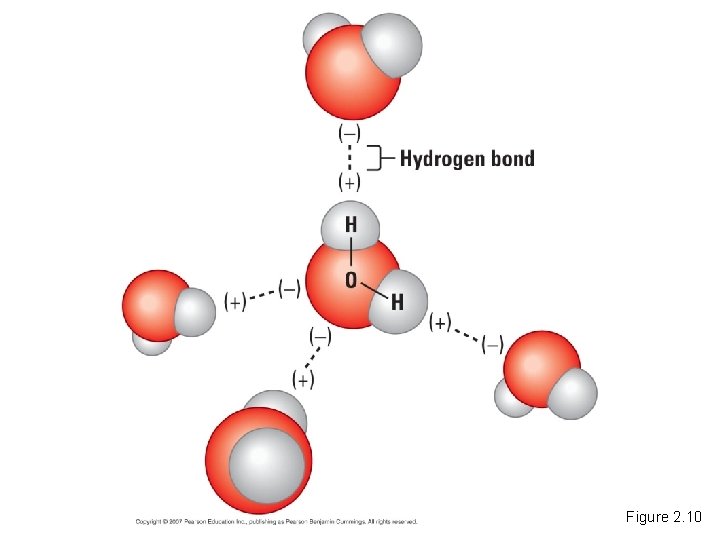
Figure 2. 10

Chemical Reactions • Cells constantly rearrange molecules by breaking existing chemical bonds and forming new ones. – Such changes in the chemical composition of matter are called chemical reactions. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
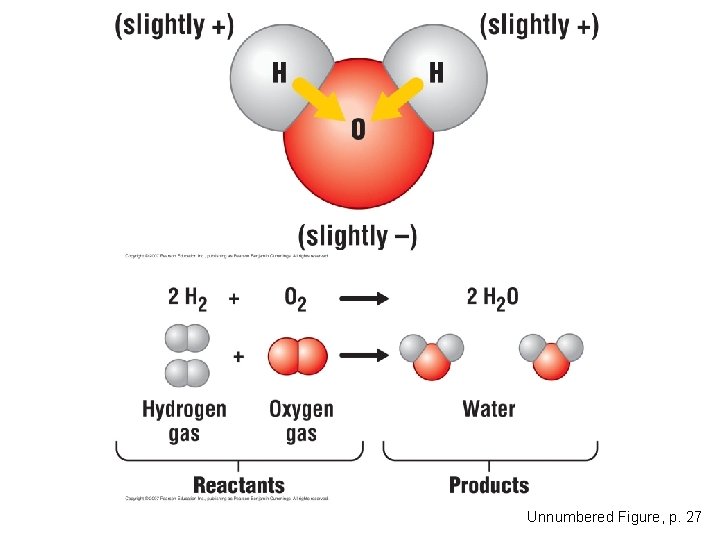
Unnumbered Figure, p. 27

• Chemical reactions include: – Reactants, the starting materials – Products, the end materials Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Chemical reactions cannot create or destroy matter, – They only rearrange it. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Water and Life • Life on Earth began in water and evolved there for 3 billion years. – Modern life still remains tied to water. – Your cells are composed of 70%– 95% water. • The abundance of water is a major reason Earth is habitable. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 11

Water’s Life-Supporting Properties • The polarity of water molecules and the hydrogen bonding that results explain most of water’s lifesupporting properties: – Water’s cohesive nature – Water’s ability to moderate temperature – Floating ice – Versatility of water as a solvent Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

The Cohesion of Water • Water molecules stick together as a result of hydrogen bonding. – This is called cohesion. – Cohesion is vital for water transport in plants. Water Transport Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 12

• Surface tension is the measure of how difficult it is to stretch or break the surface of a liquid. – Hydrogen bonds give water an unusually high surface tension. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 13

How Water Moderates Temperature • Because of hydrogen bonding, water has a strong resistance to temperature change. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Heat and temperature are related, but different. – Heat is the amount of energy associated with the movement of the atoms and molecules in a body of matter. – Temperature measures the intensity of heat. • Water can absorb and store large amounts of heat while only changing a few degrees in temperature. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Water can moderate temperatures. – Earth’s giant water supply causes temperatures to stay within limits that permit life. – Evaporative cooling removes heat from the Earth and from organisms. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 14

The Biological Significance of Ice Floating • When water molecules get cold, they move apart, forming ice. – A chunk of ice has fewer molecules than an equal volume of liquid water. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• The density of ice is lower than liquid water. – This is why ice floats. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
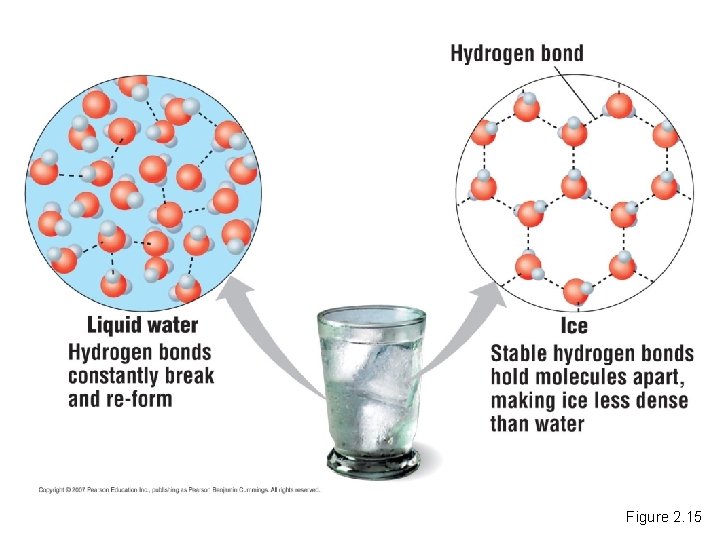
Figure 2. 15

• Since ice floats, ponds, lakes, and even the oceans do not freeze solid. – Marine life could not survive if bodies of water froze solid. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Water as the Solvent of Life • A solution is a liquid consisting of two or more substances evenly mixed. – The dissolving agent is called the solvent. – The dissolved substance is called the solute. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
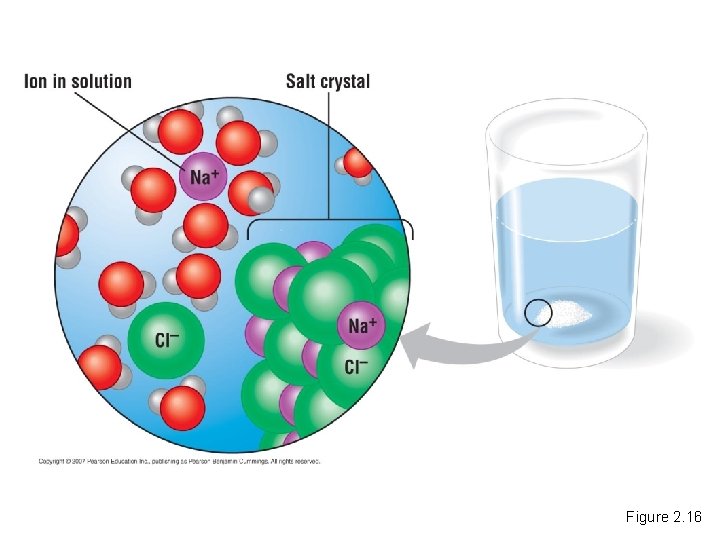
Figure 2. 16

• When water is the solvent, the result is an aqueous solution. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Acids, Bases, and p. H • Acid – A chemical compound that donates H+ ions to solutions. • Base – A compound that accepts H+ ions and removes them from solution. • To describe the acidity of a solution, we use the p. H scale. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings
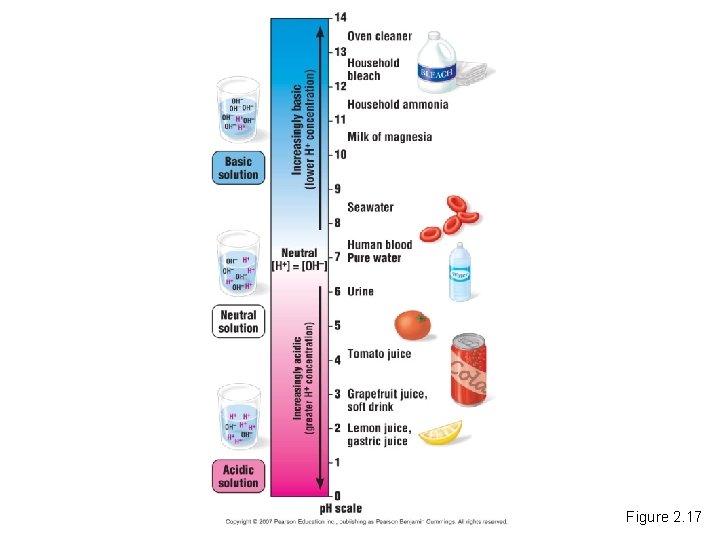
Figure 2. 17

• Buffers are substances that resist p. H change. – They accept H+ ions when they are in excess. – They donate H+ ions when they are depleted. • Buffering is not foolproof. – Example: acid precipitation Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 18

Evolution Connection: Earth Before Life • Chemical reactions and physical processes on the early Earth created an environment that made life possible. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• Earth began as a cold world about 4. 5 billion years ago. • The planet eventually melted from heat produced by: – Compaction – Radioactive decay – Impact of meteorites Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

• The first atmosphere was probably composed of hot hydrogen gas. – This gas escaped because the gravity of Earth was not strong enough. • A new atmosphere was formed from the gases belched from volcanoes. Copyright © 2007 Pearson Education Inc. , publishing as Pearson Benjamin Cummings

Figure 2. 19
- Slides: 70