CHAPTER 2 CHEMICAL REACTIONS 2 2 BALANCING CHEMICAL

CHAPTER 2: CHEMICAL REACTIONS 2 -2: BALANCING CHEMICAL EQUATIONS
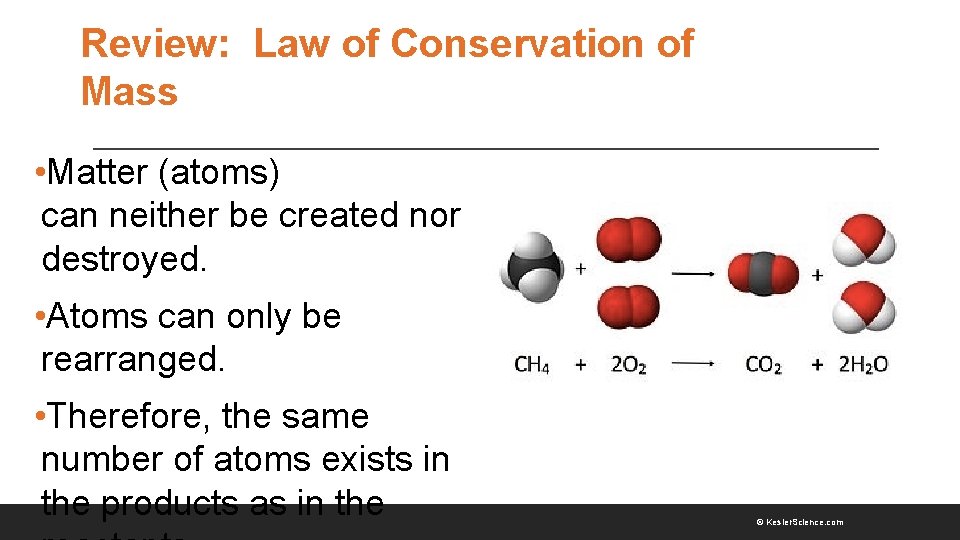
Review: Law of Conservation of Mass • Matter (atoms) can neither be created nor destroyed. • Atoms can only be rearranged. • Therefore, the same number of atoms exists in the products as in the © Kesler. Science. com
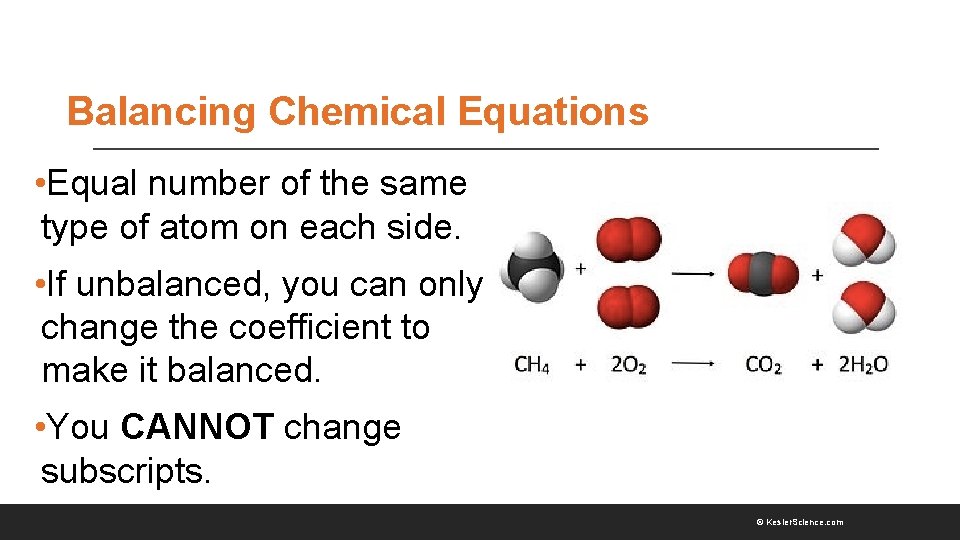
Balancing Chemical Equations • Equal number of the same type of atom on each side. • If unbalanced, you can only change the coefficient to make it balanced. • You CANNOT change subscripts. © Kesler. Science. com
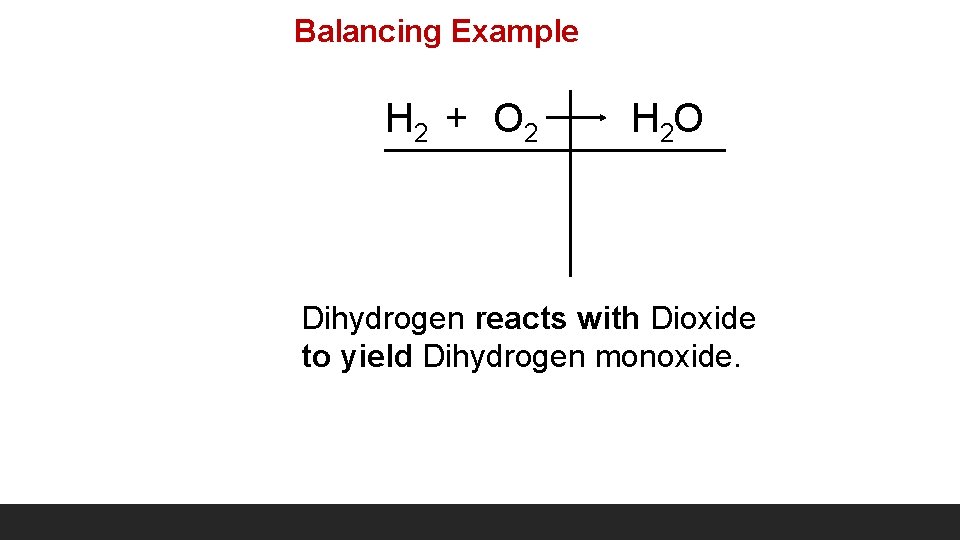
Balancing Example H 2 + O 2 H 2 O Dihydrogen reacts with Dioxide to yield Dihydrogen monoxide.
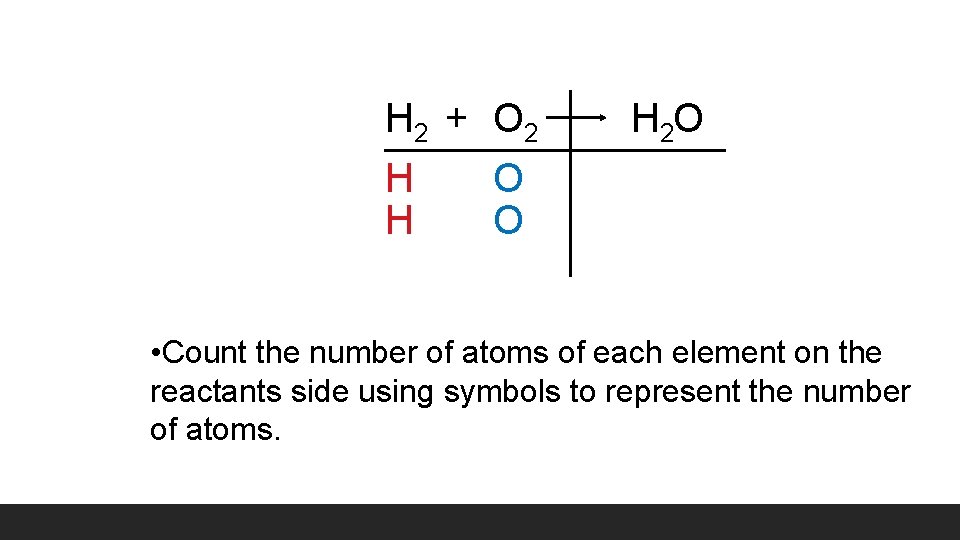
H 2 + O 2 H O H 2 O • Count the number of atoms of each element on the reactants side using symbols to represent the number of atoms.
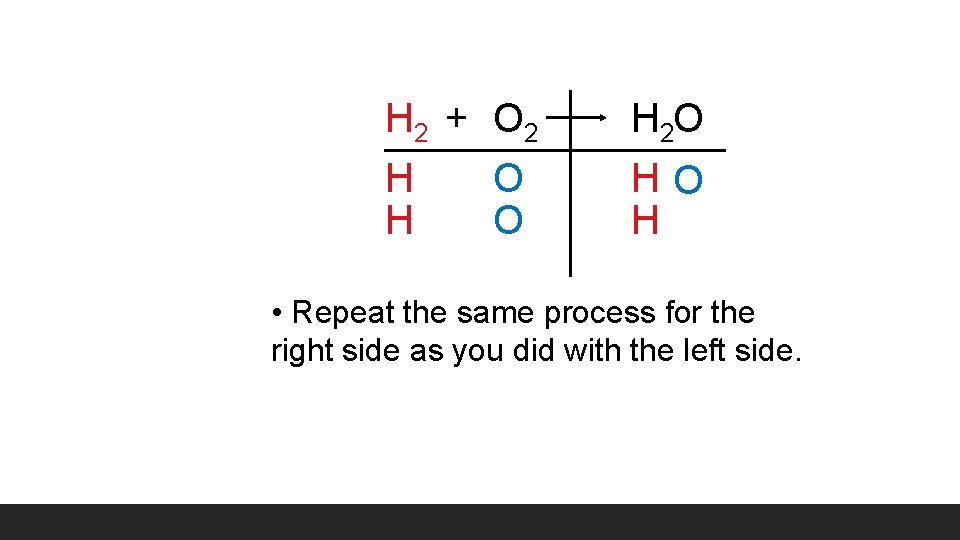
H 2 + O 2 H O H 2 O HO H • Repeat the same process for the right side as you did with the left side.
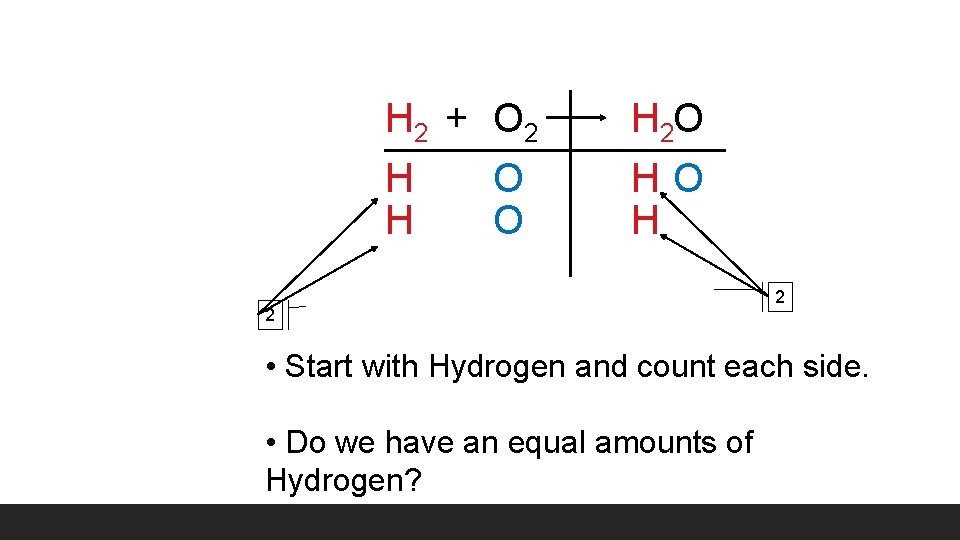
H 2 + O 2 H O H 2 O HO H 2 2 • Start with Hydrogen and count each side. • Do we have an equal amounts of Hydrogen?
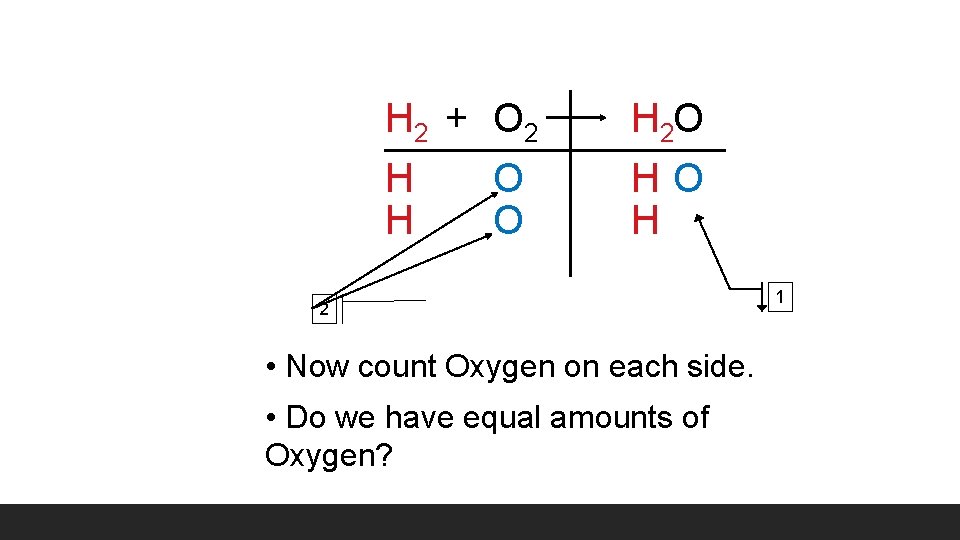
H 2 + O 2 H O H 2 O HO H 2 • Now count Oxygen on each side. • Do we have equal amounts of Oxygen? 1
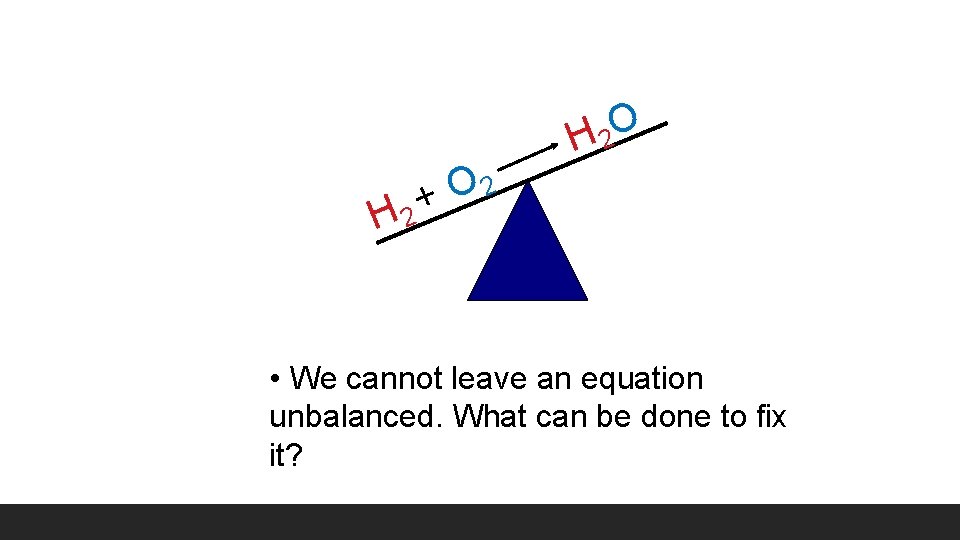
H 2 O H 2 2 O + • We cannot leave an equation unbalanced. What can be done to fix it?
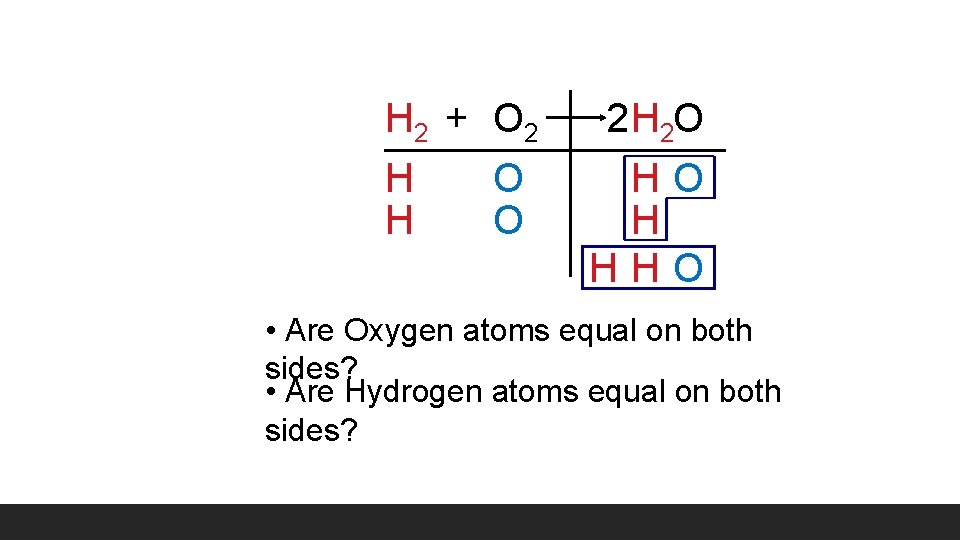
H 2 + O 2 H 2 O HO H HHO • Are Oxygen atoms equal on both sides? • Are Hydrogen atoms equal on both sides?
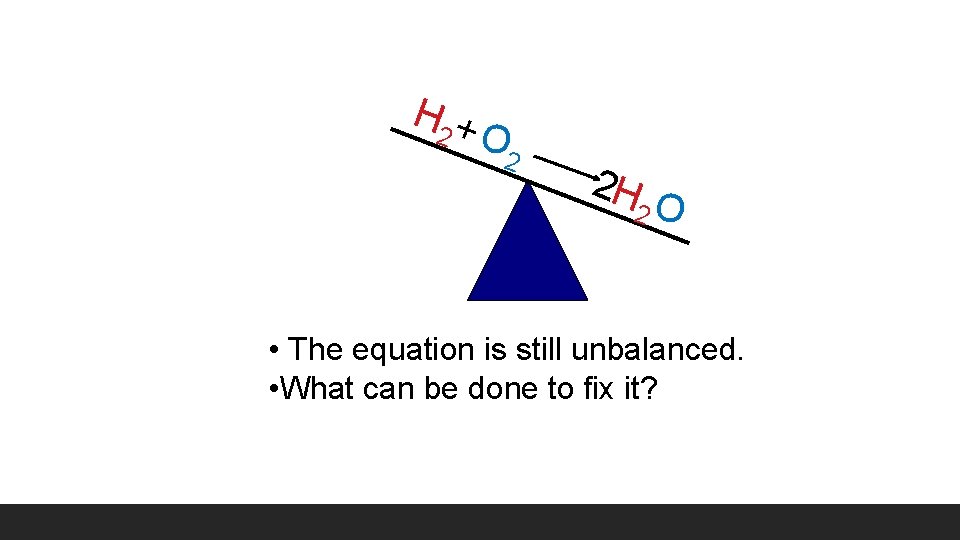
H 2 + O 2 2 H 2 O • The equation is still unbalanced. • What can be done to fix it?
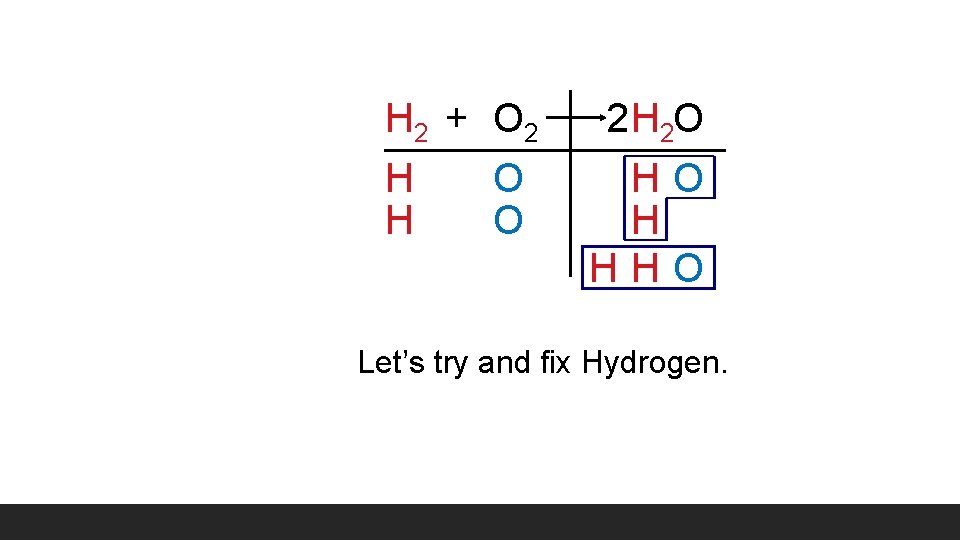
H 2 + O 2 H 2 O HO H HHO Let’s try and fix Hydrogen.
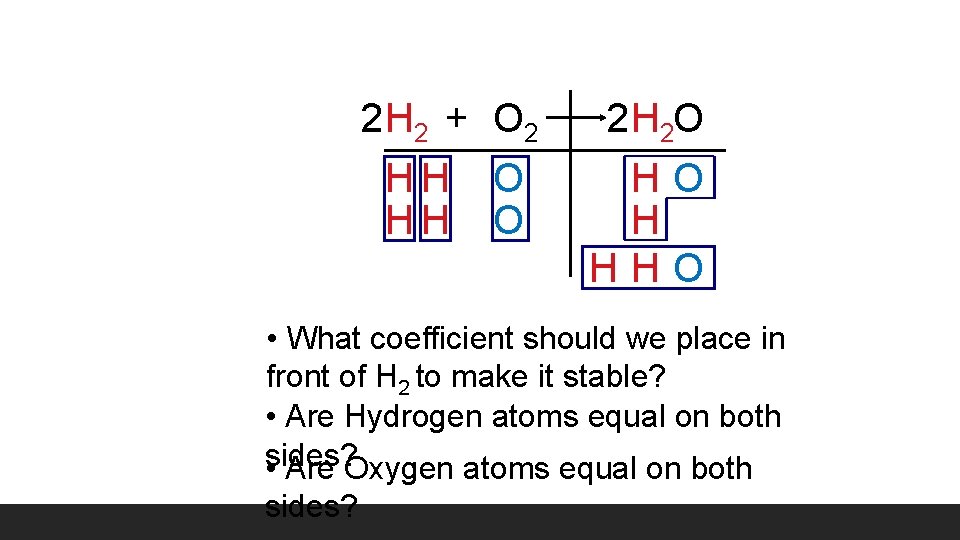
2 H 2 + O 2 HH O 2 H 2 O HO H HHO • What coefficient should we place in front of H 2 to make it stable? • Are Hydrogen atoms equal on both • sides? Are Oxygen atoms equal on both sides?
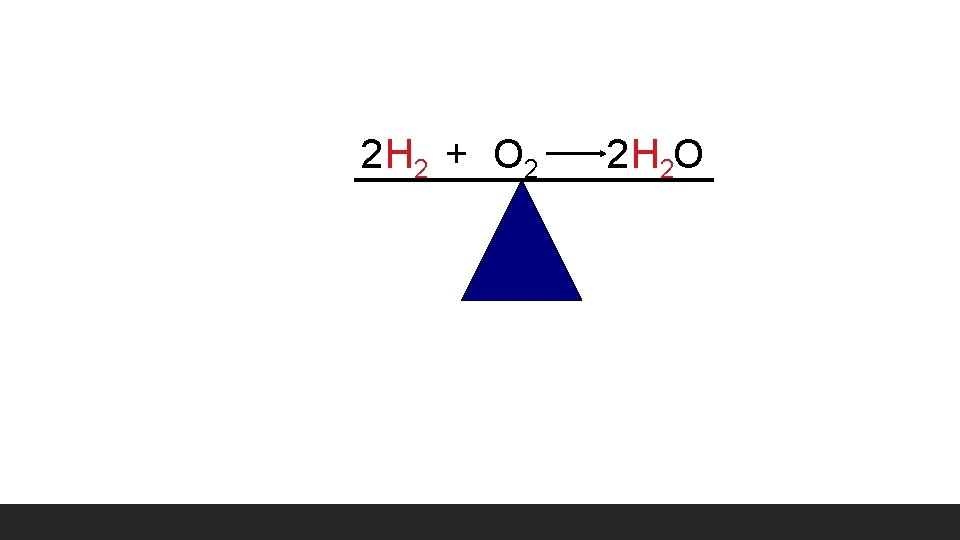
2 H 2 + O 2 2 H 2 O

Balancing Chemical Equations Practice 4 2 2 Step 1 – Take Inventory of 2 Ag 2 O g Ag + O the elements and atoms on Ag – 2 4 = Ag – 1 42 the product and reactant side. O – 1 2 = O – 2 Step 2 – Is it balanced? Step 3 – If unbalanced, change coefficients until it’s balanced. Balanced!

Balancing Chemical Equations Practice Step 1 – Take Inventory of CH + Cl 4 4 2 CCl 4 + HCl 4 the elements and atoms on = C – 1 the product and reactant side. C – 1 H – 4 = H – 1 4 Step 2 – Is it balanced? Cl – 2 8 = Cl – 5 8 Step 3 – If unbalanced, change coefficients until it’s balanced. Balanced! © Kesler. Science. com
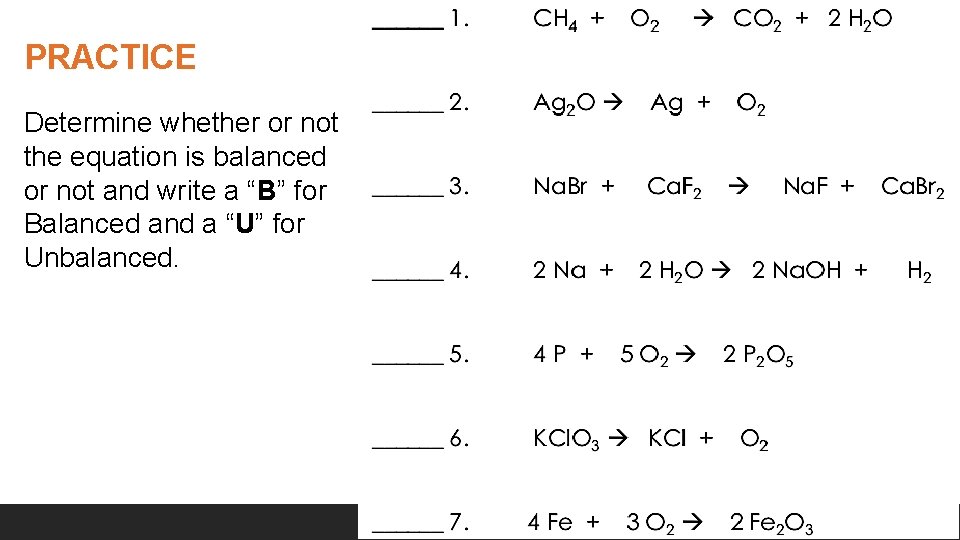
PRACTICE Determine whether or not the equation is balanced or not and write a “B” for Balanced and a “U” for Unbalanced. © Kesler. Science. com

Quick Action – Balance the Equation Step 1 – Take Inventory of the elements and atoms on the product and reactant side. Na. Br + Ca. F 2 Na. F + Ca. Br 2 Step 2 – Is it balanced? Step 3 – If unbalanced, change coefficients until it’s balanced.
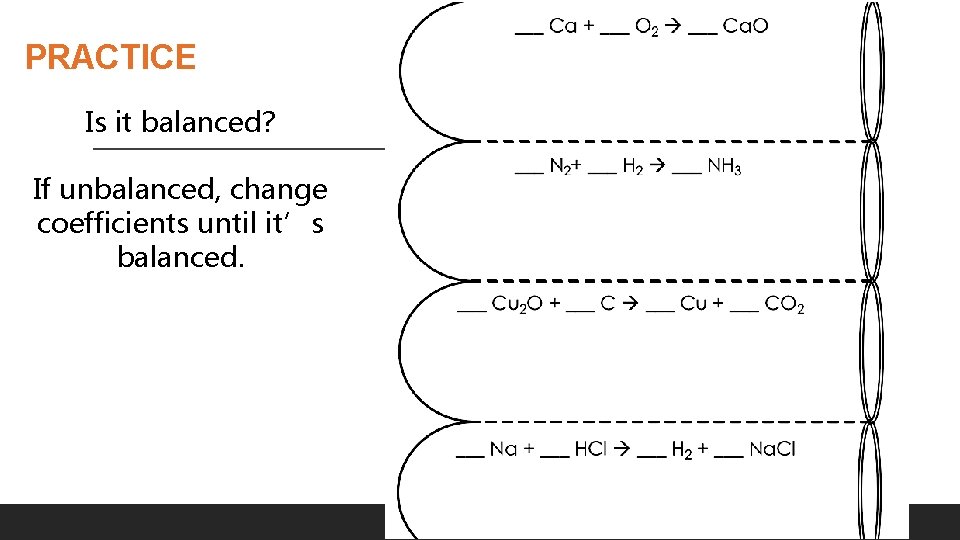
PRACTICE Is it balanced? If unbalanced, change coefficients until it’s balanced. © Kesler. Science. com
- Slides: 19