Chapter 2 Cells Tissues of Immune System CELLS



















- Slides: 43

Chapter 2 Cells & Tissues of Immune System


CELLS OF THE IMMUNE SYSTEM v The cells of the innate and adaptive immune system are normally present as circulating cells in the blood and lymph, in lymphoid organs, and as scattered cells in virtually all tissues. v The immune system faces numerous challenges to generate effective protective responses against infectious pathogens. I. The system must be able to respond rapidly to small numbers of many different microbes. II. In the adaptive immune response, very few naive lymphocytes specifically recognize and respond to any one antigen. III. The effector mechanisms of the adaptive immune system (antibodies and effector T cells) may have to locate and destroy microbes at sites that are distant from the site where the immune response was induced.

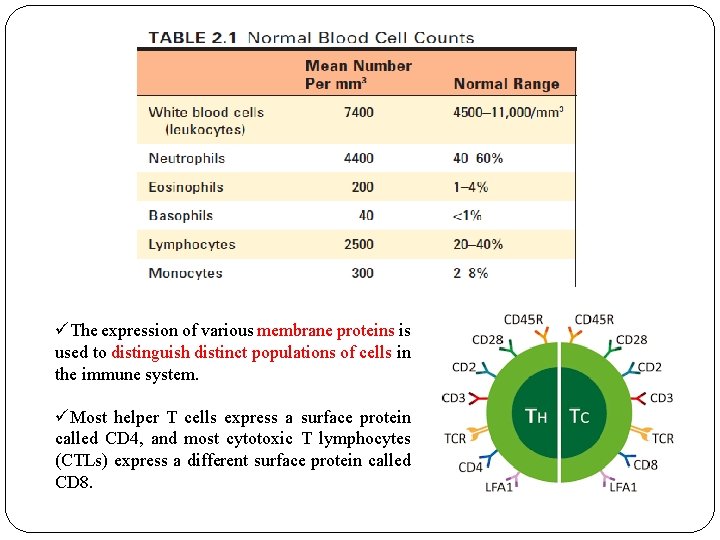
üThe expression of various membrane proteins is used to distinguish distinct populations of cells in the immune system. üMost helper T cells express a surface protein called CD 4, and most cytotoxic T lymphocytes (CTLs) express a different surface protein called CD 8.

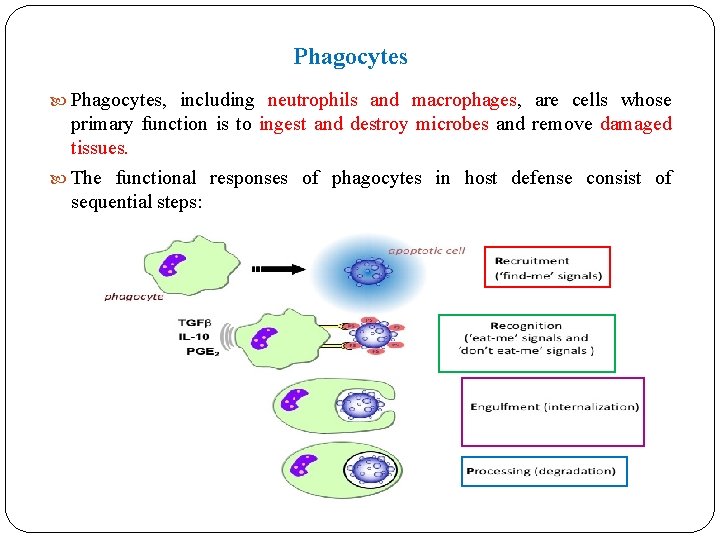
Phagocytes Phagocytes, including neutrophils and macrophages, are cells whose primary function is to ingest and destroy microbes and remove damaged tissues. The functional responses of phagocytes in host defense consist of sequential steps:

ü Polymorphonuclear leukocytes (PMN), nucleus with three to five connected lobules. ü Most abundant population of circulating WBC. ü The cytoplasm contains granules of two types, specific granules (enzyme such as lysozyme, collagenase, and elastase) that do not stain strongly with either basic or acidic dyes and azurophilic granules (containing enzymes and other microbicidal substances, including defensins and cathelicidins). Production of neutrophils is stimulated by granulocyte colony-stimulating factor (G-CSF) and granulocytemacrophage colony-stimulating factor (GM-CSF). ü An adult human produces more than 1 x 1011 neutrophils per day, Function for a few hours (6 h) and then die. Neutrophils

Mononuclear Phagocytes (MMN) The mononuclear phagocyte system includes circulating cells called monocytes, which become macrophages when they migrate into tissues, and tissue resident macrophages. Driven by a cytokine called monocyte (or macrophage) colony-stimulating factor (MCSF). üPlay central roles in innate and adaptive immunity üUnlike neutrophils, macrophages are not terminally differentiated and can division at an inflammatory site. üIncompletely differentiated is called monocyte which is divided to classical monocytes (CD 14+CD 16 -) and non-classical monocytes (CD 14+CD 16+)

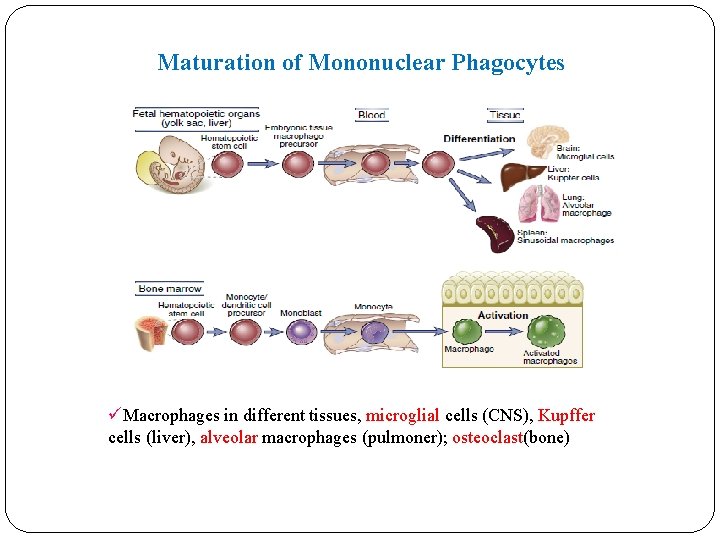
Maturation of Mononuclear Phagocytes üMacrophages in different tissues, microglial cells (CNS), Kupffer cells (liver), alveolar macrophages (pulmoner); osteoclast(bone)

Several important functions of MQ in innate and adaptive immunity: üA major function of macrophages in host defense is to ingest and kill microbes. üIngest dead host cells as part of the cleaning up process after infection or sterile tissue injury. ü Secrete several different cytokines that act on endothelial cells lining blood vessels to enhance the recruitment of more monocytes and other leukocytes from the blood into sites of infections. üMacrophages serve as APCs üPromote repair of damaged tissues by stimulating new blood vessel growth (angiogenesis) and synthesis of collagen-rich extracellular matrix (fibrosis). Recognizie many different kinds of microbial molecules by different receptors such as (Toll-like receptors).

Subsets of Macrophages can acquire distinct functional capabilities, depending on the types of activating stimuli. Some of these cytokines activate macrophages to become efficient at killing microbes, called classical activation (M 1). Other cytokines activate macrophages to promote tissue remodeling and repair, called alternative activation (M 2). Some develop abundant cytoplasm and are called epithelioid cells. Activated macrophages can fuse to form multinucleated giant cells, which occurs frequently in certain types of microbial infections, such as with mycobacteria, and in response to indigestible foreign bodies.

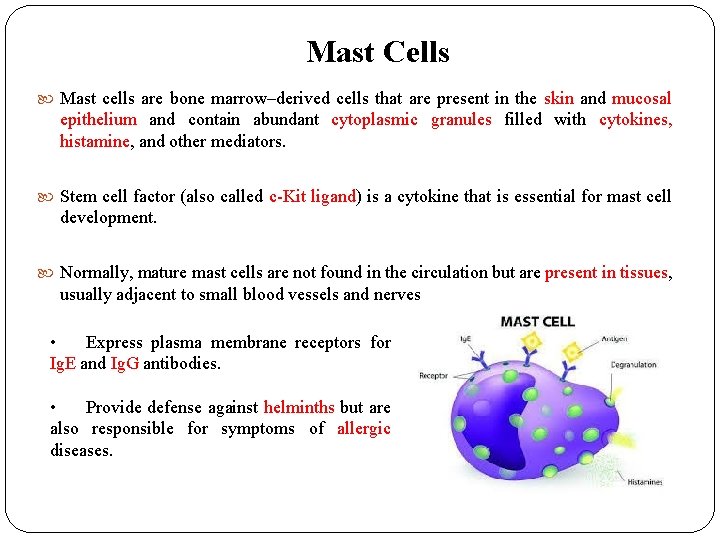
Mast Cells Mast cells are bone marrow–derived cells that are present in the skin and mucosal epithelium and contain abundant cytoplasmic granules filled with cytokines, histamine, and other mediators. Stem cell factor (also called c-Kit ligand) is a cytokine that is essential for mast cell development. Normally, mature mast cells are not found in the circulation but are present in tissues, usually adjacent to small blood vessels and nerves • Express plasma membrane receptors for Ig. E and Ig. G antibodies. • Provide defense against helminths but are also responsible for symptoms of allergic diseases.

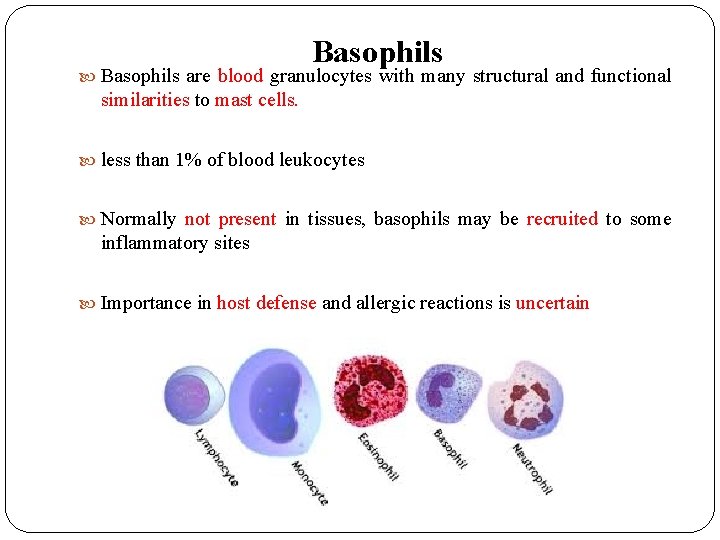
Basophils are blood granulocytes with many structural and functional similarities to mast cells. less than 1% of blood leukocytes Normally not present in tissues, basophils may be recruited to some inflammatory sites Importance in host defense and allergic reactions is uncertain

Eosinophils Blood granulocytes express cytoplasmic granules containing enzymes that are harmful to the cell walls of parasites but can also damage host tissues. Eosinophils are bone marrow–derived and circulate in the blood, from where they may be recruited into tissues. The cytokines GM-CSF, interleukin-3 (IL-3), and IL-5 promote eosinophil maturation from myeloid precursors.

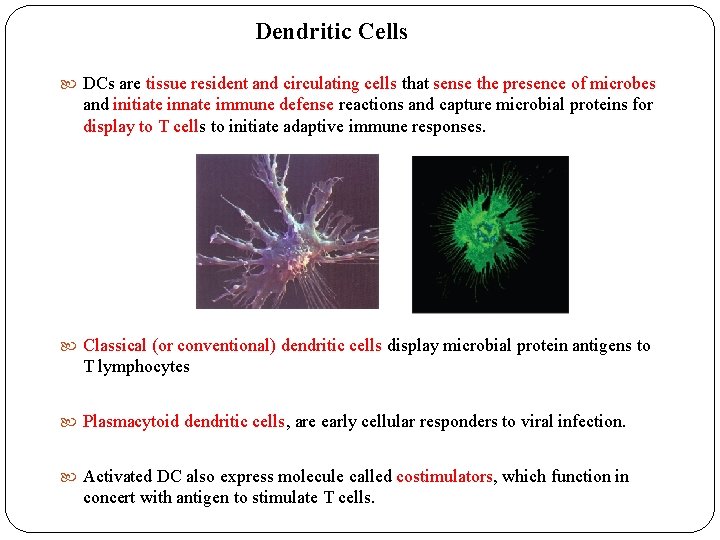
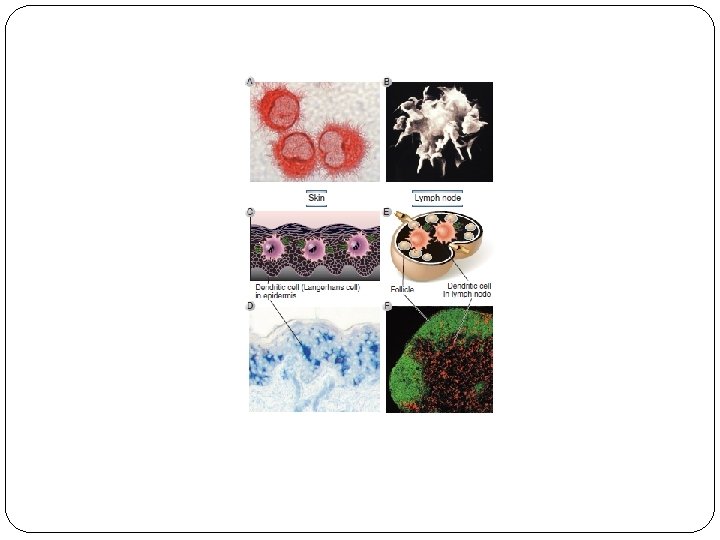
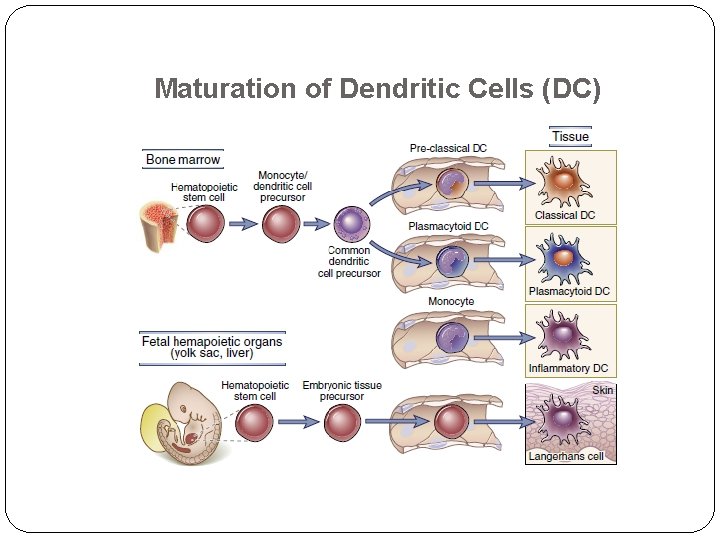
Dendritic Cells DCs are tissue resident and circulating cells that sense the presence of microbes and initiate innate immune defense reactions and capture microbial proteins for display to T cells to initiate adaptive immune responses. Classical (or conventional) dendritic cells display microbial protein antigens to T lymphocytes Plasmacytoid dendritic cells, are early cellular responders to viral infection. Activated DC also express molecule called costimulators, which function in concert with antigen to stimulate T cells.


Maturation of Dendritic Cells (DC)

Follicular Dendritic Cells (FDCs) FDCs are present involve in specialized collections of activated B cells, called germinal centers. FDCs are not derived from precursors in the bone marrow and are unrelated to the dendritic cells FDCs trap antigen-antibodies complexes to or complement products for recognition by B lymphocytes. Important for the selection of activated B lymphocytes.

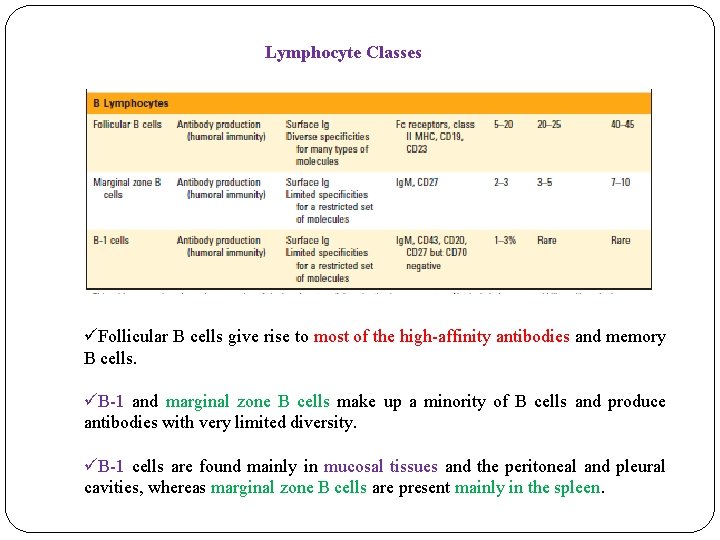
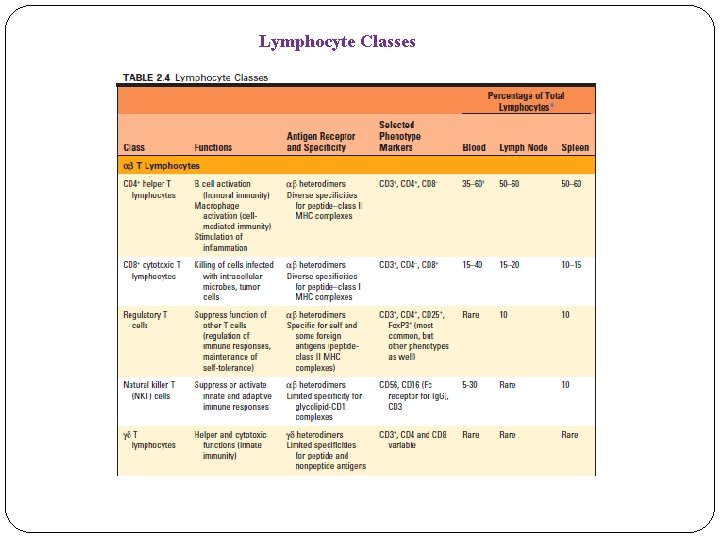
Lymohocytes Lymphocytes are the only cells in the body capable of specifically recognizing and distinguishing different antigenic determinants and are responsible for the two defining characteristics of the adaptive immune response, specificity and memory Major subsets of B cells (B 2 cell) are follicular B cells, marginal zone B cells, and B 1 cells. T cell subsets are helper T lymphocytes (Th CD 4+), cytotoxic T lymphocytes (CTLs), which express an antigen receptor called the αβ receptor, CD 4+ regulatory T cells , and γδ T cells. NKT cells are a numerically small population of lymphocytes that share characteristic of both NK cells and T cells.

Lymphocyte Classes üFollicular B cells give rise to most of the high-affinity antibodies and memory B cells. üB-1 and marginal zone B cells make up a minority of B cells and produce antibodies with very limited diversity. üB-1 cells are found mainly in mucosal tissues and the peritoneal and pleural cavities, whereas marginal zone B cells are present mainly in the spleen.

Lymphocyte Classes

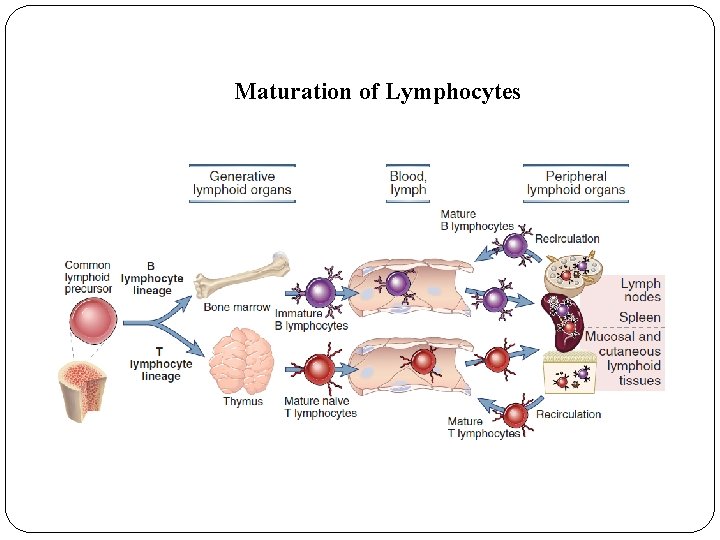
Maturation of Lymphocytes

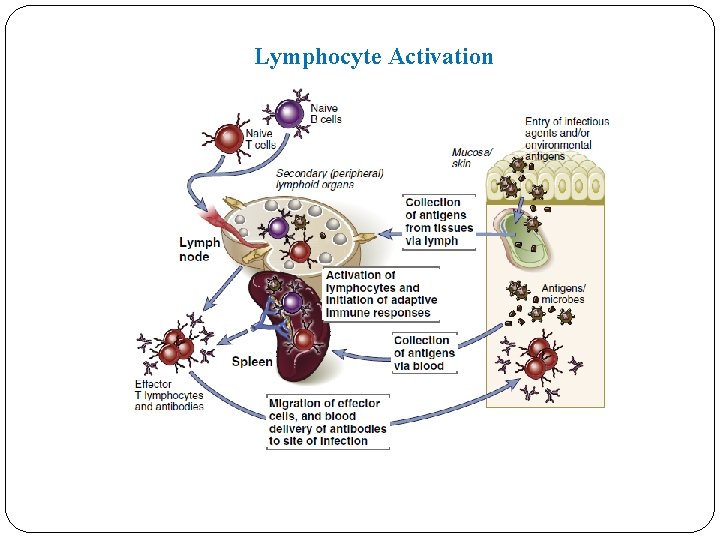
Lymphocyte Activation



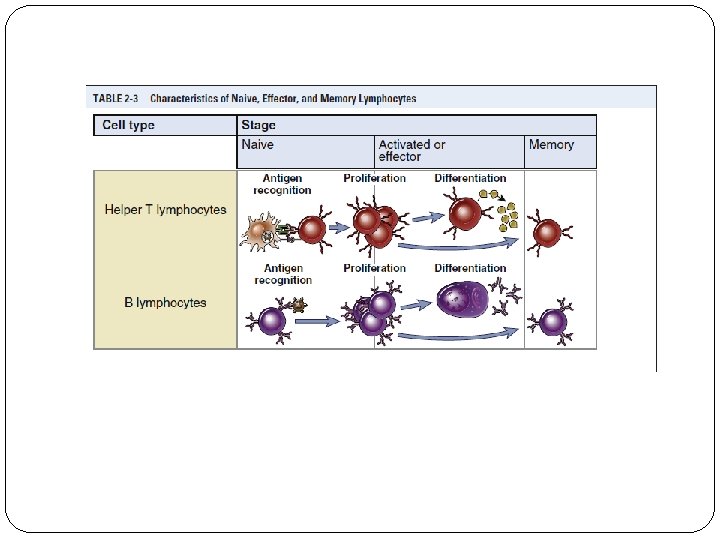
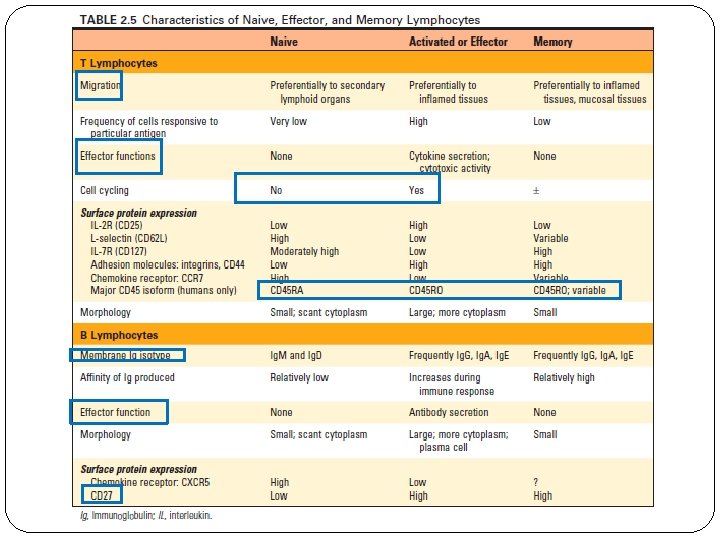
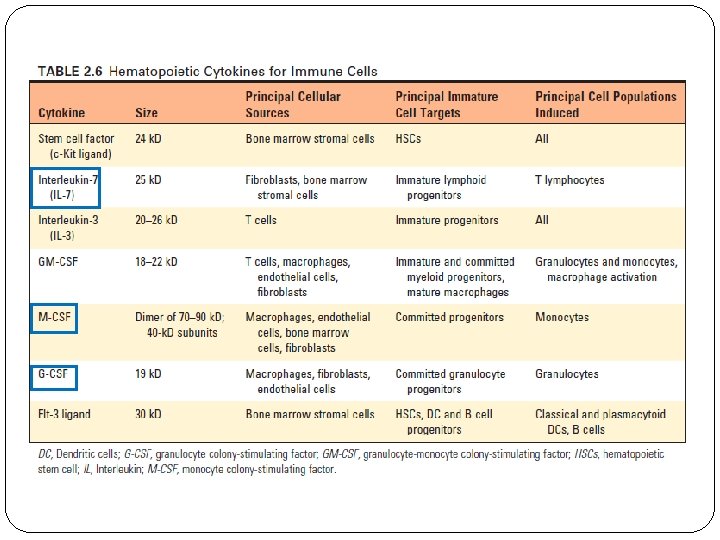
Naive Lymphocytes v Are mature T or B cell emigrants from generative lymphoid organs that have never encountered foreign antigen. v Naive lymphocytes are found in the circulation and peripheral lymphoid organs. v Naive and memory lymphocytes are both called resting lymphocytes because they are not actively dividing, nor are they performing effector functions. v Die after 1 to 3 months, if they do not recognize antigens. v Cytokines are also essential for the survival of naïve lymphocytes. The most important of these cytokines are IL-7 and B cell–activating factor (BAFF), which are required for naive T and B cell survival, respectively.

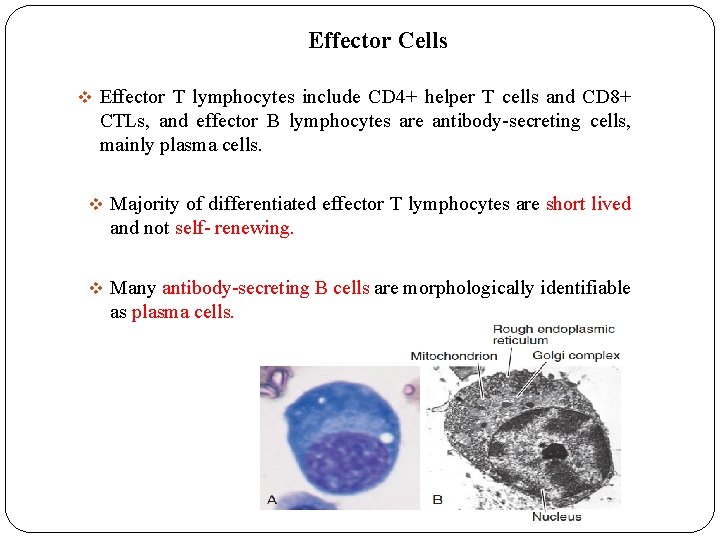
Effector Cells v Effector T lymphocytes include CD 4+ helper T cells and CD 8+ CTLs, and effector B lymphocytes are antibody-secreting cells, mainly plasma cells. v Majority of differentiated effector T lymphocytes are short lived and not self- renewing. v Many antibody-secreting B cells are morphologically identifiable as plasma cells.

Memory Cells May survive in a functionally quiescent or slowly cycling state for months or years after the microbe is eliminated. Identified by their expression of surface proteins although it is still not clear which definitive markers of memory populations. In humans, most naive T cells express a 200 -k. D isoform of a surface molecule called CD 45 RA (for restricted A). In contrast, most activated and memory T cells express a 180 -k. D isoform of CD 45 RO. Memory B lymphocytes express certain classes of membrane Ig, such as Ig. G, Ig. E, or Ig. A, whereas naive B cells express only Ig. M and Ig. D. In humans, CD 27 expression is a good marker for memory B cells.


Natural Killer Cells and Cytokine-Secreting Innate Lymphoid Cells v Bone marrow–derived cells with lymphoid morphology and effector functions similar to those of T cells but lacking T cell antigen receptors. v Natural killer (NK) cells have cytotoxic effector functions similar to CD 8+ CTLs. They circulate in the blood and are present in various lymphoid tissues. v Cytokine-secreting innate lymphoid cells (ILCs) cells have similar effector functions as CD 4+ helper T cells. v These ILCs can be grouped into three major subsets based on the cytokines they secrete. v ILCs are rare in the blood and are present mostly in tissues, especially mucosal tissues such as the lung and intestines.

ANATOMY AND FUNCTIONS OF LYMPHOID TISSUES

v Lymphoid tissues are classified as generative Lymphoid organs and Peripheral Lymphoid organs. v Generative organs or primary lymphoid organs (Thymus and BM), where lymphocytes first express antigen receptors and attain phenotypic and functional maturity. v Peripheral organs or secondary lymphoid organs (lymph nodes, spleen, and components of the mucosal immune system …) where lymphocyte responses to foreign antigens are initiated and develop. v Two important functions shared by the generative organs are to provide growth factors and other molecular signals needed for lymphocyte maturation and to present self antigens for recognition and selection of maturing lymphocytes.

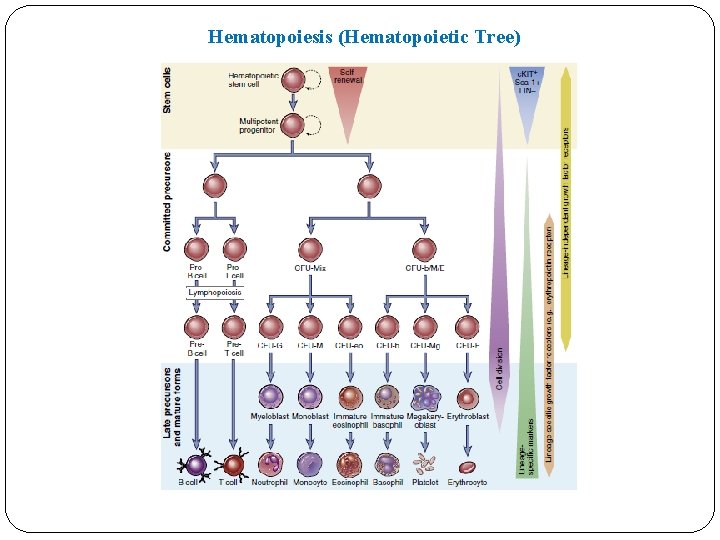
Bone Marrow v The bone marrow is the site of generation of circulating blood cells, including red blood cells, granulocytes, and monocytes, and the site of B cell maturation. v Hematopoiesis occurs initially during fetal development in the yolk sac, then shifts to the liver between the third and fourth months of gestation, and finally shifts to the bone marrow. v Red blood cells, granulocytes, monocytes, dendritic cells, mast cells, platelets, B and T lymphocytes, and ILCs all originate from a common hematopoietic stem cell (HSC) in the bone marrow. v HSCs are multipotent and self-renewing cells. v The proliferation and maturation of precursor cells in the bone marrow are stimulated by cytokines (colony-stimulating factors ). v Hematopoietic cytokines are produced by stromal cells and macrophages in the bone marrow.

Hematopoiesis (Hematopoietic Tree)


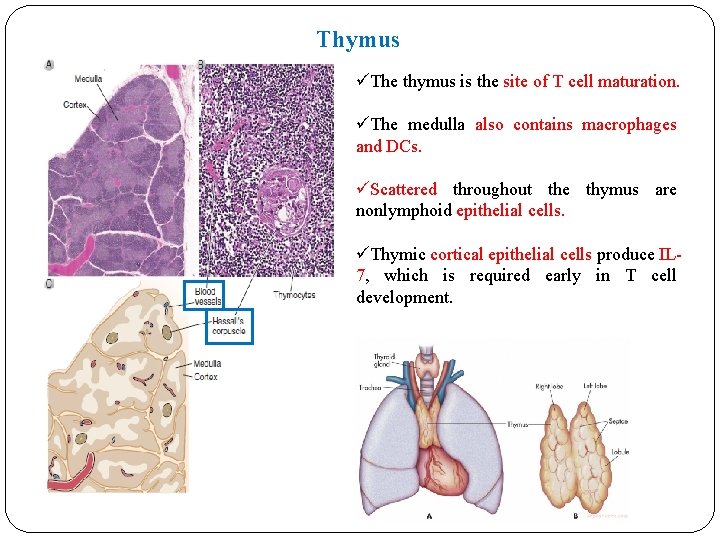
Thymus üThe thymus is the site of T cell maturation. üThe medulla also contains macrophages and DCs. üScattered throughout the thymus are nonlymphoid epithelial cells. üThymic cortical epithelial cells produce IL 7, which is required early in T cell development.

v Medullary thymic epithelial cells (MTECs), plays a special role in presenting self antigens to developing T cells and causing their elimination. v An inherited disorder of T cell immunity caused by failure of development of the thymus is called the Di. George syndrome. v These patients suffer from T cell deficiency because of a chromosomal deletion that eliminates genes required for thymus development. v The lymphocytes in the thymus, also called thymocytes, are T cells at various stages of maturation. v The most immature cells enter the thymus, and their maturation begins in the cortex.

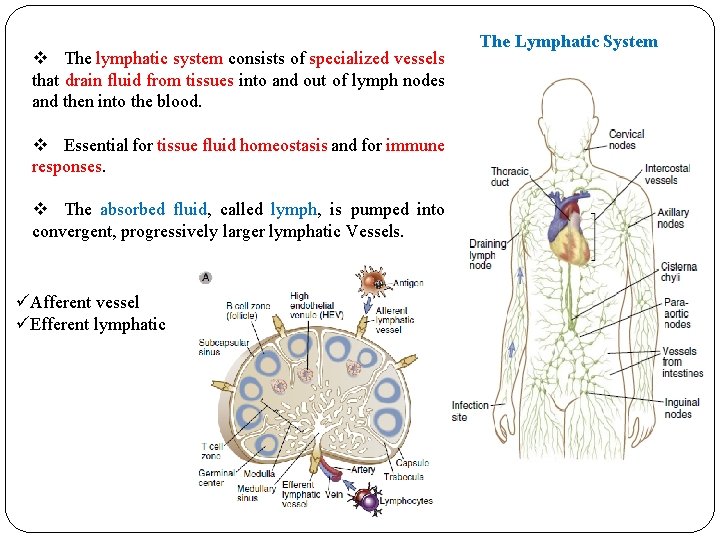
v The lymphatic system consists of specialized vessels that drain fluid from tissues into and out of lymph nodes and then into the blood. v Essential for tissue fluid homeostasis and for immune responses. v The absorbed fluid, called lymph, is pumped into convergent, progressively larger lymphatic Vessels. üAfferent vessel üEfferent lymphatic The Lymphatic System

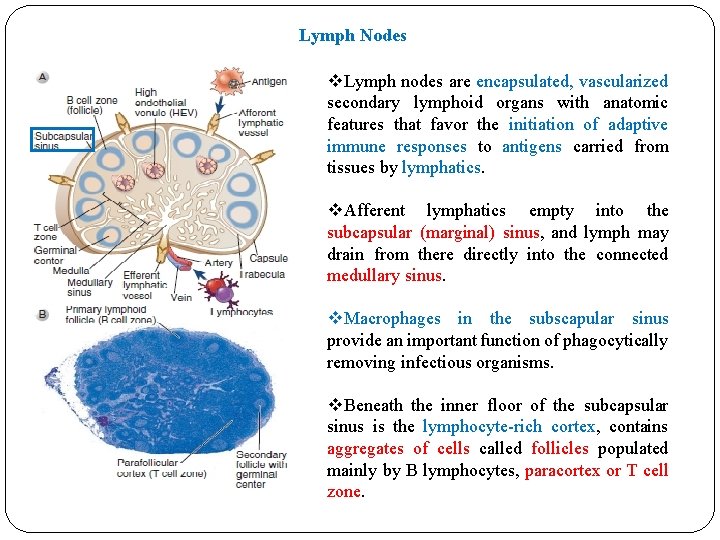
Lymph Nodes v. Lymph nodes are encapsulated, vascularized secondary lymphoid organs with anatomic features that favor the initiation of adaptive immune responses to antigens carried from tissues by lymphatics. v. Afferent lymphatics empty into the subcapsular (marginal) sinus, and lymph may drain from there directly into the connected medullary sinus. v. Macrophages in the subscapular sinus provide an important function of phagocytically removing infectious organisms. v. Beneath the inner floor of the subcapsular sinus is the lymphocyte-rich cortex, contains aggregates of cells called follicles populated mainly by B lymphocytes, paracortex or T cell zone.

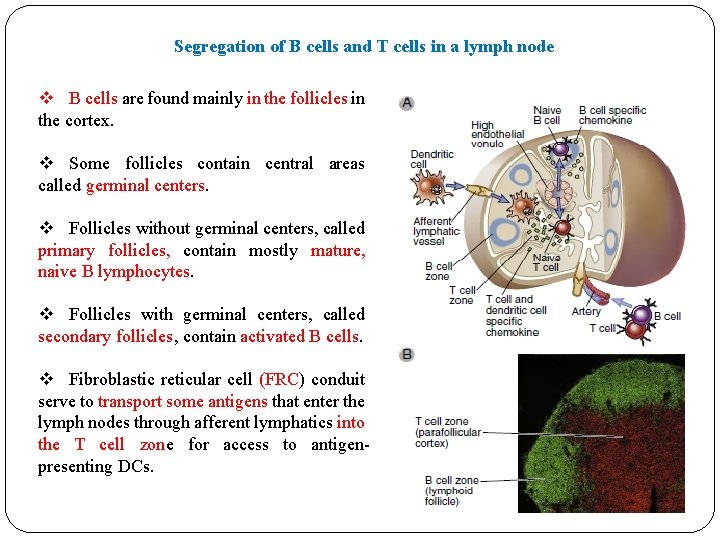
Segregation of B cells and T cells in a lymph node v B cells are found mainly in the follicles in the cortex. v Some follicles contain central areas called germinal centers. v Follicles without germinal centers, called primary follicles, contain mostly mature, naive B lymphocytes. v Follicles with germinal centers, called secondary follicles, contain activated B cells. v Fibroblastic reticular cell (FRC) conduit serve to transport some antigens that enter the lymph nodes through afferent lymphatics into the T cell zone for access to antigenpresenting DCs.

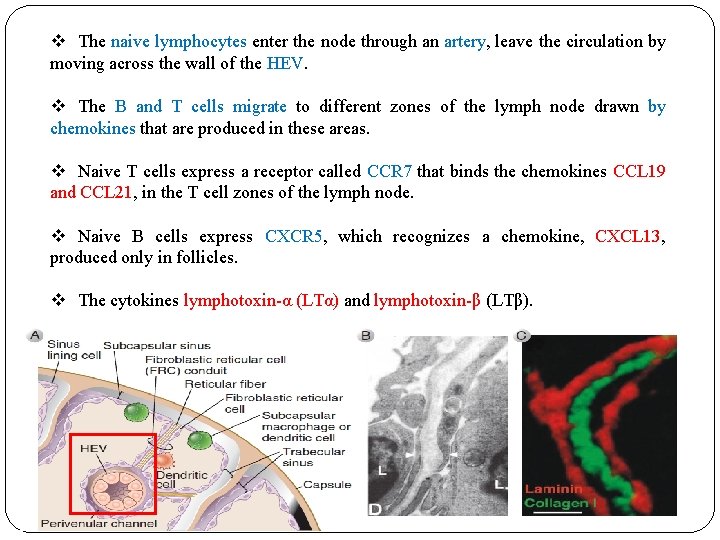
v The naive lymphocytes enter the node through an artery, leave the circulation by moving across the wall of the HEV. v The B and T cells migrate to different zones of the lymph node drawn by chemokines that are produced in these areas. v Naive T cells express a receptor called CCR 7 that binds the chemokines CCL 19 and CCL 21, in the T cell zones of the lymph node. v Naive B cells express CXCR 5, which recognizes a chemokine, CXCL 13, produced only in follicles. v The cytokines lymphotoxin-α (LTα) and lymphotoxin-β (LTβ).

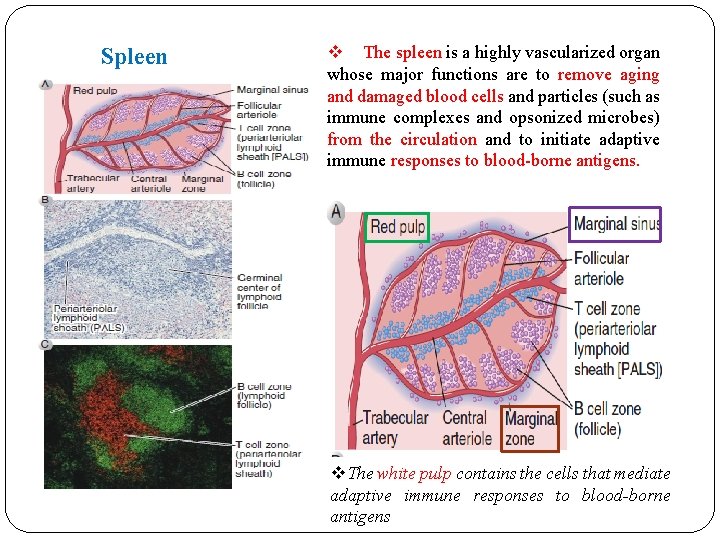
Spleen v The spleen is a highly vascularized organ whose major functions are to remove aging and damaged blood cells and particles (such as immune complexes and opsonized microbes) from the circulation and to initiate adaptive immune responses to blood-borne antigens. v. The white pulp contains the cells that mediate adaptive immune responses to blood-borne antigens

v The red pulp macrophages serve as an important filter for the blood, removing microbes, damaged cells, and antibody-coated (opsonized) cells and microbes. v In the white pulp are many collections of densely packed lymphocytes, which appear as white nodules. v A region of specialized cells surrounding the marginal sinus, called the marginal zone, forms the boundary between the red pulp and white pulp. v T cell zone: periarteriolar lymphoid sheaths (PALS). v B cell–rich follicles occupy the space between the marginal sinus and the periarteriolar sheath.
