Chapter 2 Atoms Molecules and Ions 2 2











































- Slides: 50

Chapter 2 Atoms, Molecules, and Ions

2. 2 Fundamental Chemical Laws • Atoms and molecules faithfully obey certain sets of laws that enable the study of chemistry in a systematic way. • There are three basic laws that lay down the foundation for the study of chemistry. 1. Law of Conservation of Mass 2. Law of Definite Proportions 3. Law of Multiple Proportions

Law of Conservation of Mass No detectable gain or loss in mass occurs in chemical reactions. However, the state of a substance may change in a chemical reaction. For example, substances involving in a chemical reaction can change from solid states to gaseous states but the total mass will not change.

Law of Definite Proportion ( Composition) • The elements in a given compound are always combined in the same proportion by mass. The quantity present of that compound will not in anyway change or alter this ratio. This law forms the basis for the definition of a chemical compound. • It is also known as , “Dalton’s Law”

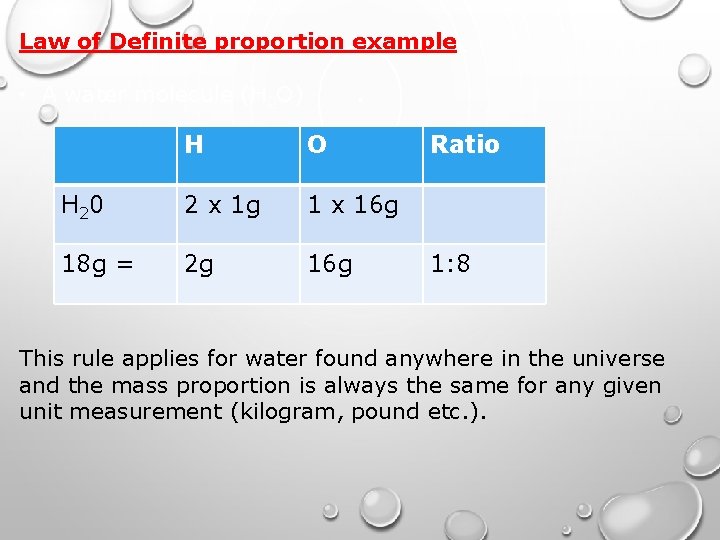
Law of Definite proportion example: • A water molecule (H 2 O) H O H 20 2 x 1 g 1 x 16 g 18 g = 2 g 16 g Ratio 1: 8 This rule applies for water found anywhere in the universe and the mass proportion is always the same for any given unit measurement (kilogram, pound etc. ).

Law of Multiple Proportion(Proust) • When the same two elements combine to form more than one compound, the ratios of the MASS OF ONE ELEMENT in the first compound to ITS MASS in the second compound, ( as it combines with the same mass of the other element) can always be expressed as ratios of small whole numbers.

Example: • Carbon combines with oxygen to make Carbon monoxide and Carbon dioxide Mass of Carbon Mass of Oxygen CO 12. 00 16. 00 CO 2 12. 00 32. 00 Ratio of O in CO 2 to CO 2: 1

2. 3 JOHN DALTON ) A Quaker schoolmaster (became a teacher at the age of 12) who studied all sciences, but made his greatest contributions in chemistry. Developed Atomic Theory and Law of Multiple Proportions. Atomic Theory helped to explain many of the observations that scientists were making. Law of Multiple Proportions helped to explain that 2 elements could combine to form more than 1 compound; for example CO and CO 2.

DALTON’S ATOMIC THEORY • 1. ELEMENTS ARE MADE UP OF ATOMS WHICH ARE INDIVISIBLE. • 2. ATOMS OF THE SAME ELEMENT ARE IDENTICAL. ATOMS OF DIFFERENT ELEMENTS ARE DIFFERENT. • 3. COMPOUNDS ARE FORMED WHEN ATOMS COMBINE. EACH COMPOUND HAS A SPECIFIC NUMBER AND KIND OF ATOM. • 4. CHEMICAL REACTIONS ARE A REARRANGEMENT OF ATOMS ARE NOT CREATED OR DESTROYED.

INDIVISIBLE? • WELL, DALTON DID THIS WORK IN THE EARLY 1800’S. • WE KNOW THAT ATOMS ARE COMPOSED OF PROTONS, NEUTRONS AND ELECTRONS. DALTON DIDN’T KNOW ABOUT THEM—THEY HADN’T BEEN DISCOVERED YET! • HOWEVER, THE ATOM IS “THE SMALLEST PART OF AN ELEMENT THAT RETAINS THE PROPERTIES OF THAT ELEMENT. ” • SO AN ATOM OF GOLD IS STILL GOLD AND IS DIFFERENT FROM AN ATOM OF CARBON.

2. 4 JJ THOMPSON In 1897, Thompson discovered the electron. Electrons are negatively charged and have almost no mass at all, compared to a proton. Thompson revised Dalton’s model of the atom with one of his own, called the “Plum Pudding Model. ”

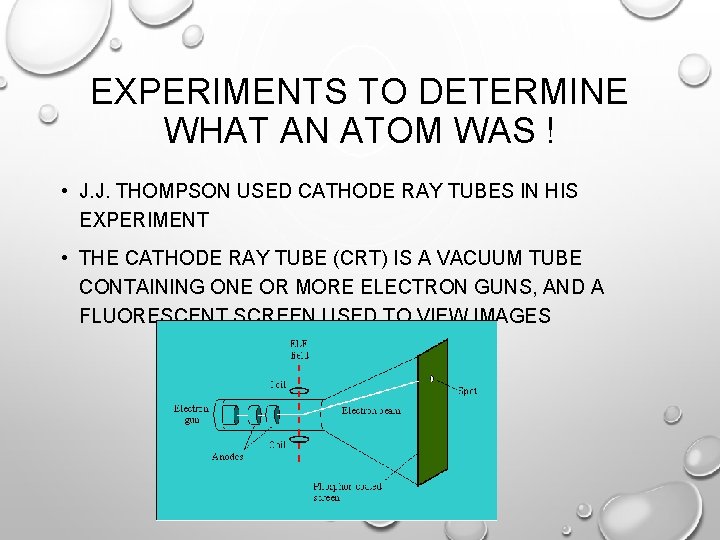
EXPERIMENTS TO DETERMINE WHAT AN ATOM WAS ! • J. J. THOMPSON USED CATHODE RAY TUBES IN HIS EXPERIMENT • THE CATHODE RAY TUBE (CRT) IS A VACUUM TUBE CONTAINING ONE OR MORE ELECTRON GUNS, AND A FLUORESCENT SCREEN USED TO VIEW IMAGES

Thomson’s Experiment Voltage source n + Passing an electric current makes a beam appear to move from the negative to the positive end.

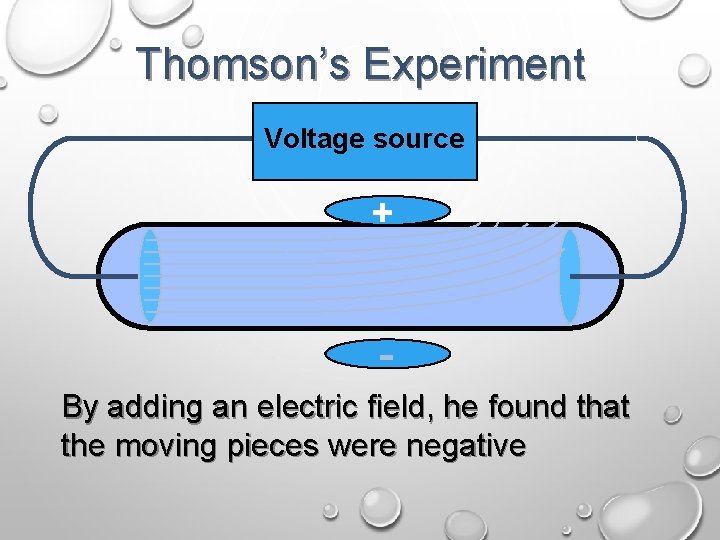
Thomson’s Experiment Voltage source + By adding an electric field, he found that the moving pieces were negative

THOMPSON’S PLUM PUDDING MODEL • FOUND THE ELECTRON. • ATOM WAS LIKE PLUM PUDDING. • A BUNCH OF POSITIVE STUFF, WITH THE ELECTRONS ABLE TO BE REMOVED. • PLUM PUDDING IS A BRITISH DESSERT IN WHICH PLUMS ARE SCATTERED MORE OR LESS RANDOMLY THROUGHOUT A CAKE (THE PUDDING).

ERNEST RUTHERFORD The Plum Pudding Model wouldn’t last long, because one of JJ’s former students did some experiments that forced the model to be revised again. Rutherford was from New Zealand, and like his mentor, Thompson, also won a Nobel Prize for his work. His “work” was the famous “gold foil” experiments, where he was researching alpha particles. As sometimes happened, Rutherford didn’t set out to discover what he actually did.

RUTHERFORD’S EXPERIMENT • USED URANIUM TO PRODUCE ALPHA PARTICLES. • AIMED ALPHA PARTICLES AT GOLD FOIL BY DRILLING HOLE IN LEAD BLOCK. • SINCE THE MASS IS EVENLY DISTRIBUTED IN GOLD ATOMS ALPHA PARTICLES SHOULD GO STRAIGHT THROUGH.

What he expected

What he got

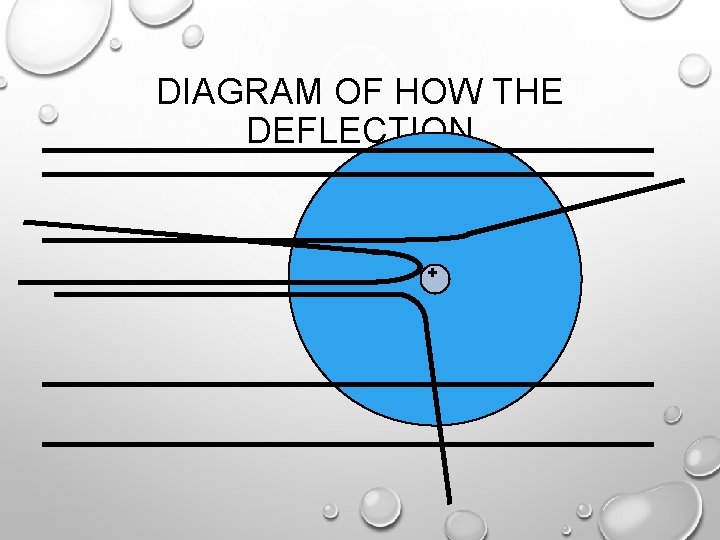
DIAGRAM OF HOW THE DEFLECTION +

How he explained it • ATOM IS MOSTLY EMPTY • SMALL DENSE, POSITIVE PIECE AT CENTER. + • ALPHA PARTICLES ARE DEFLECTED BY IT IF THEY GET CLOSE ENOUGH.

NEILS BOHR Rutherford’s nuclear model only really lasted for about 3 years, before Neils Bohr (who, oh by the way, also won a Nobel Prize for this) revised it again. Bohr asked a question: if the electrons are rotating around the nucleus, why don’t they “run out of energy. ” As they did, they would come closer and closer, attracted by the opposite charge of the nucleus, and eventually collapse onto the nucleus, destroying the atom in the process. Soccer goalie on Denmark’s 1908 Olympic team AND a Nobel Prize winner!! This doesn’t happen, and Bohr answered why. His model is usually called “the Planetary model, ” because in his model, electrons “orbit” the nucleus much as our planets orbit the Sun.

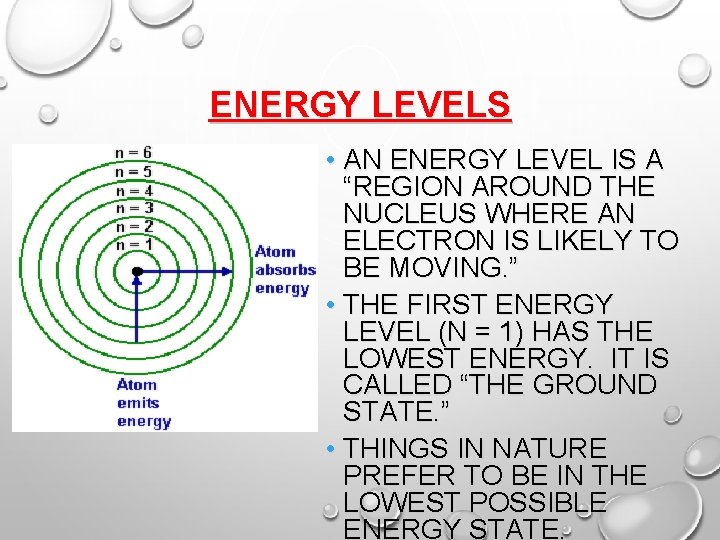
BOHR’S PLANETARY MODEL But the electrons don’t just orbit anywhere. They actually exist in orbits that Bohr called “energy levels. ” Each energy level has a certain amount of energy. Electrons can move to a higher energy level by gaining energy. Or they can drop to a lower energy level by losing (or emitting) energy.

ENERGY LEVELS • AN ENERGY LEVEL IS A “REGION AROUND THE NUCLEUS WHERE AN ELECTRON IS LIKELY TO BE MOVING. ” • THE FIRST ENERGY LEVEL (N = 1) HAS THE LOWEST ENERGY. IT IS CALLED “THE GROUND STATE. ” • THINGS IN NATURE PREFER TO BE IN THE LOWEST POSSIBLE ENERGY STATE.

MODERN VIEW • THE ATOM IS MOSTLY EMPTY SPACE. • TWO REGIONS • NUCLEUS- PROTONS AND NEUTRONS. • ELECTRON CLOUDREGION WHERE YOU MIGHT FIND AN ELECTRON.

2. 7 Periodic Table Review

2. 6 IONS THEY ARE ATOMS WITH A + OR - CHARGE. • CATIONS: POSITIVE IONS – LOST ELECTRONS • ANIONS: NEGATIVE IONS – GAIN ELECTRONS • IONIC BONDING: HELD TOGETHER BY THE OPPOSITE + AND - CHARGES. • IONIC SOLIDS ARE CALLED SALTS.

Reminder about Ions The periodic table can be used as a predictor for the ion that an element will become. Group 1 Group 2 Group 15 Group 16 Group 17 +1 cations +2 cations -3 anions -2 anions -1 anions

2. 6 POLYATOMIC IONS THEY ARE COVALENTLY BONDED ELEMENTS THAT ACT AS ONE UNIT AND THEY HAVE A CHARGE. THEY ARE USUALLY FOUND BONDED TO METALS IN THE FORMATION OF IONIC COMPOUNDS. EXAMPLES: OH- HYDROXIDE ION CO 3 -2 CARBONATE ION NO 3 - NITRATE ION …. . AND MANY MORE!

2. 8 TWO TYPES OF COMPOUNDS IONIC COMPOUNDS § METAL BONDED TO A NON METAL § METAL BONDED TO A POLYATOMIC ION. COVALENT MOLECULES § NONMETAL BONDED TO A NONMETAL

REVIEW OF COMMON METALLIC ROOTS THESE WILL PRIMARILY BE USED WHEN NAMING IONIC COMPOUNDS: H = HYDR- B = BOR- C = CARB- N = NITR- O = OX- F = FLUOR- S = SULFUR- CL = CHLOR- BR = BROM- I = IOD- P = PHOSPHOR - SI = SILC SE = SELEN- AS = ARSEN-

REMEMBER ! • MOST COMPOUNDS ARE BINARY. THIS JUST MEANS THAT THEY ARE MADE UP OF ” 2 PARTS” • THIS MEANS THAT WHEN YOU ARE NAMING COMPOUNDS THEY WILL NEED A FIRST AND A LAST NAME!

NAMING IONIC COMPOUNDS (TYPE I) Write the full name of the cation and use the root of the anion with “-ide” at the end. Example: Na. Cl : sodium and chlorine Name: sodium chloride Charges: Na +1 Cl -1 (cancel each other out) The charges of an ionic compound MUST ALWAYS balance out to zero!

METAL IONS WITH MULTIPLE CHARGES (TYPE II) Transition metals frequently form more than one type of compound. The oxidation number of the metal is indicated in the name by a ROMAN NUMERAL in parentheses following the name of the cation. Example: Cu + Copper (1) Cu. Cl: Cu+1 Cl-1 Copper(1)chloride Cu+2 copper (2) Cu. Cl 2 : Cu+2 Cl 2 copper(2)chloride

Helpful Hint! Not all transition metals are capable of forming different cations. If they only form one cation, then it is not necessary to use a roman numeral in their names. The following transition metals only have one ion. There a few others but these are the ones that you will encounter the most. Zn +2 Al +3 Ag +1 Cd +1

IONIC COMPOUNDS PRACTICE YOU MUST KNOW POLYATOMIC ION NAMES. FOR NOW, YOU MAY USE TABLE 2. 5 ON PAGE 65 OR THE CHEAT SHEET THAT PROVIDED. YOU WILL NEED TO MEMORIZE THE MORE COMMONLY USED ONES TO BE SUCCESSFUL IN THIS CLASS. WHEN NAMING, THEY ARE TREATED AS ONE UNIT ALL WORKING TOGETHER. • Ca. S • K 2 S • Al. PO 4 • K 2 SO 4 • Fe. S • Co. I 3

IONIC COMPOUNDS (MORE PRACTICE) Fe 2(C 2 O 4) Mg. O Mn. O KMn. O 4 NH 4 NO 3 KCl. O 4 Na. Cl. O 3 Cr 2 O 3 Hg 2 Cl 2

REVIEW OF GREEK PREFIXES These will be used when naming covalently bonded compounds. mono- one di- two tri- three tetra- four penta- five hexa- six hepta- seven octa- eight nona- nine deca- ten

NAMING COVALENT (MOLECULAR) COMPOUNDS (TYPE III) • FORMED BETWEEN 2 NONMETALS • NAME THE FIRST ELEMENT USING THE FULL NAME OF THE MORE METALLIC ELEMENT • FOLLOWED BY THE ROOT OF THE SECOND ELEMENT • ADD “IDE” TO THE ROOT • USE NUMERIC PREFIXES TO SHOW MANY • THE EXCEPTION : YOU DO NOT USE A PREFIX FOR THE FIRST ELEMENT WHEN THERE IS ONLY ONE OF IT. YOU WILL NEVER WRITE “MONO” IN

• CO 2 • CO • CCL 4 • N 2 O 4 • SF 6 • N 4 O 4 • P 2 O 10 • LIH • NH 4 NO 3 NAMING COVALENT COMPOUNDS

NAMING ACIDS Acids are compounds that produce H+ ions when dissolved in water. Naming an acid depends on whether or not it contains oxygen! • If the acid DOES NOT have oxygen Add the prefix hydro. The suffix -ic attached to the root of the second element • HCl • H 2 S • HF

NAMING ACIDS WITH OXYGEN IN THE FORMULA (OXYACIDS) If the formula does have oxygen in it: • Write the name of the anion, but change its ending! -ate becomes -ic acid -ite becomes -ous acid Examples: • H 2 SO 4 • H 2 SO 3 • HNO 2

WRITING FORMULAS BASED ON THE NAME OF THE COMPOUND • Ionic and covalent compounds have different sets of rules to follow. • To decide which to use, decide what the first name is identifying! If is a metal or polyatomic use ionic rules If it is a non-metal use covalent rules

WRITING IONIC FORMULAS • Charges must add up to zero. • Get charges from table, name of metal ion, or memorized from the list. • Use parenthesis to indicate multiple polyatomics.

WRITING IONIC FORMULAS • Sodium nitride • Sodium- Na is always +1 • Nitride - ide tells you it comes from the table • Nitride is N -3 • The charges will not cancel out! Use the crisscross method and move the charges down. Na +1 N -3 ……. . Na 3 N

PRACTICE WITH WRITING FORMULAS FOR IONIC COMPOUNDS • SODIUM SULFITE • CALCIUM IODIDE • LEAD (II) OXIDE • LEAD (IV) OXIDE • MERCURY (I) SULFIDE • BARIUM CHROMATE • CERIUM (IV) NITRITE

WRITING COVALENT FORMULAS THE NAME TELLS YOU HOW TO WRITE THE FORMULA !!! • SULFUR DIOXIDE • DIFLOURINE MONOXIDE • NITROGEN TRICHLORIDE • DIPHOSPHORUS PENTOXIDE

WRITING FORMULAS FOR ACIDS • You work backwards from the names. • If it has the hydro prefix in the name it has no oxygen and it will end in –ic • If there is No hydro prefix, it will end in -ate or –ite In both cases, you first write the H in the front of the formula. Next you write in the correct anion. Finally, you need to make sure that the charges are balanced

PRACTICE WRITING FORMULAS FOR BOTH TYPES OF ACIDS • Hydrofluoric acid • Dichromic acid • Carbonic acid • Hydrophosphoric acid • Perchloric acid • Phosphorous acid

UNDERSTANDING HYDRATES • Hydrates are salts that trap water crystals when they form crystals. • Both the name and the formula needs to indicate how many water molecules are trapped. • In the name we add the word hydrate with a prefix that tells us how many water molecules. • In the formula you put a dot and then write the number of molecules. Copper sulfate pentahydrate