Chapter 2 Chemical Basis of Life Anatomy Physiology
Chapter 2 Chemical Basis of Life Anatomy & Physiology I (Dr. Rothschild)

2. 1: Introduction Why study chemistry in an Anatomy and Physiology class? - Body functions depend on cellular functions - Cellular functions result from chemical changes - Biochemistry helps to explain physiological processes 2

Matter Properties Mass, Volume • Atoms smallest unit of matter • Elements- one type of atom 3
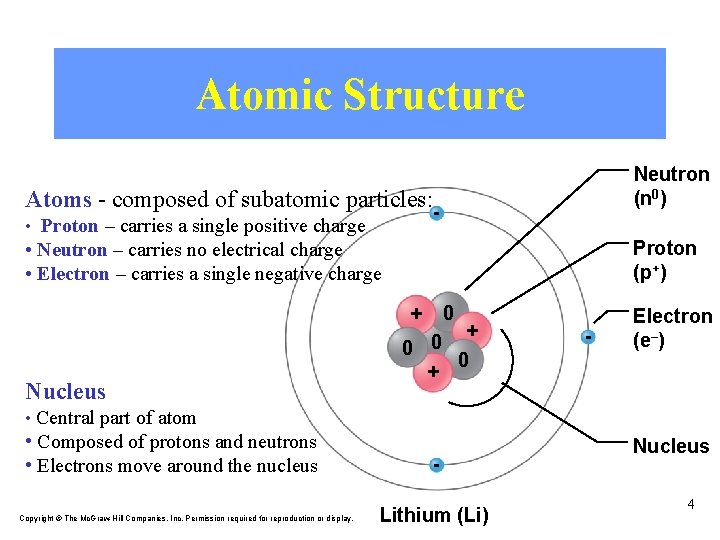
Atomic Structure Neutron (n 0) Atoms - composed of subatomic particles: • Proton – carries a single positive charge Proton (p+) • Neutron – carries no electrical charge • Electron – carries a single negative charge 0 + Nucleus + 0 0 0 + - Electron (e–) • Central part of atom • Composed of protons and neutrons • Electrons move around the nucleus Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Lithium (Li) Nucleus 4

Chemical Behavior of an Atom 1. Depends on electrons in outermost shell 2. Valence electrons (valence shell) 5. Orbitals describe the 3 -D space occupied (90% of the time) by an electron as it orbits a nucleus. 6. Reactivity of an atom arises 3. Atoms with the same from the presence of number of electrons in their unpaired electrons in one or valence shell exhibit similar more orbitals of an atom’s chemical behavior valence shell • Consider: F and Cl – Both combine with Na 1: 1 – Na. F and Na. Cl 4. Completed valence shell atoms are unreactive (inert) 7. Unpaired electrons of atoms interact to complete their valence shells

Elemental Isotopes • Atoms have same number of protons • Isotopes have varying numbers of neutrons in their nucleus • Isotopes behave similarly in chemical reactions despite different masses. • Carbon (6 C) Isotopes – 12 C (stable) – 13 C (stable) – 14 C (radioactive) • Radioactive isotopes decay spontaneously giving off particles and energy • Atomic mass number is actually an average of the atomic masses of all the element’s isotopes

Bonding of Atoms Bonds form when atoms combine with other atoms Electrons occupy regions of space called orbitals (subshells) Different energy levels (shells) circle the nucleus Outermost (valence) electrons responsible for bonding • Atoms may lose/gain electrons • Atoms may share electrons 7

Bonding of Atoms: Ions Ion • An atom that gains or loses electrons to become stable • An electrically charged atom Cation • A positively charged ion • Formed when an atom loses electrons Anion • A negatively charged ion • Formed when an atom gains electrons 11 p+ 12 n 0 Sodium atom (Na) 17 p+ 18 n 0 Chlorine atom (Cl) (a) Separate atoms If a sodium atom loses an electron to a chlorine atom, the sodium atom becomes a sodium ion (Na+), and the chlorine atom becomes a chloride ion (Cl–). 8

Formula is a ratio Na. Cl is a compound, not a molecule Ionic Compounds Highly unequally electronegative atom strips an electron from its partner’s valence shell

Covalent Bonds Formed when atoms share electrons Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H - H 2 - + + - Hydrogen atom + Hydrogen atom Hydrogen molecule All organic (carbon) substances are formed by covalent bonds, are molecules 10

Structural formulas show atoms bond are arranged in various molecules H H O O C O O H H H 2 O CO 2 H 2 O Polar Molecules Slightly negative end Molecule with a slightly negative end a slightly positive end Results when electrons are not shared equally in covalent bonds • Water is an important polar molecule (a) Slightly positive ends 11 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Elements and Compounds Element: Cannot be broken down • Bulk elements – required by the body in large amounts • Trace elements - required by the body in small amounts • Ultratrace elements – required by the body in very minute amounts Compound • Two or more elements in a fixed ratio • Chemical bonds maintain ratio • Example: Sodium Chloride (Na. Cl) Compound properties different than elements that form it

Molecules and Compounds Compound – particle formed when two or more atoms of different elements chemically combine Molecule – particle formed when two or more atoms chemically combine Molecular formulas – depict the elements present and the number of each atom present in the molecule H 2 C 6 H 12 O 6 H 2 O 13

Hydrogen Bonds A weak attraction between the positive end of one polar molecule and the negative end of another polar molecule • Formed between water molecules • Important for protein and nucleic acid structure H H O H Hydrogen bonds O H H 14 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Chemical Reactions Chemical reactions occur when chemical bonds form or break among atoms, ions, or molecules Reactants are the starting materials of the reaction - the atoms, ions, or molecules Products are substances formed at the end of the chemical reaction Na. Cl ’ Na+ + Cl. Reactant Products 15

Chemical Reactions • Do not gain/lose atoms in a reaction 6 CO 2 + 6 H 2 O C 6 H 12 O 6 + 6 O 2 • Most reactions are reversible: products can change back to the reactants 3 H 2 + N 2 2 NH 3 • Chemical equilibrium: forward and reverse reactions occur at the same rate

Types of Chemical Reactions Synthesis Reaction – more complex chemical structure is formed A + B ’ AB Decomposition Reaction – chemical bonds are broken to form a simpler chemical structure AB ’ A + B Exchange Reaction – chemical bonds are broken and new bonds are formed AB + C ’ A + CB AB + CD ’ AD + CB 17

Acids, Bases, and Salts Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Na+ Cl– Salt crystal Na+ Cl– Ions in solution 18

Acid and Base Concentration p. H scale - indicates the concentration of hydrogen ions in solution Neutral – p. H 7; indicates equal concentrations of H+ and OH- Acidic – p. H less than Acidic Relative + Amounts H + of H (red) 3. 0 and OH– 2. 0 apple (blue) gastric juice p. H 0 1 Acidic Each p. H unit represents a 10 fold increase in hydrogen ion concentration 5. 3 4. 2 cabbage tomato juice 6. 0 corn 2 3 4 5 H+ concentration increases 7; indicates a greater concentration of H+ 8. 4 7. 4 6. 6 Sodium cow’s Human biocarbonate blood milk 6 8. 0 7. 0 Egg Distilled white water 7 Neutral 8 10. 5 milk of magnesia 11. 5 Household ammonia Basic OH– 9 10 11 12 13 14 OH– concentration increases Basic (alkaline) p. H = -log [H+] Power of hydrogen Basic or alkaline – p. H greater than 7; indicates a greater concentration of OH 19 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Acids, Bases, and Salts Electrolytes – substances that release ions in water Na. Cl Na+ + Cl- Salts –formed by the reaction between an acid & base HCl + Na. OH H 2 O + Na. Cl 20

Changes in p. H and Buffers Blood p. H • Normal blood p. H is 7. 35 – 7. 45 • Alkalosis occurs when blood p. H rises to 7. 5 – 7. 8 • Acidosis occurs when blood p. H drops to 7. 0 – 7. 3 • Homeostatic mechanisms help regulate p. H • Buffers are chemicals which act to resist p. H changes 21

Buffers • Minimize p. H changes • Accepts or donates hydrogen ions • Most are a weak acid and its conjugate base • H 2 CO 3 HCO 3 - + H+

Water Hydrogen bonds produce a phenomenon known as cohesion Water molecules adhere to substances Water has a greater surface tension than most other liquids

Water and Energy Transfer • Heat is the measure of the kinetic energy due to molecular motion in a body of matter • Temperature measures the heat due to the average kinetic energy • Kinetic energy is transferred from a body with a higher kinetic energy to a body with a lower kinetic energy • Temperature is measured using Celsius scale • Calorie is a measure of heat • Kilocalorie (kcal) – Unit used in for cellular reactions

Water: The Solvent of Life • • • Solutions Solvents Solutes Aqueous Solutions Water forms hydration shells around dissolved ions of an ionic compound • Water also surrounds organic compounds if they have ionic and polar regions

Dissociation of Water H 2 O H+ + OH-

2. 3: Chemical Constituents of Cells Organic v. Inorganic Molecules Organic molecules • Contain C and H • Usually larger than inorganic molecules • Dissolve in water and organic liquids • Carbohydrates, proteins, lipids, and nucleic acids Inorganic molecules • Generally do not contain C • Usually smaller than organic molecules • Usually dissociate in water, forming ions • Water, oxygen, carbon dioxide, and inorganic salts 27

Inorganic Substances Water • Most abundant compound in living material • Two-thirds of the weight of an adult human • Major component of all body fluids • Medium for most metabolic reactions • Important role in transporting chemicals in the body • Absorbs and transports heat Oxygen (O 2) • Used by organelles to release energy from nutrients in order to drive cell’s metabolic activities • Necessary for survival 28

Inorganic Substances Carbon dioxide (CO 2) • Waste product released during metabolic reactions • Must be removed from the body Inorganic salts • Abundant in body fluids • Sources of necessary ions (Na+, Cl-, K+, Ca 2+, etc. ) • Play important roles in metabolism 29

Chemistry of Living Things – C, H, O, N = 96% – All biological compounds are based on hydrocarbon chains • C-C-C-C • Chains are very long – 4 Main types of biological compounds • • Proteins Carbohydrates Lipids Nucleic Acids

II. Proteins – Composed of amino acid building Protein blocks • COOH group • NH 2 Group – Protein = Many amino acids hooked together • Special covalent bond = peptide bond – Links C-N-C • Only found in proteins • Proteins = polypeptides
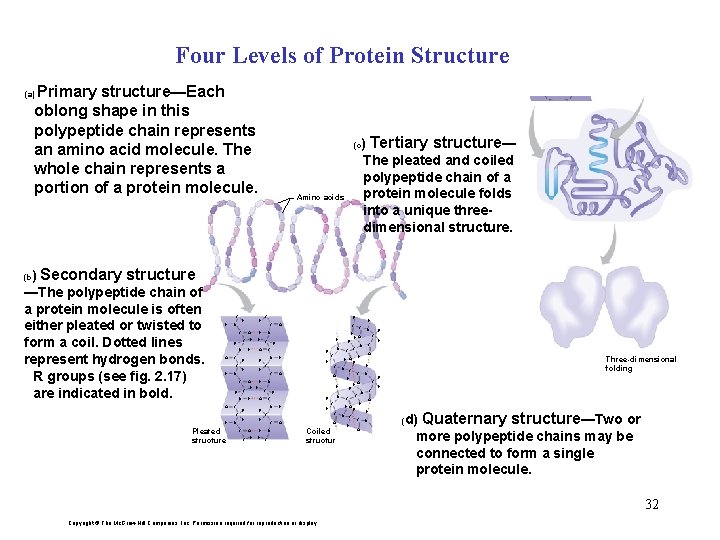
Four Levels of Protein Structure Primary structure—Each oblong shape in this polypeptide chain represents an amino acid molecule. The whole chain represents a portion of a protein molecule. (a) (c Amino acids ) Tertiary structure— The pleated and coiled polypeptide chain of a protein molecule folds into a unique threedimensional structure. H ) Secondary structure —The polypeptide chain of a protein molecule is often either pleated or twisted to form a coil. Dotted lines represent hydrogen bonds. R groups (see fig. 2. 17) are indicated in bold. H (b C H N H C O C R N O C C H N H O H Pleated structure N H R H O C H H R C R N H C N O R C N R H HO O O Coiled structure N C H O H Three-dimensional folding H C H H C N N R C O C N C N H C C R R C H N HO H N C C H O C O R N H C C H C R H C O O N H H H R C O R N C H H C C H R R C H H C O H H R H C O H O N d) Quaternary structure—Two or more polypeptide chains may be connected to form a single protein molecule. ( 32 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Animation: Protein Denaturation Please note that due to differing Please note that duesome to differing operating systems, animations operating systems, some animations is will not appear until the presentation will not in appear until the. Mode presentation viewed Presentation (Slide is viewed in Presentation Mode (Slide Show view). You may see blank slides Show view). You may see blank in the “Normal” or “Slide Sorter”slides views. in the “Normal” or “Slide Sorter” views. All animations will appear after viewing in Presentation Mode and playing each animation. Most animations will require the latest version of the Flash Player, the latest version of which is available at the Flash Player, which is available at http: //get. adobe. com/flashplayer. 33

C. Functions of proteins 1) Enzymes 2) Muscles 3) Cell Membranes 4) Structure of cells 5) Some Hormones a) insulin

III. Carbohydrates = sugars and starches – Composed of monosaccharides (single sugars) • m. s. +m. s. disaccharide + HOH – Dehydration synthesis – (m. s. +m. s. polysaccharide+many HOH (polysaccharide = starch) • Examples of monosaccharides – Glucose – Fructose – Galactose • Examples of disaccharides – Sucrose – Lactose – Maltose

Functions • Provide energy to cells • Supply materials to build cell structures • Water-soluble • Contain C, H, and O • Ratio of H to O close to 2: 1 (C 6 H 12 O 6) • Monosaccharides – glucose, fructose • Disaccharides – sucrose, lactose • Polysaccharides – glycogen, cellulose 36
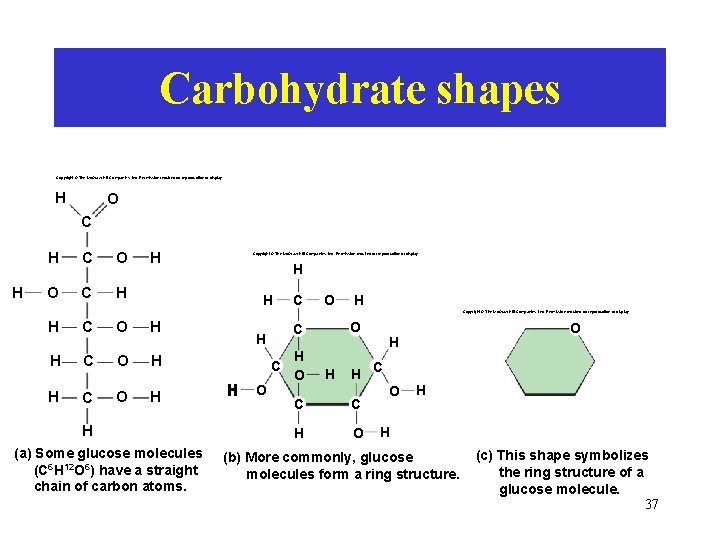
Carbohydrate shapes Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H O C H H C O O C H H Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H C O H Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H C O H H (a) Some glucose molecules (C 6 H 12 O 6) have a straight chain of carbon atoms. C O O C H H O H H C C C H O O H H (b) More commonly, glucose molecules form a ring structure. (c) This shape symbolizes the ring structure of a glucose molecule. 37

Lipids • Soluble in organic solvents; insoluble in water • Fats (triglycerides) • Used primarily for energy; most common lipid in the body • Contain C, H, and O but less O than carbohydrates (C 57 H 110 O 6) • Building blocks are 1 glycerol and 3 fatty acids per molecule • Saturated and unsaturated H H C C C O O O H Glycerol portion O H H H H C C C C H H H H O H H H H H C C C C C H H H H H O H H H C C C C H H H C C H H H Fatty acid portions H H H 38 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

Lipids • Steroids • Four connected rings of carbon • Widely distributed in the body, various functions • Component of cell membrane • Used to synthesize hormones • Cholesterol H 2 C HO (a) General structure of a steroid C H H 2 C CH 3 C HC C C H 2 C H CH 3 H 2 CH 3 H C CH CH 2 CH CH 3 CH 2 (b) Cholesterol 39

Organic Substances Lipids • Phospholipids • Building blocks are 1 glycerol, 2 fatty acids, and 1 phosphate per molecule • Hydrophilic and hydrophobic • Major component of cell membranes Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H H C O Fatty acid H Glycerol portion (a) A fat molecule Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. H C O Fatty acid O H C H O P O– O H H C C H H Water-insoluble (hydrophobic) “tail” H N H Water-soluble (hydrophilic) “head” Phosphate portion (b) A phospholipid molecule (the unshaded portion may vary) (c) Schematic representation of a phospholipid molecule 40

V. Nucleic Acids = DNA + RNA • Made of 3 components – 5 -C sugar – Phosphate – Nitrogen Base – Together these make 1 nucleotide – Nucleotides = Building Blocks • Functions – Carries genetic code/blueprint – Cell fuel (ATP) – Important in cell cycles

Nucleic Acids • Carry genes • Encode amino acid sequences of proteins • Building blocks are nucleotides Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. P B S • DNA (deoxyribonucleic acid) – double polynucleotide • RNA (ribonucleic acid) – single polynucleotide 42

Nucleic Acids Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. P B P S P S P S B B B B P S P S B P S (a) B S S B P B S B P B S (b) S P 43

44

Important Points in Chapter 2: Outcomes to be Assessed 2. 1: Introduction ü Give examples of how the study of living materials requires and understanding of chemistry. 2. 2: Structure of Matter ü Describe how atomic structure determines how atoms interact. ü Describe the relationships among matter, atoms, and molecules. ü Explain how molecular and structural formulas symbolize the composition of compounds. ü Describe three types of chemical reactions. ü Explain what acids, bases, and buffers are. ü Define p. H. 45

Important Points in Chapter 2: Outcomes to be Assessed Continued 2. 3: Chemical Constituents of Cells ü List the major groups of inorganic chemicals common in cells. ü Describe the general functions of the main classes of organic molecules in cells. 46
- Slides: 46