Chapter 2 Elements are the building blocks of

Chapter 2 Elements are the building blocks of matter

Chapter 2 Objectives In this chapter, you will • Distinguish between metals, non-metals, and metalloids • Explain the organization of the periodic table • Predict the properties of a family of elements in the periodic table • Compare the characteristics and atomic structures of an element • Draw Bohr model diagrams
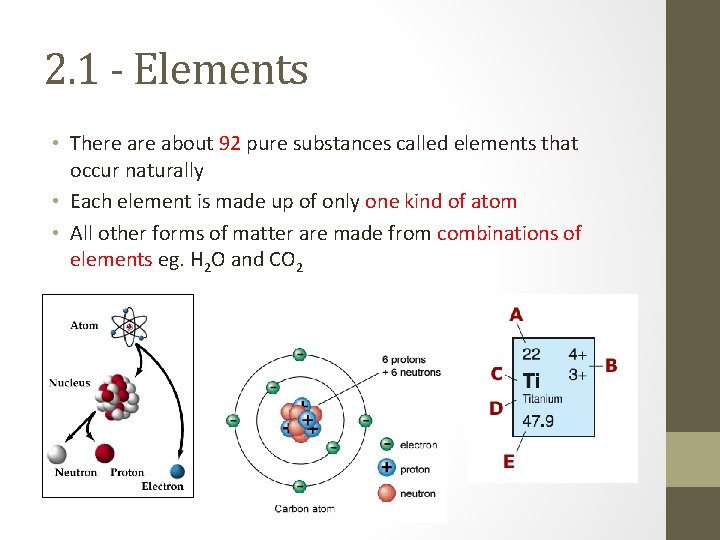
2. 1 - Elements • There about 92 pure substances called elements that occur naturally • Each element is made up of only one kind of atom • All other forms of matter are made from combinations of elements eg. H 2 O and CO 2

Elements • Each element is represented by a one- or two-letter symbol • Common elements include; • • Hydrogen (H) Iron (Fe) Oxygen (O) Sodium (Na) Chlorine (Cl) Mercury (Hg) Silver (Ag) Silicon (Si)

Chemical Symbol • There are 118 different elements (92 naturally occurring and others synthetic (man made) • Each element symbol consists of one or two letters • The first letter is always capitalized, whether it is a single letter or two lettered element, the second letter is always lower case.

Chemical Symbol • The symbols on the Periodic Table are universal (used world-wide) • Symbols come from Latin (or Greek) words • CAPITALIZE the first letter, lower case 2 nd letter (if it is needed) e. g. No vs. NO • (No = nobelium - an element VS NO = nitrogen monoxide – a compound) • Check out page 44 for some highlights!

Elements How do we separate one element from one another? • From Chapter 1, we looked at physical properties like state, color, melting and boiling points, malleability, ductility, magnetism, crystallinity, density etc • Each element has its own set of physical properties as well as chemical properties. • We use Chemical properties to describe a substance’s ability to react chemically with other substances to form new products

Elements (pg. 44) • Your textbook talks about 8 specific common elements • All elements are separated into either metals or non-metals by the staircase on the period table • Metals are typically hard, shiny, malleable, ductile and good conductors of heat and electricity • Non-metals tend not to have these properties and are usually gases or brittle solids at room temperature

Hydrogen (H) • • Colorless, odourless, tasteless, and highly flammable gas It is the lightest element Hydrogen makes up over 90% of the atoms in the universe Lighter than air

Iron (Fe) • • Very strong metal Combines with carbon to make steel Iron is very ductile (wires in foundations) Rusts when exposed to oxygen and water

Oxygen (O) • • • Non-metal Gaseous element Combines with sugar in our body to release energy 21% of atmosphere is oxygen Very reactive with other elements

Sodium (Na) • Is a soft metal • It looks very metallic and is shiny but cannot be used like your typical metal. It is too soft! • A pot made of sodium cannot be used to boil water because its boiling point is lower than water (98 degrees) • Sodium and water react violently to produce hydrogen gas and heat

Chlorine (Cl) • Pale yellow-green gas • Added to swimming pools and some water supplies to kill bacteria • Chlorine can combine with Sodium to make table salt

Mercury (Hg) Metal Liquid at room temperature Extremely toxic Mercury is used to create a spark free weld when working with dangerous gases • Found in older thermometers • •

Silver (Ag) • The abundance of Silver created a ‘Silver Rush’ early in the 1800 s in the Kootenays in around areas like Nelson, Kaslo, and Slocan • White metallic element • It is considered a precious metal • It can be modeled, polished, and stretched into jewellery and silverware

Silicon (Si) • Silicon is the second most common element in Earth’s crust • It is not a metal but is brittle, grey, and has a metallic lustre • It is used mainly as a semiconductor in electronics • A semiconductor is effective at conducting electricity in controllable conditions • It is found in computer chips and hardware • Also used to make the frames of automobiles when combined with aluminum.

Elements • Meet the Elements
- Slides: 17