Chapter 2 Essential Chemistry for Biology Power Point

Chapter 2 Essential Chemistry for Biology Power. Point® Lectures for Campbell Essential Biology, Fifth Edition, and Campbell Essential Biology with Physiology, Fourth Edition – Eric J. Simon, Jean L. Dickey, and Jane B. Reece Lectures by Edward J. Zalisko © 2013 Pearson Education, Inc.

Biology and Society: More Precious than Gold • A drought is a period of abnormally dry weather that changes the environment and one of the most devastating disasters. • Droughts can cause • severe crop damage, shortages of drinking water, • dust storms, famine, habitat loss, and mass migration. • Throughout human history, droughts have helped wipe out societies and even whole civilizations. • Droughts are catastrophic because life cannot exist without water. © 2013 Pearson Education, Inc.

Matter: Elements and Compounds • Matter is anything that occupies space and has mass. – found on the Earth in three physical states: Solid, Liquid and Gas © 2013 Pearson Education, Inc.

Matter: Elements and Compounds • Matter is composed of chemical elements: . – substances that cannot be broken down into other substances. – There are 92 naturally occurring elements on Earth. – All of the elements are listed in the periodic table. – Twenty-five are essential to life. – Four make up about 96% of the weight of the human body – CHON: Carbon, Hydrogen, Oxygen and Nitrog © 2013 Pearson Education, Inc.
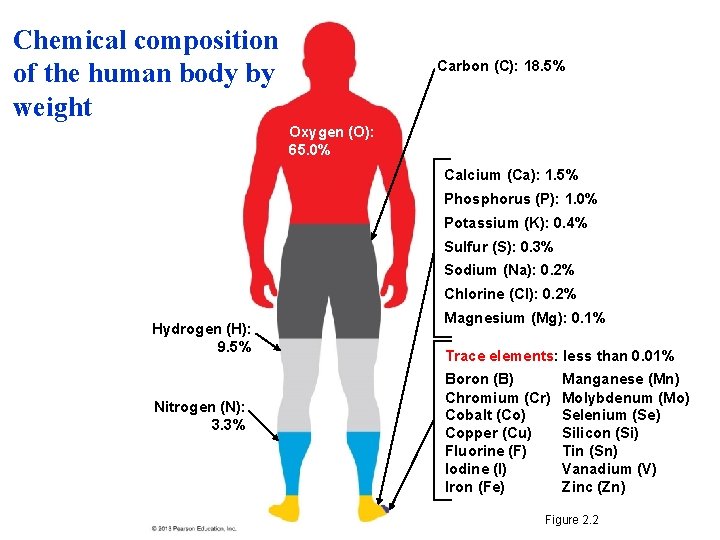
Chemical composition of the human body by weight Carbon (C): 18. 5% Oxygen (O): 65. 0% Calcium (Ca): 1. 5% Phosphorus (P): 1. 0% Potassium (K): 0. 4% Sulfur (S): 0. 3% Sodium (Na): 0. 2% Chlorine (Cl): 0. 2% Hydrogen (H): 9. 5% Nitrogen (N): 3. 3% Magnesium (Mg): 0. 1% Trace elements: less than 0. 01% Boron (B) Chromium (Cr) Cobalt (Co) Copper (Cu) Fluorine (F) Iodine (I) Iron (Fe) Manganese (Mn) Molybdenum (Mo) Selenium (Se) Silicon (Si) Tin (Sn) Vanadium (V) Zinc (Zn) Figure 2. 2
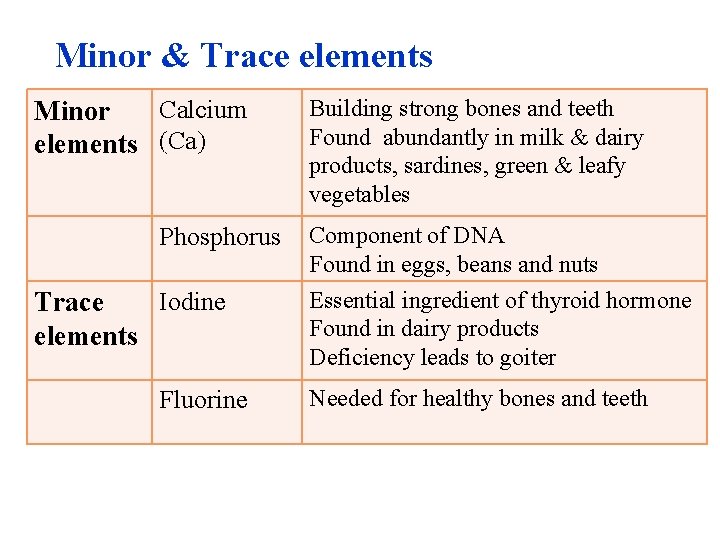
Minor & Trace elements Calcium Minor elements Ca Phosphorus Iodine Trace elements Fluorine Building strong bones and teeth Found abundantly in milk & dairy products, sardines, green & leafy vegetables Component of DNA Found in eggs, beans and nuts Essential ingredient of thyroid hormone Found in dairy products Deficiency leads to goiter Needed for healthy bones and teeth

Matter: Elements and Compounds • Trace elements – are required in only very small amounts and – are essential for life. • An iodine deficiency causes goiter. • Fluorine – is added to dental products and drinking water and – helps to maintain healthy bones and teeth. © 2013 Pearson Education, Inc.
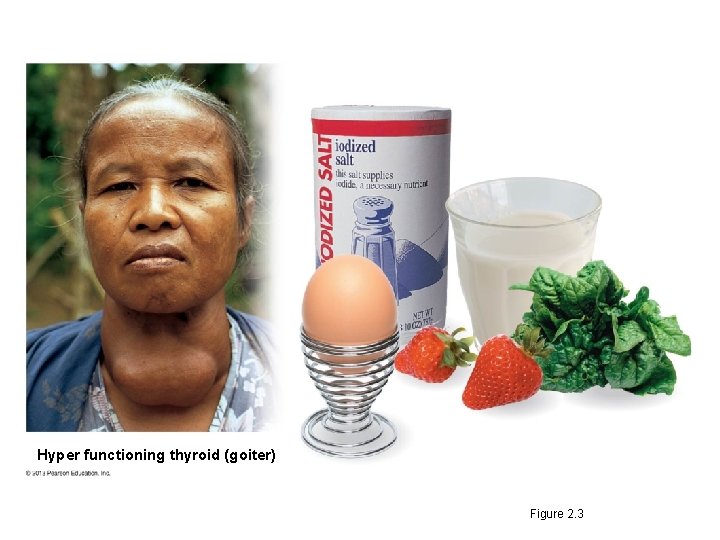
Hyper functioning thyroid (goiter) Figure 2. 3

Matter: Elements, Atoms and Compounds • Each element consists of one kind of atom. – An atom is the smallest unit of matter that still retains the properties of an element. • Elements can combine to form compounds. – Compounds are substances that contain two or more elements in a fixed ratio. – Common compounds include – Na. Cl (table salt) and – H 2 O (water). © 2013 Pearson Education, Inc.
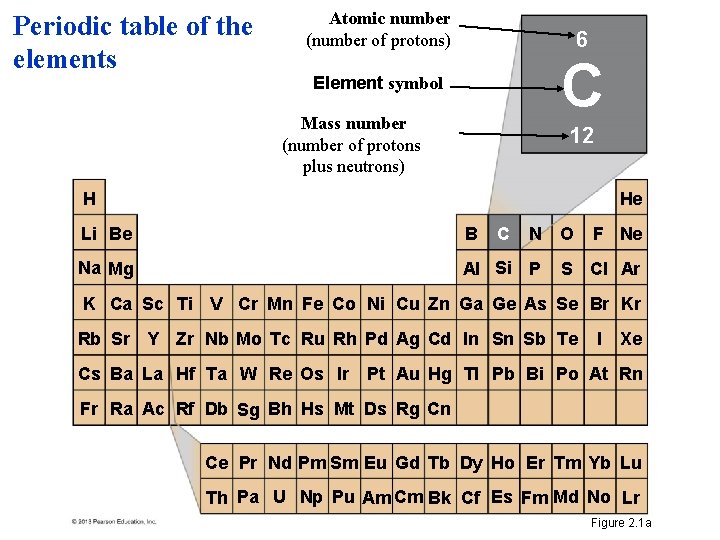
Periodic table of the elements Atomic number (number of protons) 6 C Element symbol Mass number (number of protons plus neutrons) 12 H He Li Be B Na Mg Al Si P C N O F Ne S Cl Ar K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta W Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg Cn Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Figure 2. 1 a
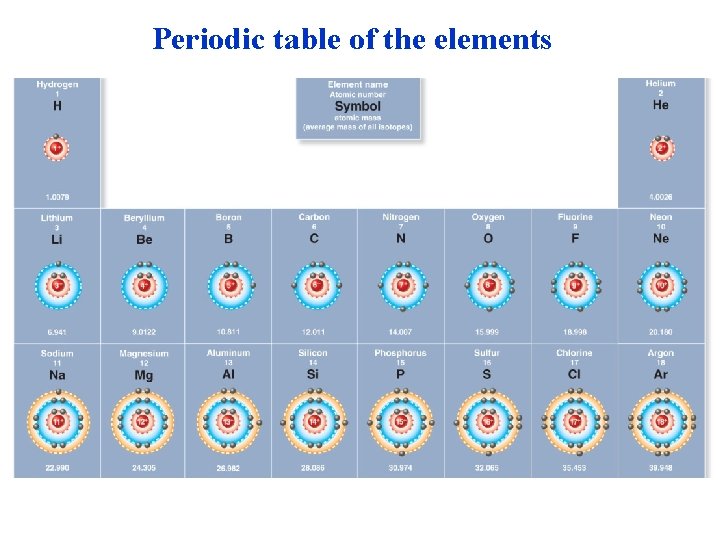
Periodic table of the elements

Practice 1. Ordinary oxygen has an atomic mass of 16 and an atomic number of 8. How many neutrons does this type of oxygen have? 2. Phosphorous has an atomic number of 15 and an atomic weight of 30. 974. How many neutrons does this type of oxygen have? 3. Silicon has an atomic number of 14 and a mass number of 28. How many neutrons are found in silicon?

The Structure of Atoms • Atoms are composed of three subatomic particles – A proton is positively charged, mass of 1 dalton – An element is defined by its number of protons – A neutron is electrically neutral mass of 1 dalton – Neutrons can vary between isotopes – An electron is negatively charged minimal mass 1/2000 th 1 dalton, found outside (orbiting) the nucleus. • Most atoms have protons and neutrons packed tightly into the nucleus. – The nucleus is the atom’s central core. – Electrons orbit the nucleus. © 2013 Pearson Education, Inc. Electron shell or Electron energy level
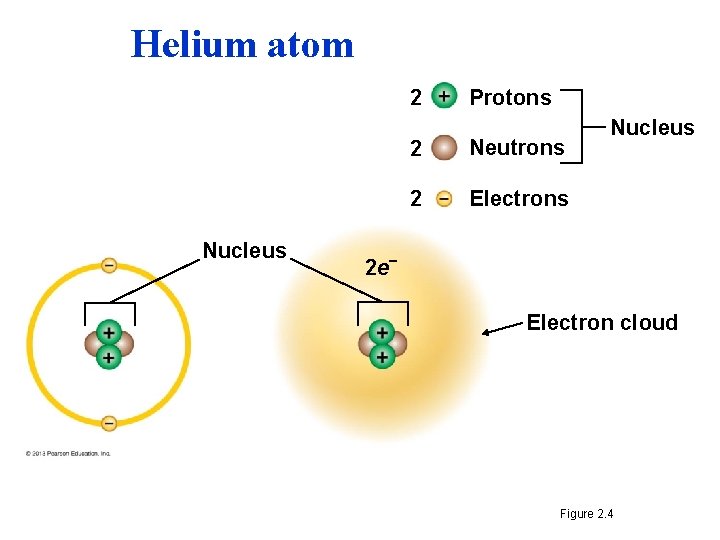
Helium atom 2 Nucleus Protons 2 Neutrons 2 Electrons Nucleus 2 e– Electron cloud Figure 2. 4
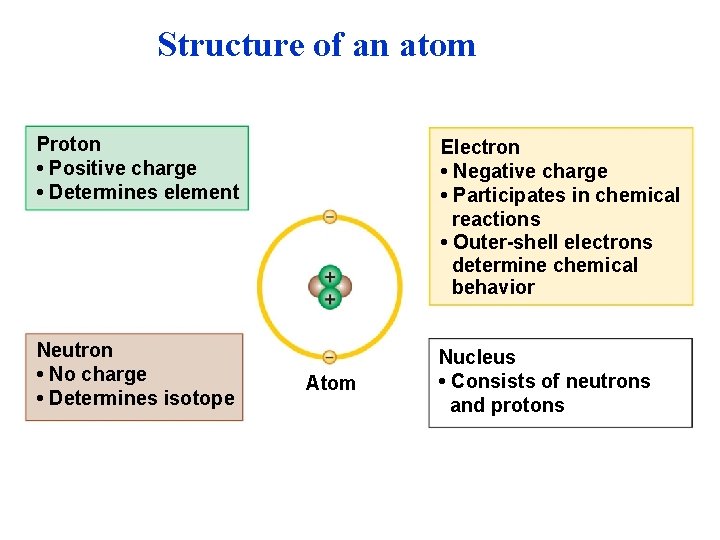
Structure of an atom Proton • Positive charge • Determines element Electron • Negative charge • Participates in chemical reactions • Outer-shell electrons determine chemical behavior Neutron • No charge • Determines isotope Nucleus • Consists of neutrons and protons Atom

Atomic and mass number • Elements differ in the number of subatomic particles in their atoms. – The number of protons, the atomic number, determines which element it is. – Mass is a measure of the amount of material in an object. – An atom’s mass number is the sum of the number of protons and neutrons in its nucleus. © 2013 Pearson Education, Inc.

Isotopes • Isotopes are alternate mass forms of an element. – Isotopes have the same number of protons and electrons, – but they have a different number of neutrons.

Radioactive Isotope • The nucleus of a radioactive isotope decays, giving off particles and energy. • Radioactive isotopes have many uses in research and medicine. – They can be used to determine the fate of atoms in living organisms. – They are used in PET scans to diagnose heart disorders and some cancers. • Uncontrolled exposure to radioactive isotopes can harm living organisms by damaging DNA. – The 1986 Chernobyl nuclear accident released large amounts of radioactive isotopes. – Naturally occurring radon gas may cause lung cancer.
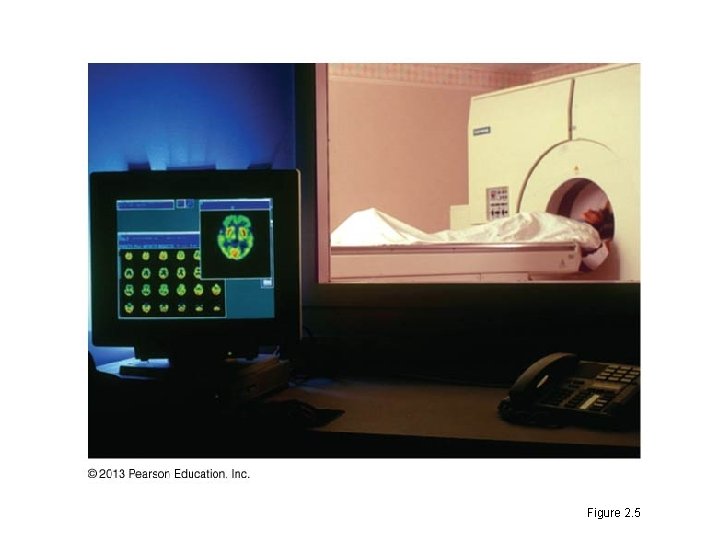
Figure 2. 5

Electron Arrangement and the Chemical Properties of Atoms • Of the three subatomic particles, only electrons are directly involved in the chemical activity of an atom. • Electrons orbit the nucleus of an atom in specific electron shells. • The farther an electron is from the nucleus, the greater its energy. • The number of electrons in the outermost shell (valance) determines the chemical properties of an atom. • Valance electrons determine how an atom behaves when it encounter other atoms © 2013 Pearson Education, Inc.
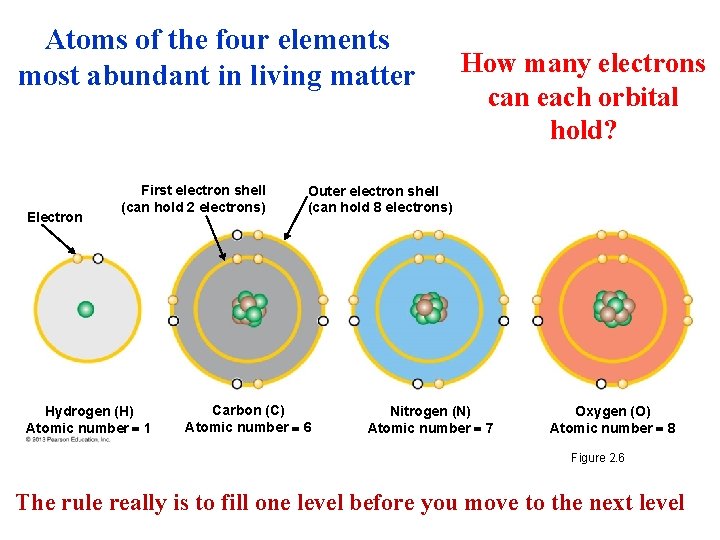
Atoms of the four elements most abundant in living matter Electron First electron shell (can hold 2 electrons) Hydrogen (H) Atomic number 1 How many electrons can each orbital hold? Outer electron shell (can hold 8 electrons) Carbon (C) Atomic number 6 Nitrogen (N) Atomic number 7 Oxygen (O) Atomic number 8 Figure 2. 6 The rule really is to fill one level before you move to the next level

Practice 1. By definition all atoms of carbon have exactly six _______ , but the number of _______ varies from one isotope to another. 2. As two atoms approach each other, which subatomic particles will first come into contacts?

Chemical Bonding and Molecules • In chemical reactivity, a toms tend to - complete a partially filled valence or - empty a partially filled valence • Chemical reactions enable atoms to give up or acquire electrons, in order to complete their outer shells. • Chemical reactions usually result in atoms - staying close together and - being held together by attractions called chemical bonds. • EIGHT is GREAT. Will steal or share electrons to get eight valance electrons © 2013 Pearson Education, Inc.

Chemical bonding between atoms Covalent bonds are formed when atoms share electrons – Non polar covalent bonds = Equal attraction; ie: H 2 – Polar covalent bonds = Assymetric attraction; ie: HO 2 Ionic bonds are attractions between ions of opposite charge – steal electrons from other = Unequal attraction Hydrogen bonds form between a positively charged hydrogen atom and any other negatively charged atom van der Waals interactions form between negatively charged and positively charged regions in molecules.
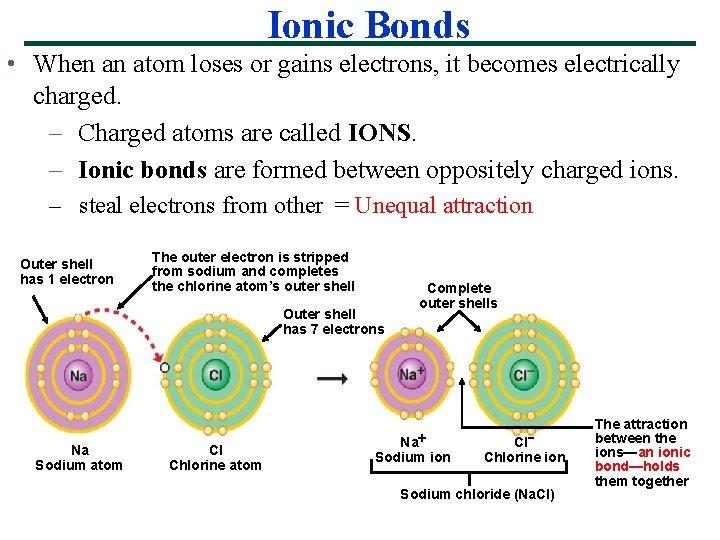
Ionic Bonds • When an atom loses or gains electrons, it becomes electrically charged. – Charged atoms are called IONS. – Ionic bonds are formed between oppositely charged ions. – steal electrons from other = Unequal attraction Outer shell has 1 electron The outer electron is stripped from sodium and completes the chlorine atom’s outer shell Outer shell has 7 electrons Na Sodium atom Cl Chlorine atom Complete outer shells Na Sodium ion Cl Chlorine ion Sodium chloride (Na. Cl) The attraction between the ions—an ionic bond—holds them together
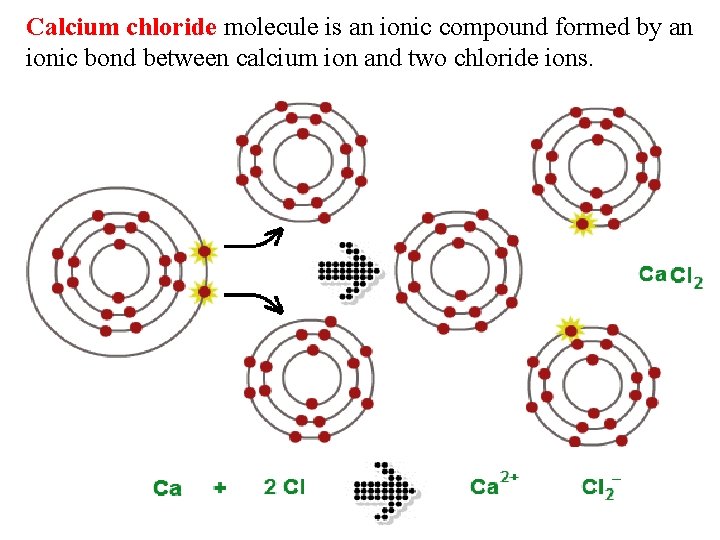
Calcium chloride molecule is an ionic compound formed by an ionic bond between calcium ion and two chloride ions.

Covalent Bonds • A covalent bond forms when two atoms share one or more pairs of outer-shell electrons. – Non polar covalent bonds = Equal attraction (H 2) – Polar covalent bonds = Asymmetric attraction (HO 2 • Covalent bonds are the strongest of the various bonds. • Covalent bonds hold atoms together in a molecule. • The number of covalent bonds an atom can form is equal to the number of additional electrons needed to fill its outer shell. © 2013 Pearson Education, Inc.
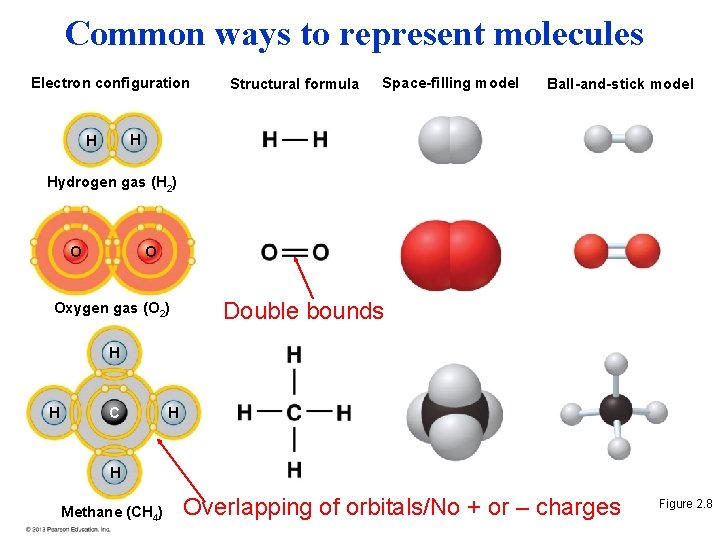
Common ways to represent molecules Electron configuration Structural formula Space-filling model Ball-and-stick model H H Hydrogen gas (H 2) O O Oxygen gas (O 2) Double bounds H H C H H Methane (CH 4) Overlapping of orbitals/No + or – charges Figure 2. 8
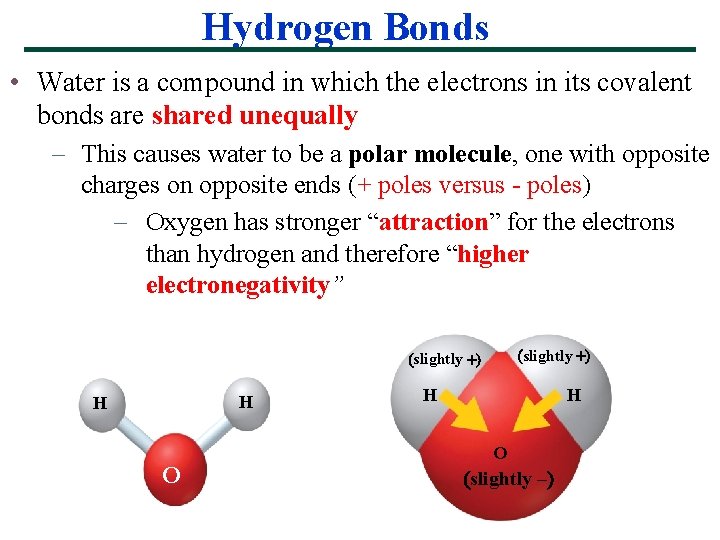
Hydrogen Bonds • Water is a compound in which the electrons in its covalent bonds are shared unequally – This causes water to be a polar molecule, one with opposite charges on opposite ends (+ poles versus - poles) – Oxygen has stronger “attraction” for the electrons than hydrogen and therefore “higher electronegativity” slightly H H O slightly –
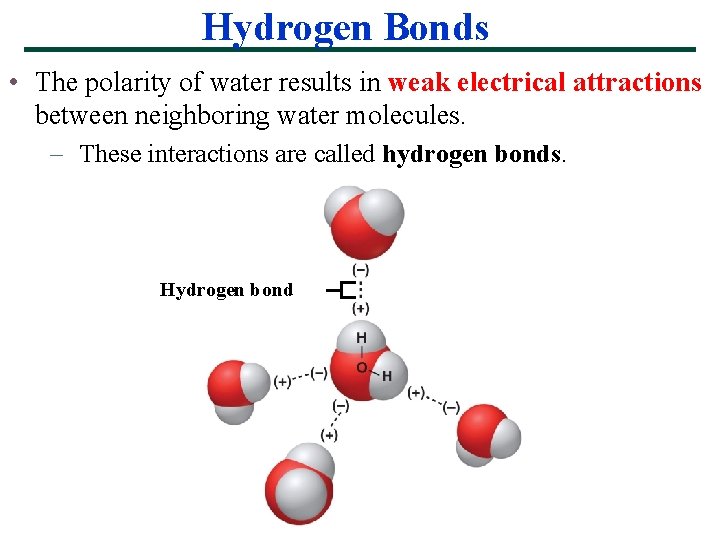
Hydrogen Bonds • The polarity of water results in weak electrical attractions between neighboring water molecules. – These interactions are called hydrogen bonds. Hydrogen bond

Practice What happens when the outermost electron shell of an element such as neon contains the maximum number of eight electrons? a. It becomes extremely unstable and reactive b. It becomes stable and inert c. It forms covalent bonds in an aqueous solution An atom with an unfilled outer shell is a. b. c. d. extremely unreactive likely to bond with another atom unable to bond with another atom polar

Practice The number of protons of an atom of an element determines its a. weight b. isotopic status c. mass number d. tendency to form ionic bonds e. atomic number Fluorine’s atomic number is 9 and its mass number is 19. which of the following is true of fluorine? a. It contains 9 protons and 10 neutrons b. It contains 19 protons and 9 neutrons c. It has no net charge when it has 19 electrons d. It contains 9 protons and 19 neutrons e. It is lighter than helium

Chemical Reactions • Chemical reactions occurs when: a. One substance combine with another b. One substance is broken down into another c. Chemical bonds are broken and other bonds form • Cells constantly rearrange molecules by breaking existing chemical bonds and forming new ones. – Such changes in the chemical composition of matter are called chemical reactions. – Reaction between oxygen gas & hydrogen gas that forms water. • Chemical reactions include reactants, the starting materials, and products, the end materials. • Chemical reactions can rearrange matter but cannot create or destroy matter.
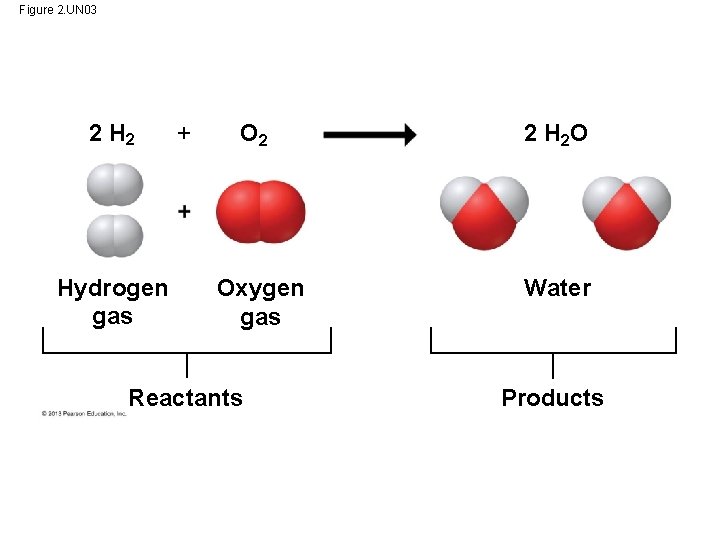
Figure 2. UN 03 2 Hydrogen gas + O 2 2 H 2 O Oxygen gas Water Reactants Products

Water: The Molecule That Supports All of Life • Water is the biological medium on Earth - All life occurs in water • The abundance of water is the main reason Earth is habitable • All living organisms require water more than any other substance • Most cells are surrounded by water, and cells themselves are about 70– 95% water • Water is the only common substance in the natural environmen that exists in all three physical states of matter: solid, liquid, and gas. • Water is a reactant in many of the chemical reactions of life.

Figure 2. 10

Water’s Life-Supporting Properties • The polarity of water molecules and the hydrogen bonding that results explain most of water’s life-supporting properties. – Water molecules stick together – forming weak attractions between partially negative -O and partially positive +H of another polar molecule. – Water has a strong resistance to change in temperature. – Frozen water floats – Water is a common solvent for life. © 2013 Pearson Education, Inc.
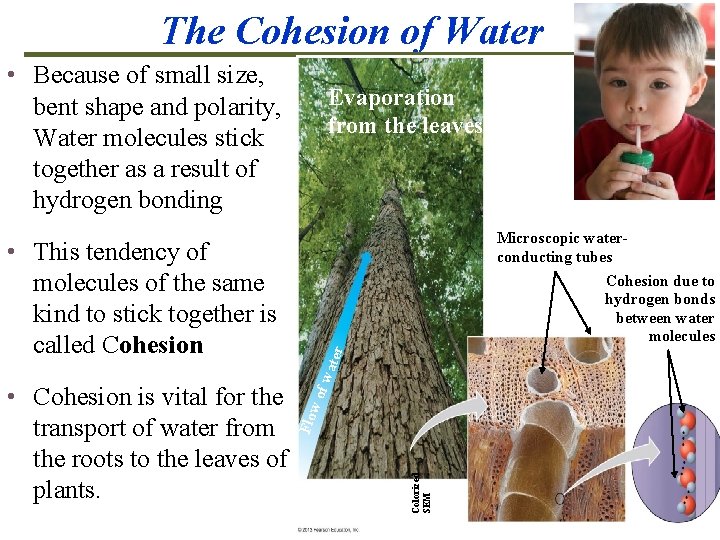
The Cohesion of Water • Because of small size, bent shape and polarity, Water molecules stick together as a result of hydrogen bonding f wa ter Cohesion due to hydrogen bonds between water molecules wo Colorized SEM • Cohesion is vital for the transport of water from the roots to the leaves of plants. Microscopic waterconducting tubes Flo • This tendency of molecules of the same kind to stick together is called Cohesion Evaporation from the leaves

The Cohesion of Water • Surface tension is the measure of how difficult it is to stretch or break the surface of a liquid. – Hydrogen bonds give water an unusually high surface tension. Figure 2. 12 © 2013 Pearson Education, Inc.

2. How Water Moderates Temperature • Because of hydrogen bonds water has a strong resistance to temperature change. • Heat and temperature are related, but different. – Heat is the amount of energy associated with the movement of the atoms and molecules in a body of matter. – Temperature measures the intensity of heat. • Water can absorb and store large amounts of heat while only changing a few degrees in temperature. • Water can moderate temperatures. – Earth’s giant water supply causes temperatures to stay within limits that permit life. – Evaporative cooling occurs when a substance evaporates and the surface of the liquid remaining behind cools down – removes heat from Earth and from organisms © 2013 Pearson Education, Inc.

Evaporative cooling • When a substance evaporates by changing the physical state from a liquid to gas , the surface of the liquid remaining behind cools down. • The molecules with the greatest energy (hottest one) tend to vaporize first • Evaporative cooling removes heat from the Earth and from organisms.

3. The Biological Significance of Ice Floating • When water molecules get cold enough, they move apart (the movement of its molecules slows), forming ice. - A chunk of ice has fewer molecules than an equal volume of liquid water. • Ice floats because it is less dense than the liquid water around it. - the water molecules are spaced out more than in liquid water • Oceans & lakes don’t freeze solid b/c surface insulates water below allowing life to survive the winter • If ice sank, ponds, lakes, & even oceans would freeze solid • Life in water could not survive if bodies of water froze solid.
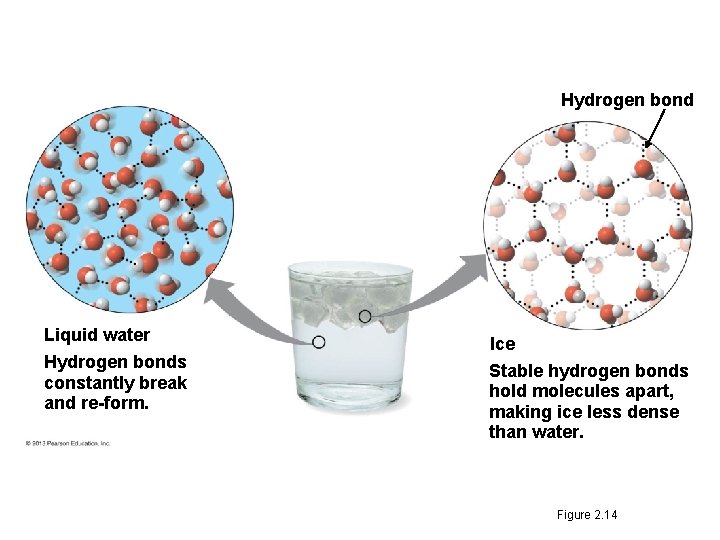
Hydrogen bond Liquid water Hydrogen bonds constantly break and re-form. Ice Stable hydrogen bonds hold molecules apart, making ice less dense than water. Figure 2. 14

4. Water as the Solvent of Life • A solution is a liquid consisting of a homogeneous mixture of two or more substances. – The dissolving agent is the solvent (medium of for chemical reaction). – The dissolved substance is the solute (table salt ). • When water is the solvent, the result is an aqueous solution § Water dissolve an enormous variety of solutes necessary for life. § Hydrophilic substance dissolves in water, have attraction to water (polar molecules) § Hydrophobic substance that don’t have an attraction to water (non- polar molecules, oil and butter) § Water is the solvent inside all cells, in blood, and in plant sap. © 2013 Pearson Education, Inc.
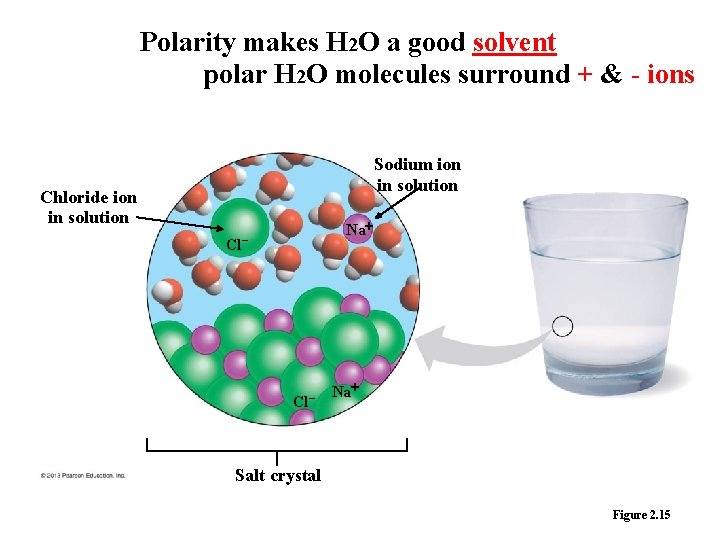
Polarity makes H 2 O a good solvent polar H 2 O molecules surround + & - ions Sodium ion in solution Chloride ion in solution Cl Na – Cl – Na Salt crystal Figure 2. 15

Practice Surface tension and capillary action occur in water because it a. has hydrogen bonds b. has ionic bonds c. is dense d. is nonpolar e. is wet In the reaction glucose + fructose sucrose + water, _____ is a reactant and _____ is a product.

Practice Why does ice float on water? a. Ice has a higher p. H than water b. Molecules of water are spaced father apart than molecules of ice c. Ice has a lower p. H than water d. Molecules of ice are spaced father apart than molecules of water Cocoa is mixed with hot water to make hot chocolate. The cocoa is the ______, the hot water is the _____, and the hot chocolate is the _____. a. b. c. d. e. solute ……. . . solvent ……… solution solvent ……. . … solution ……. . solute solution………. . . solvent ………. . . solute solvent ……. . … solute……… solution solute……. . . solution …. …. . solvent

Practice The hydrogen bond between two molecules arises because water is a. b. c. d. polar hydrophobic a liquid nonpolar Which of the following is not a property of water? a. b. c. d. e. polarity a good solvent cohesion nonpolarity store heat well

Practice Which of the following p. H values indicates a slightly acidic solution? a. b. c. d. e. 9 7 3 11 6. 5 H 2 SO 4 can ionize to yield two H+ ions and one H 2 SO 4 is a. b. c. d. e. molar an acid a base a solution a buffer ion.

Practice A basic solution contains a. b. c. d. more OH- ions than H+ ions the same number of OH- ions as H+ ions no OH- ions more H+ ions than OH- ions Which of the following has concentration of hydrogen ions? a. b. c. d. baking soda at p. H 9 Household ammoniac at p. H 11 human blood at p. H 7 black coffee at p. H 5 the greatest

Practice Which of these refers to atoms with the same atomic number but different atomic masses? a. b. c. d. e. these atoms are different elements these atoms have different numbers of elements these atoms are isomers these atoms are isotopes these atoms have different numbers of protons An ionic bond involves______. a. b. c. d. e. the sharing of a single pair of electrons an attraction between ions of opposite charge no atoms other than sodium and chlorine the unequal sharing of an electron pair water avoidance

Practice Which of these refers to atoms with the same atomic number but different atomic masses? a. b. c. d. e. these atoms are different elements these atoms have different numbers of elements these atoms are isomers these atoms are isotopes these atoms have different numbers of protons An ionic bond involves______. a. b. c. d. e. the sharing of a single pair of electrons an attraction between ions of opposite charge no atoms other than sodium and chlorine the unequal sharing of an electron pair water avoidance

Practice What determines the types of chemical reactions that an atom participates in ? a. b. c. d. e. its atomic mass the number of electrons in the innermost electron shell the number of electrons in the outermost electron shell the number of protons it contains its atomic number An unchanged atom of boron has an atomic number of 5 and an atomic mass of 11. How many protons does boron have?

Practice The unequal sharing of electrons within a water molecule makes the water molecule _____. In a neutral solution the concentration of _____. a. hydrogen ions is equal to the concentration of hydroxide ions b. water molecules is less than the concentration of hydrogen ions c. hydrogen ions is less than the concentration of hydroxide ions d. water molecules is less than the concentration of hydroxide ions e. hydrogen ions is greater than the concentration of hydroxide ions

Practice Which of the following substance is a substance that resists changes in p. H? a. b. c. d. e. acid buffer base Solvent solution What is the atomic number of an atom that has 6 protons, 6 neutrons, and 6 electrons?
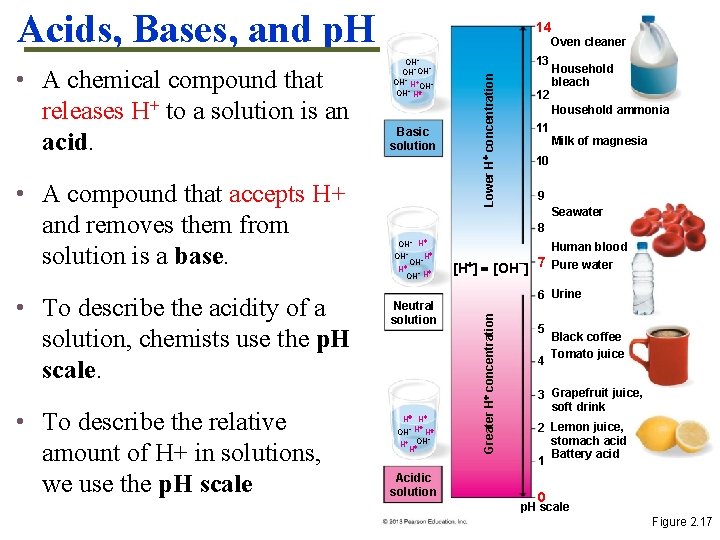
Acids, Bases, and p. H • To describe the acidity of a solution, chemists use the p. H scale. • To describe the relative amount of H+ in solutions, we use the p. H scale OH− − OH−OH − OH H OH− H Basic solution 13 Lower H concentration • A compound that accepts H+ and removes them from solution is a base. Oven cleaner 12 Household bleach Household ammonia 11 Milk of magnesia 10 9 Seawater 8 OH− H H OH− − OH H − OH H Neutral solution H H OH− H H − OH H H Acidic solution Human blood [H ] [OH−] 7 Pure water 6 Urine Greater H concentration • A chemical compound that releases H+ to a solution is an acid. 14 5 4 Black coffee Tomato juice 3 Grapefruit juice, soft drink 2 Lemon juice, stomach acid Battery acid 1 0 p. H scale Figure 2. 17
![p. H 14 Lower H concentration Basic Neutral − [H ] [OH ] Acidic p. H 14 Lower H concentration Basic Neutral − [H ] [OH ] Acidic](http://slidetodoc.com/presentation_image/cedb0bd1ea20cc97632589bef110350c/image-57.jpg)
p. H 14 Lower H concentration Basic Neutral − [H ] [OH ] Acidic p. H 7 H concentration equal to OH− concentration Greater H concentration p. H 0 p. H scale Figure 2. UN 10

Acids, Bases, and p. H • Biological fluids contain buffers which are substances that resist p. H change ü accept H+ ions when they are in excess and ü donate H+ ions when they are depleted • Buffers & Cellular regulation - p. H of cells must be kept at ~ 7 - p. H affects shape of molecules - shape of molecules affect function - therefore p. H affects cellular function © 2013 Pearson Education, Inc.

• Increases in global CO 2 concentrations may lead to - the acidification of the oceans and - ecological disasters Figure 2. 18

Can Exercise Boost Your Brain Power? • Observation: Human brains shrink as we age. • Question: Can aerobic exercise slow or reverse brain loss? • Hypothesis: MRI (magnetic resonance imaging) scans would reveal differences between people who regularly exercised aerobically and those who did not. • Prediction: Brains of active people would shrink less than the brains of less active people. • Experiment: Twenty-nine people in their 60 s and 70 s exercised for three one-hour sessions per week. A control group of 29 people engaged in non-aerobic stretching exercises for the same periods. • Results: The aerobic group showed significant increases in brain volume compared to the non-aerobic group

Figure 2. UN 04
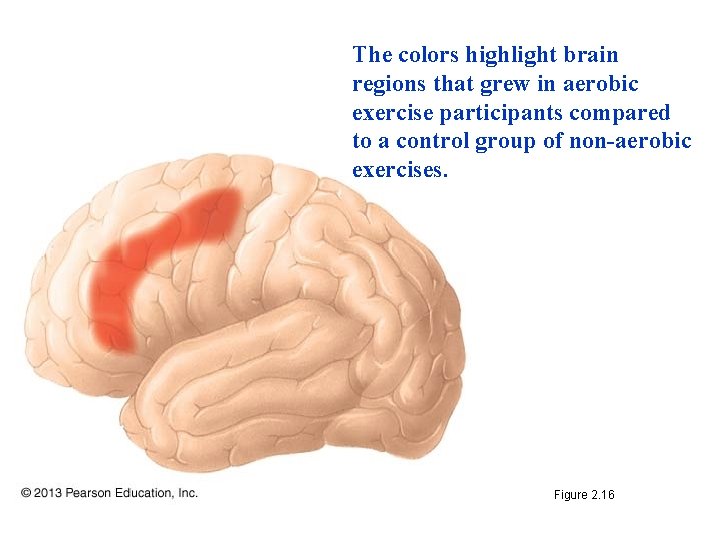
The colors highlight brain regions that grew in aerobic exercise participants compared to a control group of non-aerobic exercises. Figure 2. 16

Fluoride in the Water • Fluoride – Is a common ingredient in Earth’s crust – Helps maintain healthy teeth • Fluoride prevents cavities by – Affecting the metabolism of oral bacteria – Promoting the replacement of lost minerals on the tooth surface © 2010 Pearson Education, Inc.

Evolution Connection: The Search for Extraterrestrial Life • If life similar to ours has evolved elsewhere in the universe, then it too would depend upon water. • Researchers at NASA missions have found evidence that water was once abundant on Mars. • Microbial life may exist below the Martian surface. © 2013 Pearson Education, Inc.
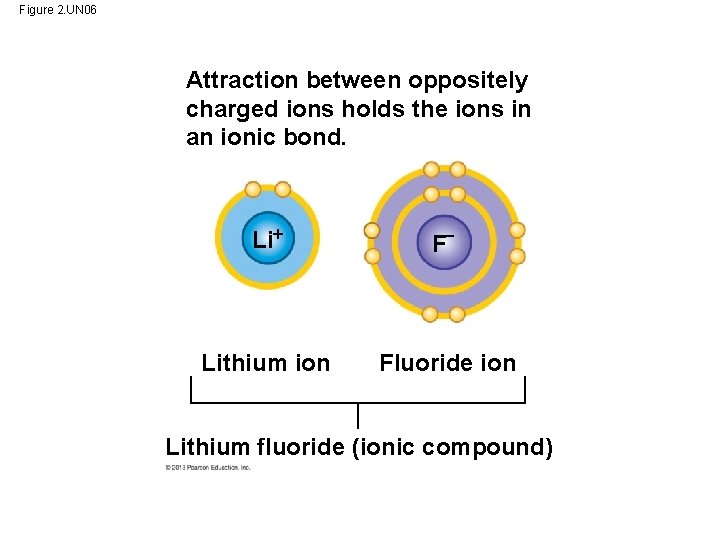
Figure 2. UN 06 Attraction between oppositely charged ions holds the ions in an ionic bond. Li F− Lithium ion Fluoride ion Lithium fluoride (ionic compound)
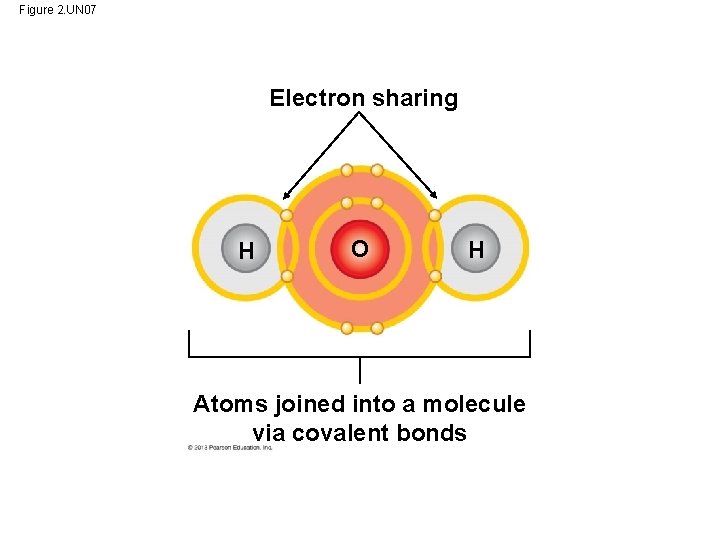
Figure 2. UN 07 Electron sharing H O H Atoms joined into a molecule via covalent bonds
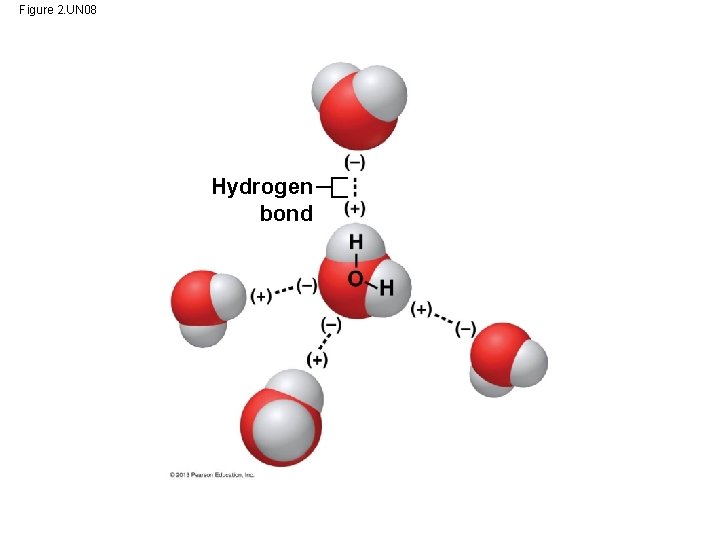
Figure 2. UN 08 Hydrogen bond
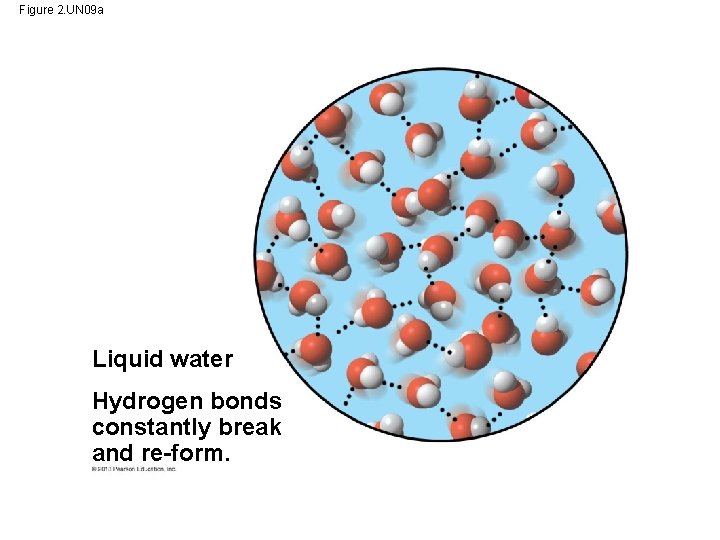
Figure 2. UN 09 a Liquid water Hydrogen bonds constantly break and re-form.
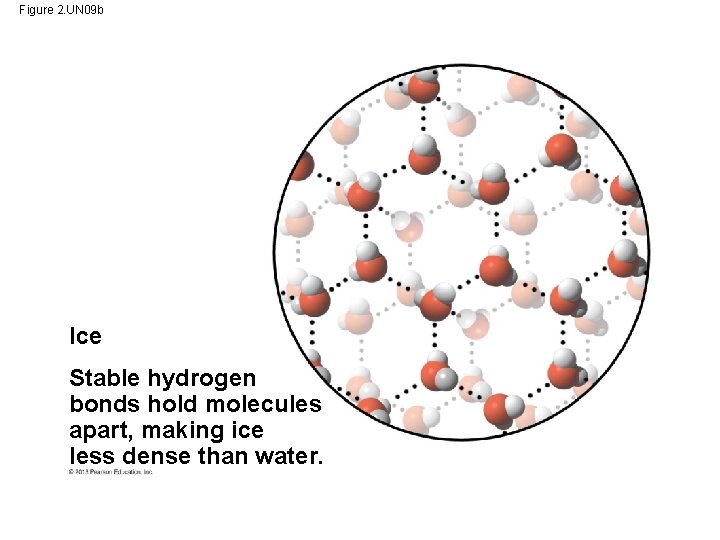
Figure 2. UN 09 b Ice Stable hydrogen bonds hold molecules apart, making ice less dense than water.

Figure 2. UN 11
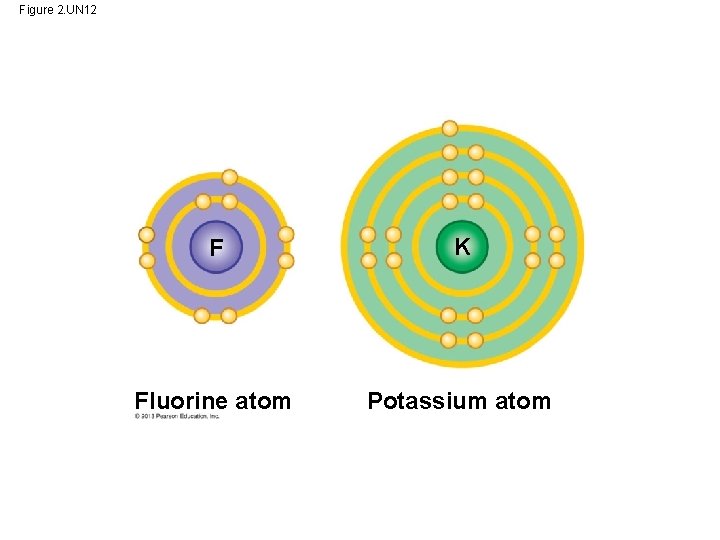
Figure 2. UN 12 F K Fluorine atom Potassium atom
- Slides: 71