Chapter 2 Alkanes and Cycloalkanes Based on Mc

Chapter 2. Alkanes and Cycloalkanes Based on: Mc. Murry’s Fundamental of Organic Chemistry, 4 th edition, Chapter 2

Families of Organic Compounds n Organic compounds can be grouped into families by their common structural features n We shall survey the nature of the compounds in a tour of the families in this course n This chapter deals with alkanes, compounds that contain only carbons and hydrogens, all connected exclusively by single bonds 2

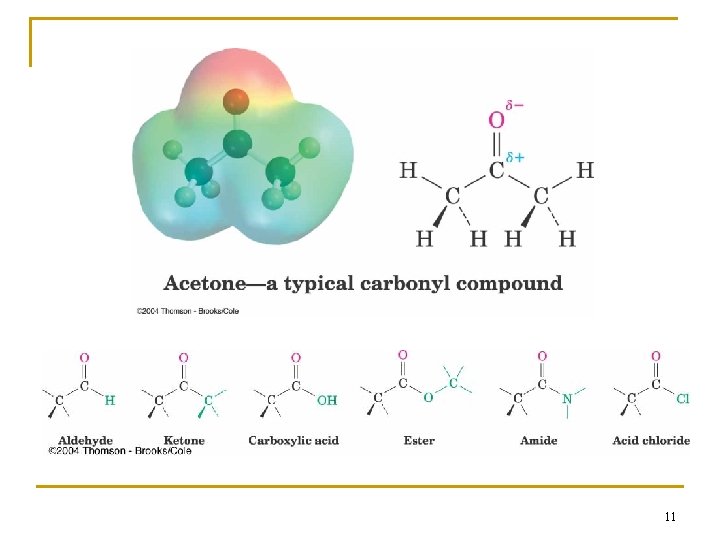
2. 1 Functional Groups n Functional group - an atom or group of atoms within a molecule that shows a characteristic set of physical and chemical properties n The group reacts in a typical way, generally independent of the rest of the molecule n For example, the double bonds in simple and complex alkenes react with bromine in the same way 3
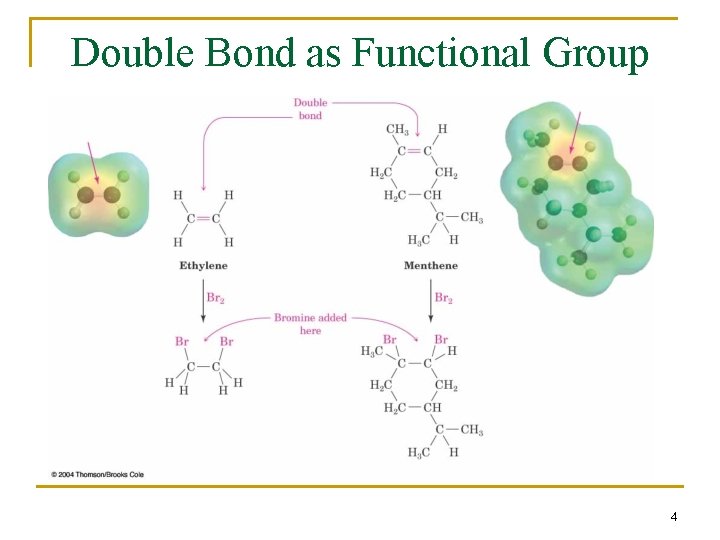
Double Bond as Functional Group 4

Survey of Functional Groups n n n Table 2. 1 (page 46 and 47) lists a wide variety of functional groups that you should recognize As you learn about them in each chapter it will be easier to recognize them The functional groups affect the reactions, structure, and physical properties of every compound in which they occur 5
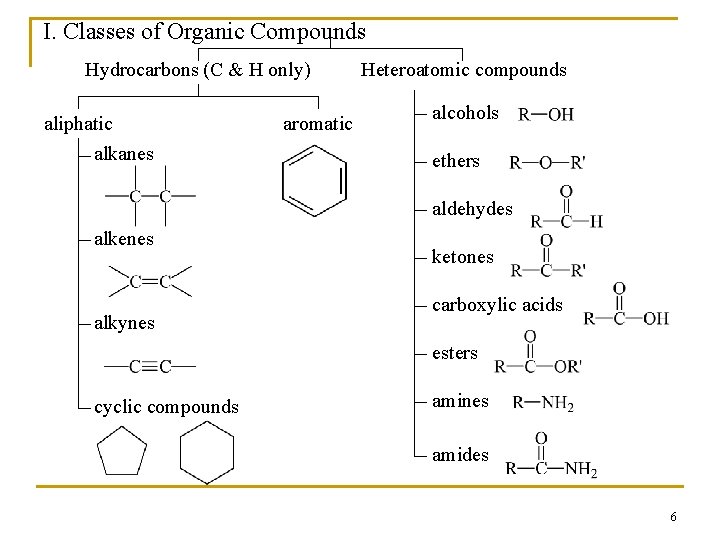
I. Classes of Organic Compounds Hydrocarbons (C & H only) aliphatic alkanes aromatic Heteroatomic compounds alcohols ethers aldehydes alkenes alkynes ketones carboxylic acids esters cyclic compounds amines amides 6

n Functional groups are important for three reason; they are 1. the units by which we divide organic compounds into classes 2. the sites of characteristic chemical reactions 3. the basis for naming organic compounds 7
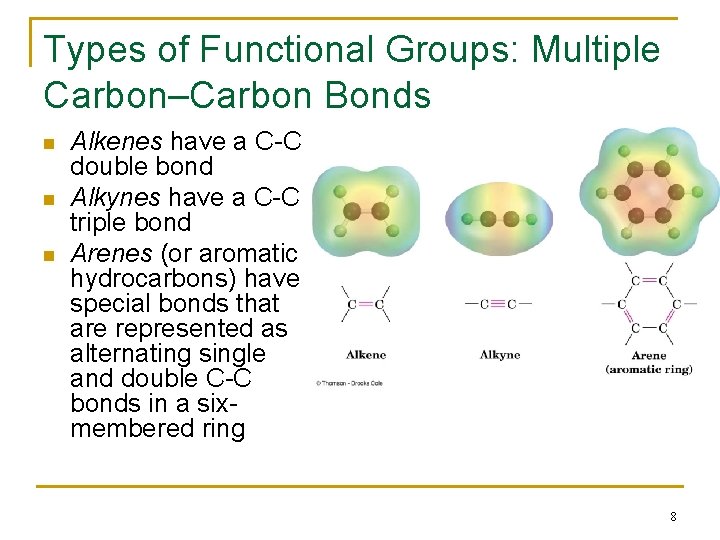
Types of Functional Groups: Multiple Carbon–Carbon Bonds n n n Alkenes have a C-C double bond Alkynes have a C-C triple bond Arenes (or aromatic hydrocarbons) have special bonds that are represented as alternating single and double C-C bonds in a sixmembered ring 8

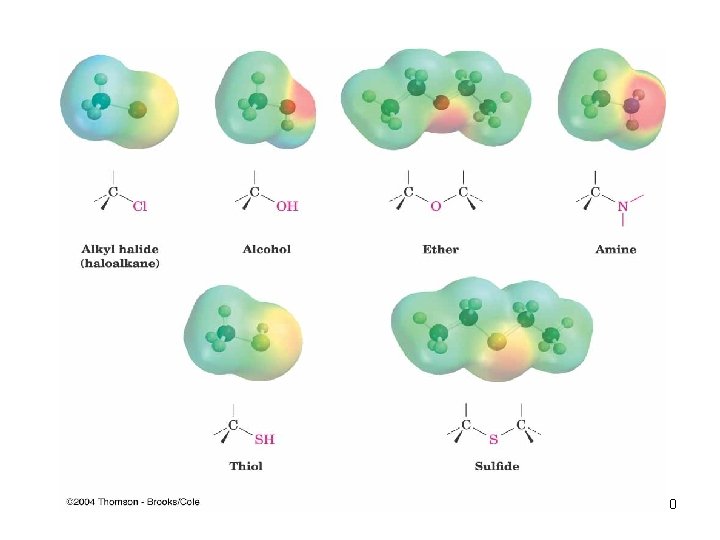
Functional Groups with Carbon Singly Bonded to an Electronegative Atom n n n n Alkyl halide: C bonded to halogen (C-X) Alcohol: C bonded O of a hydroxyl group (C�OH) Ether: Two C’s bonded to the same O (C�O�C) Amine: C bonded to N (C�N) Thiol: C bonded to SH group (C�SH) Sulfide: Two C’s bonded to same S (C�S�C) Bonds are polar, with partial positive charge on C ( +) and partial negative charge ( ) on electronegative atom 9
10
11

2. 2 Alkanes and Alkane Isomers n Alkanes: Compounds with C-C single bonds and C-H bonds only (no functional groups) n The formula for an alkane with no rings (acyclic) must be Cn. H 2 n+2 where the number of C’s is n n Alkanes are saturated with hydrogen (no more can be added) (saturated hydrocarbon) n They are also called aliphatic compounds (aleiphas=fat) 12

A “saturated” fat (glyceryl stearate): 13
Hydrocarbons Aliphatic Aromatic 14
Hydrocarbons Aliphatic Alkanes Aromatic Alkenes Alkynes 15
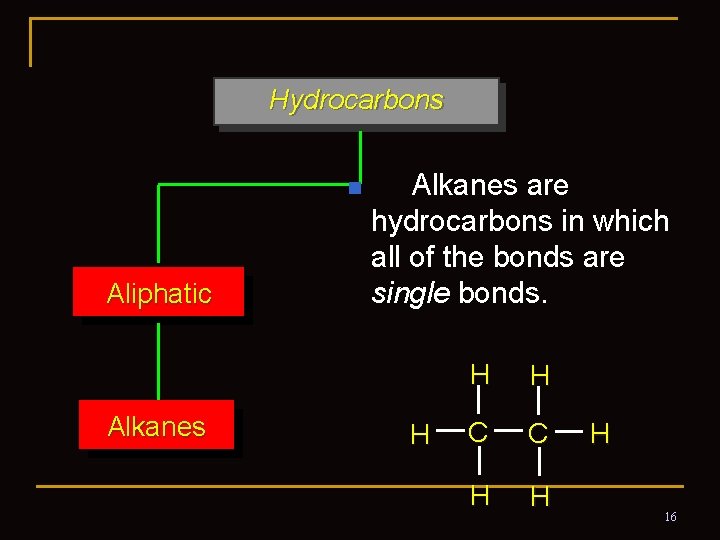
Hydrocarbons n Aliphatic Alkanes are hydrocarbons in which all of the bonds are single bonds. H H H C C H H H 16

Alkanes Recall that alkanes are aliphatic hydrocarbons having C—C and C—H bonds. They can be categorized as acyclic or cyclic. • Acyclic alkanes have the molecular formula Cn. H 2 n+2. They are also called saturated hydrocarbons because they have the maximum number of hydrogen atoms per carbon. • Cycloalkanes contain carbons joined in one or more rings. Because their general formula is Cn. H 2 n, (n = 3 or more) they have two fewer H atoms than an acyclic alkane with the same number of carbons.
Sources of Alkanes n n Natural gas 90 -95% methane Petroleum - mixture q q q n gases gasoline, kerosene ( ﺳﻔیﺪ )ﻧﻔﺖ fuel oil ( کﻮﺭﻩ )ﻧﻔﺖ lubricating oils asphalt Coal ( ﺳﻨگ )ﺫﻏﺎﻝ 18

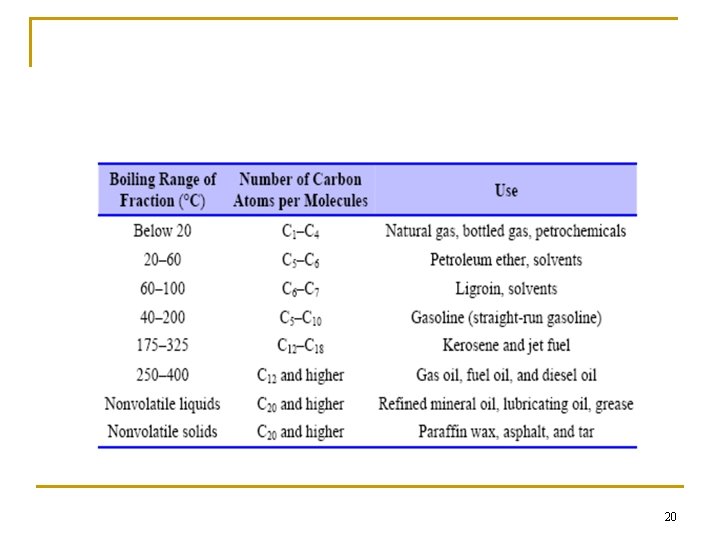
n Refining crude petroleum into usable fuel and other petroleum products. (a) An oil refinery. At an oil refinery, crude petroleum is separated into fractions of similar boiling point by the process of distillation. (b) Schematic of a refinery tower. As crude petroleum is heated, the lower-boiling, more volatile components distill first, followed by fractions of progressively higher boiling point. 19
20

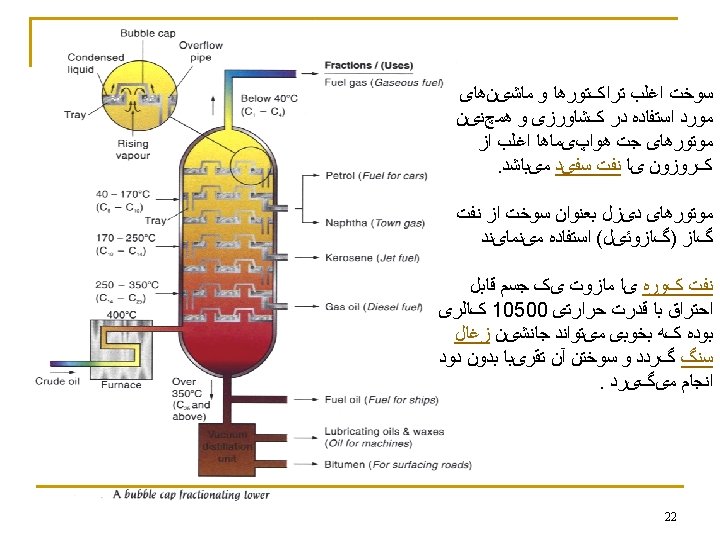

n n In cracking, high molecular weight fractions and catalysts are heated to the point where the carbon-carbon bonds break. Products of the reaction include alkenes and alkanes of lower molecular weight than were present in the original fraction. alkane C 13 H 28(l) alkane C 8 H 18(l) + alkene C 2 H 4(g) + alkene C 3 H 6(g) 23

Names of Small Hydrocarbons Parent names: CH 4 CH 3 CH 2 CH 3(CH 2)3 CH 3(CH 2)4 CH 3 n-C 7 H 16 n-C 8 H 18 n-C 9 H 20 n-C 10 H 22 methane propane butane pentane hexane heptane octane nonane decane n-C 11 H 24 n-C 12 H 26 n-C 13 H 28 n-C 14 H 30 ¦ n-C 20 H 42 n-C 30 H 62 n-C 40 H 82 ¦ etc. undecane dodecane tridecane tetradecane icosane triacontane tetracontane 25

Same Formula Alkane Isomers: Different Structure n CH 4 = methane, C 2 H 6 = ethane, C 3 H 8= propane n The molecular formula of an alkane with more than three carbons can give more than one structure. When we get to Butane, C 4 H 10, we find that there are two different structures that can share the same formula q q C 4 (butane) = butane and isobutane C 5 (pentane) = pentane, 2 -methylbutane (isopentane), and 2, 2 -dimethylpropane (neopentane) 26

3 -D Representation of Alkanes • All C atoms in an alkane are surrounded by four groups, making them sp 3 hybridized and tetrahedral, and all bond angles are 109. 50. • The three-carbon alkane CH 3 CH 2 CH 3, called propane, has a molecular formula C 3 H 8. Note in the 3 -D drawing that each C atom has two bonds in the plane (solid lines), one bond in front (on a wedge) and one bond behind the plane (on a dashed line).
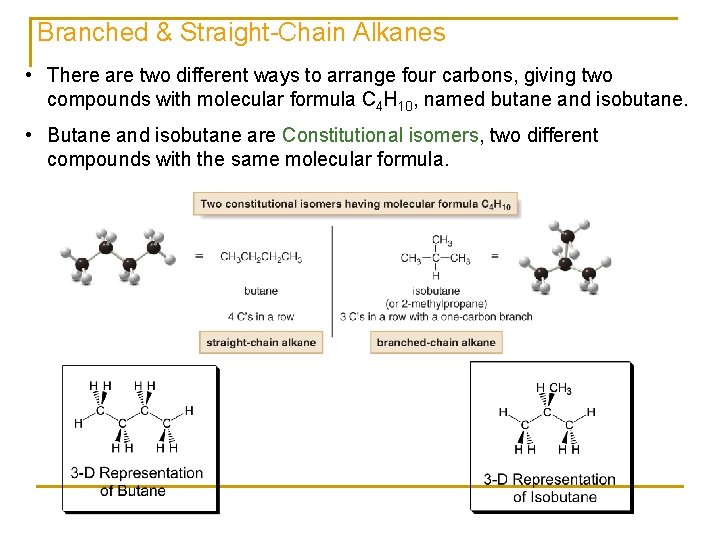
Branched & Straight-Chain Alkanes • There are two different ways to arrange four carbons, giving two compounds with molecular formula C 4 H 10, named butane and isobutane. • Butane and isobutane are Constitutional isomers, two different compounds with the same molecular formula.
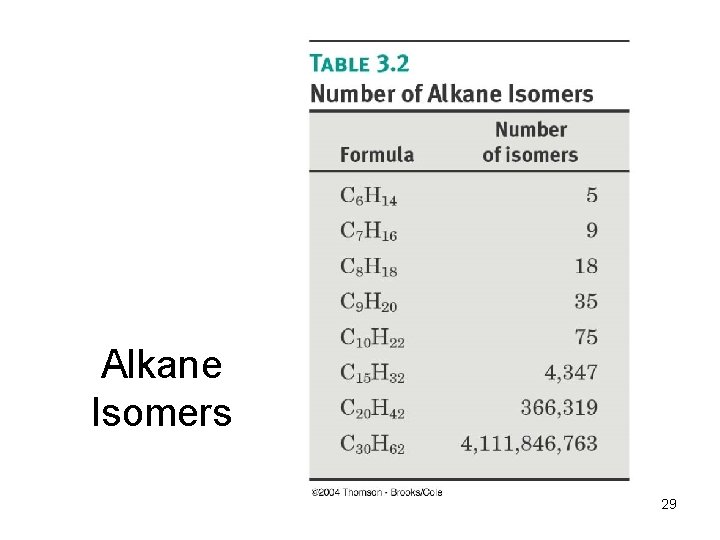
Alkane Isomers 29
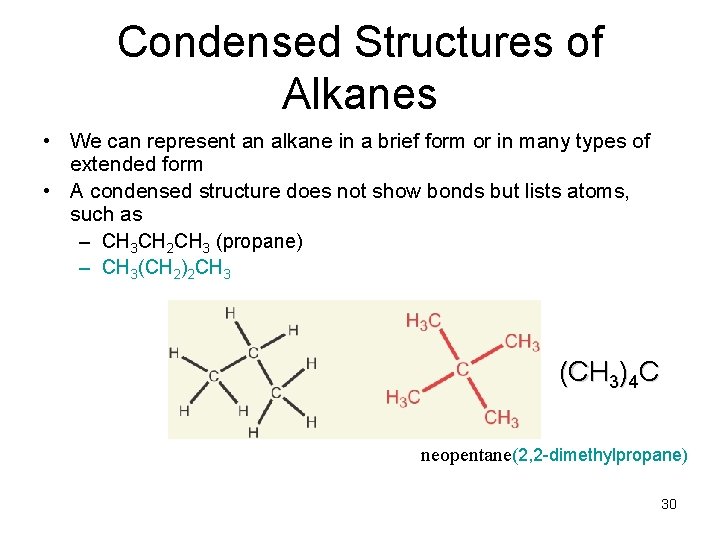
Condensed Structures of Alkanes • We can represent an alkane in a brief form or in many types of extended form • A condensed structure does not show bonds but lists atoms, such as – CH 3 CH 2 CH 3 (propane) – CH 3(CH 2)2 CH 3 (CH 3)4 C neopentane(2, 2 -dimethylpropane) 30
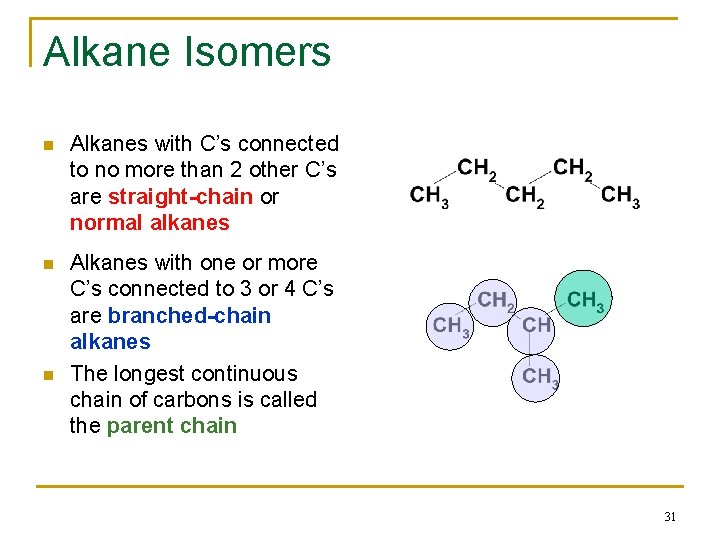
Alkane Isomers n Alkanes with C’s connected to no more than 2 other C’s are straight-chain or normal alkanes n Alkanes with one or more C’s connected to 3 or 4 C’s are branched-chain alkanes The longest continuous chain of carbons is called the parent chain n 31

Constitutional (Structural) Isomers n Isomers that differ in how their atoms are arranged in chains are called constitutional (or structural) isomers n Structural isomers ﺍیﺰﻭﻣﺮﻫﺎی ﺳﺎﺧﺘﻤﺎﻧی are molecules ﺍیﺰﻭﻣﺮﻫﺎی with the same chemical formulas but different molecular structures - different “connectivity”. n Compounds other than alkanes can also be Structural isomers of one another n They must have the same molecular formula to be isomers 32

Constitutional Isomers 33
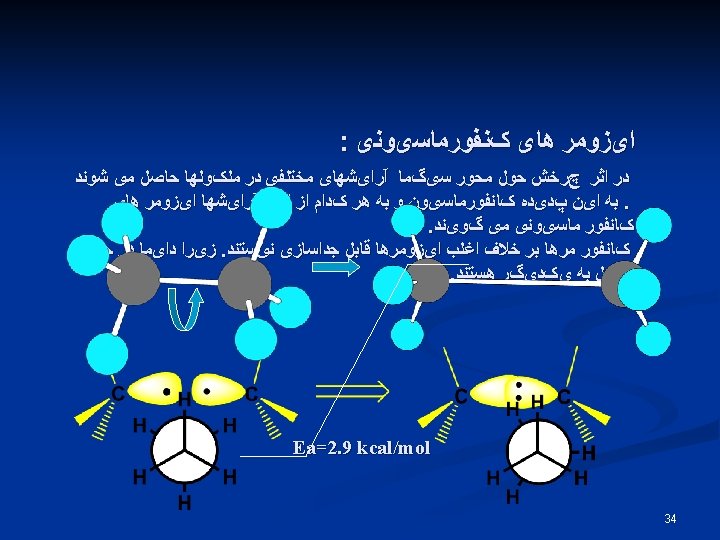
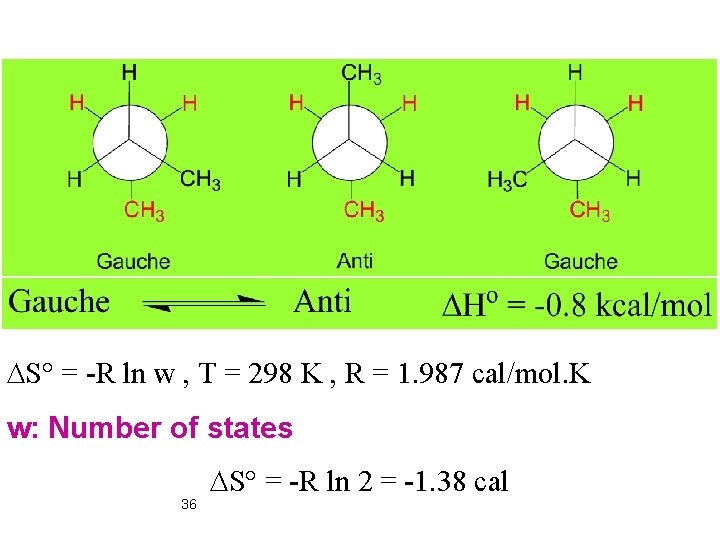
DS° = -R ln w , T = 298 K , R = 1. 987 cal/mol. K w: Number of states 36 DS° = -R ln 2 = -1. 38 cal

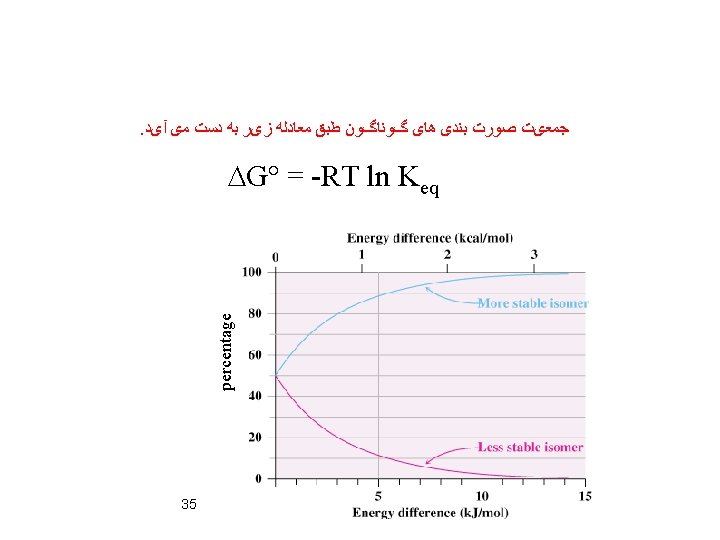
DG° = DH° - TDS° DG° = -0. 8 x 1000 – 298(-1. 38) DG° = -388. 8 cal= -0. 39 kcal DG° = -RT ln Keq = 1. 9 37 at 298 K

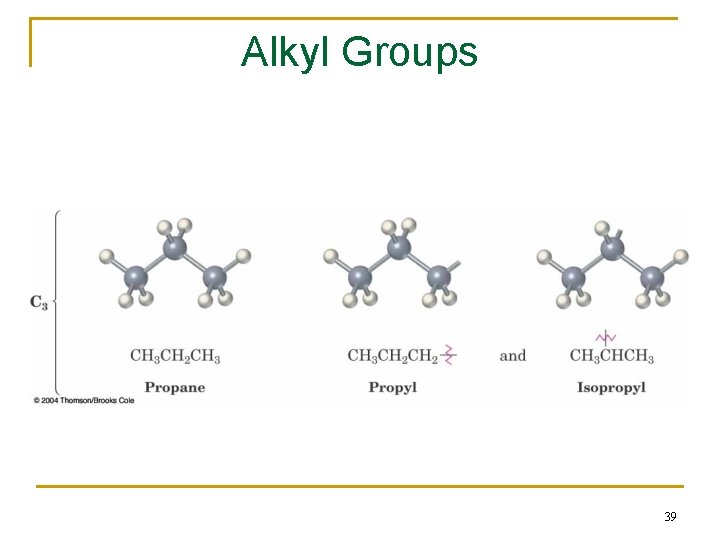
2. 3 Alkyl Groups n Alkyl group – remove one H from an alkane (a part of a structure) n General abbreviation “R” (for Radical, an incomplete species or the “rest” of the molecule) n Name: replace -ane ending of alkane with -yl ending q �CH 3 is “methyl” (from methane) q �CH 2 CH 3 is “ethyl” from ethane 38
Alkyl Groups 39
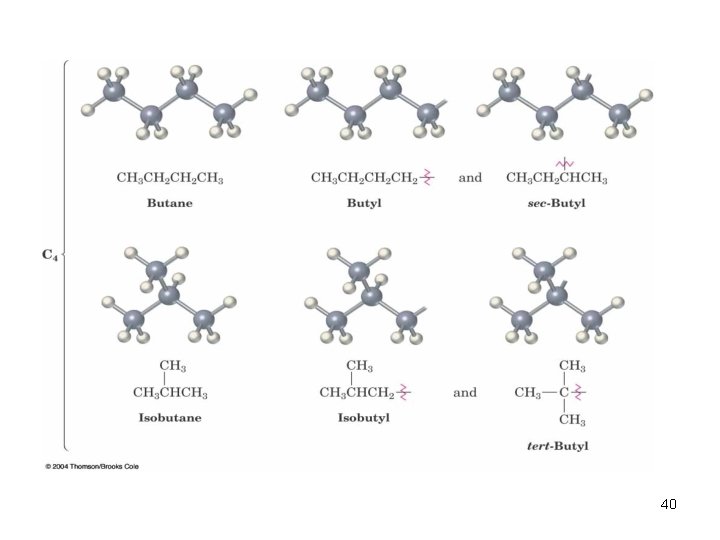
40
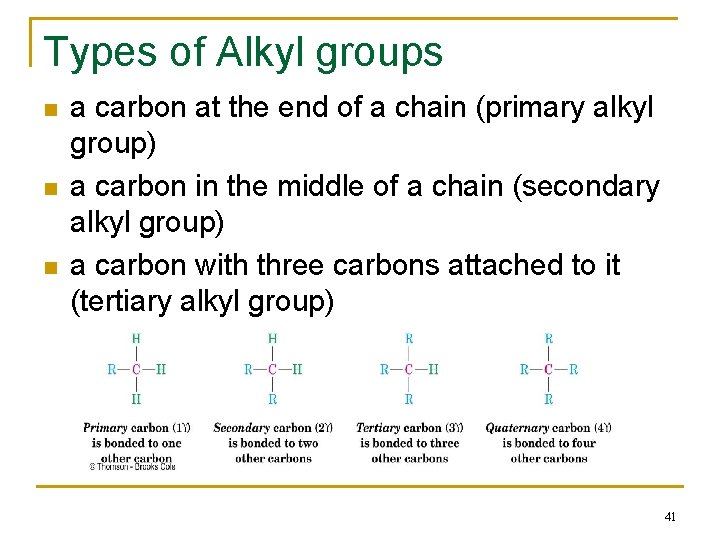
Types of Alkyl groups n n n a carbon at the end of a chain (primary alkyl group) a carbon in the middle of a chain (secondary alkyl group) a carbon with three carbons attached to it (tertiary alkyl group) 41
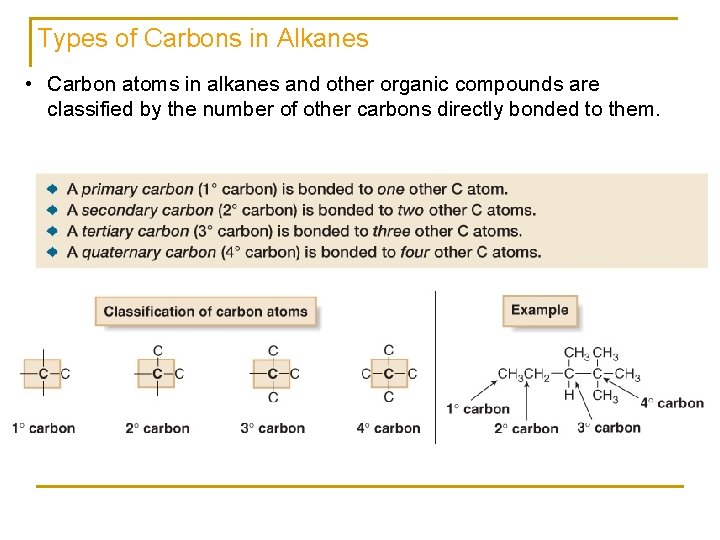
Types of Carbons in Alkanes • Carbon atoms in alkanes and other organic compounds are classified by the number of other carbons directly bonded to them.

Types of Hydrogens in Alkanes • Hydrogen atoms are classified as primary (10), secondary (20), or tertiary (30) depending on the type of carbon atom to which they are bonded.
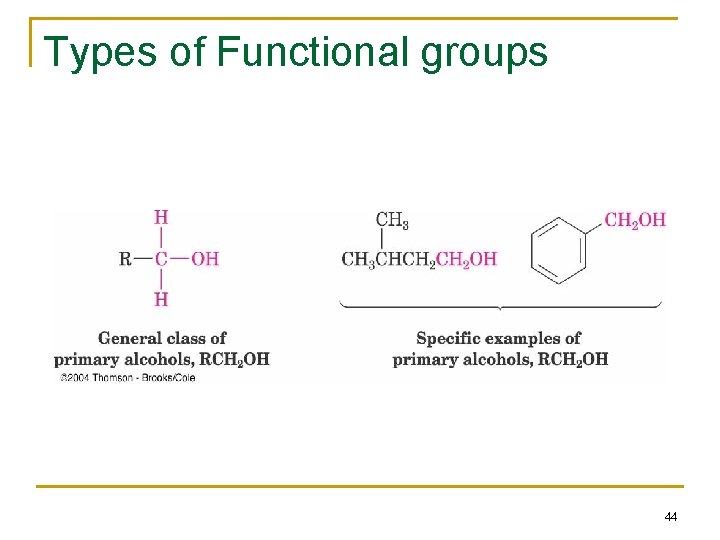
Types of Functional groups 44
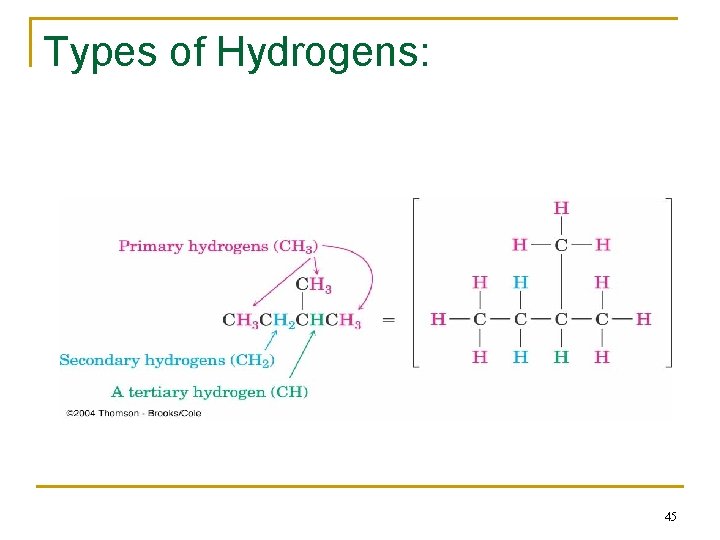
Types of Hydrogens: 45

2. 4 Naming Alkanes: IUPAC n Compounds are given systematic names by a process that uses: 46

Naming Branched-Chain Alkanes 1. Find the Parent: longest continuous carbon chain 2. Number the atoms in the chain n Number the chain so that the substituent gets the lowest possible number. 3. Identify & number the substituents n If two or more substituents are the same, the prefixes “di, ” “tri, ” and “tetra” are used to indicate how many identical substituents the compound has. n Substituents are listed in alphabetical order. 4. Write the name (A number and a word are separated by a hyphen; numbers are separated by a comma) 47
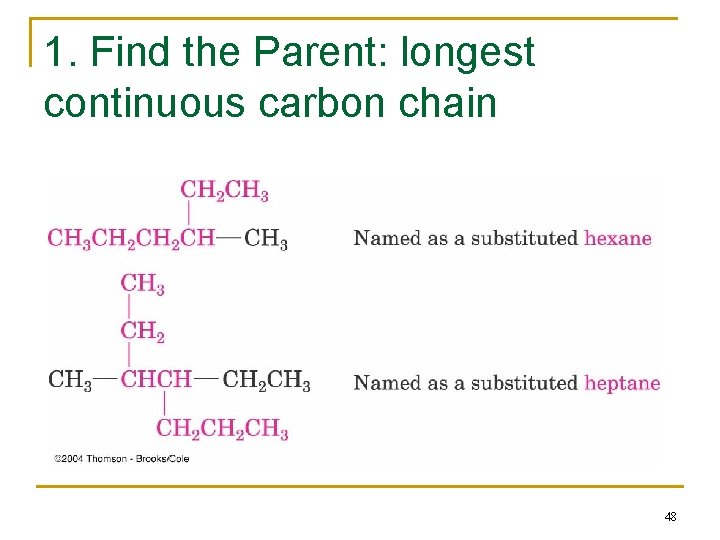
1. Find the Parent: longest continuous carbon chain 48

2. Number the atoms in the chain • number from the end closest to substituent 49
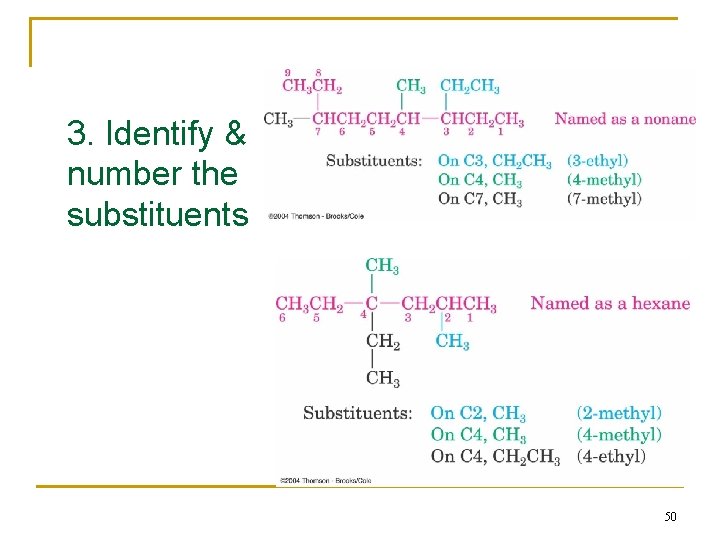
3. Identify & number the substituents 50
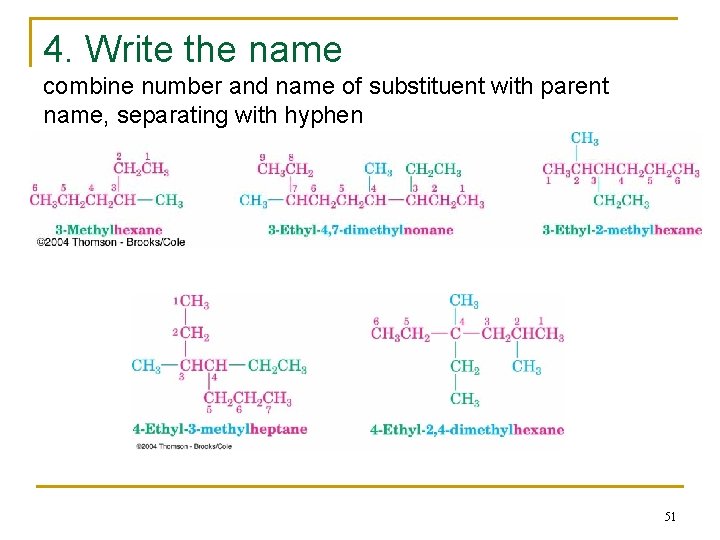
4. Write the name combine number and name of substituent with parent name, separating with hyphen 51

Additional points to keep in mind. . . n n di, tri, tetra, sec, and tert are ignored in alphabetizing. iso, neo, and cyclo are not ignored in alphabetizing. 52
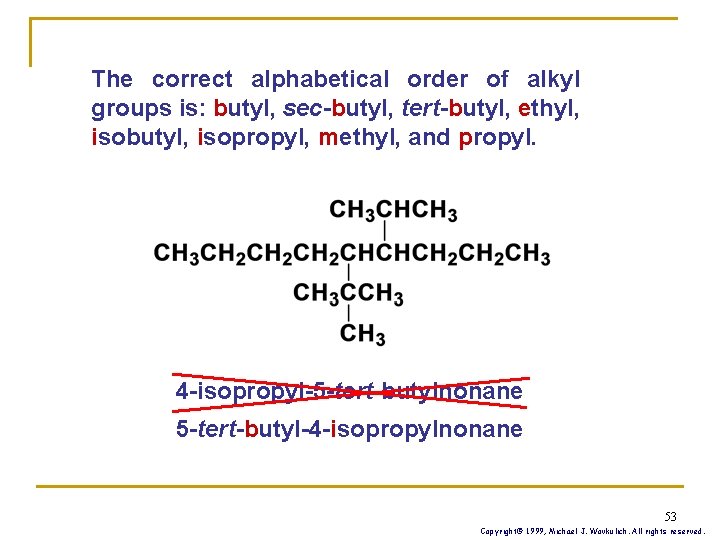
The correct alphabetical order of alkyl groups is: butyl, sec-butyl, tert-butyl, ethyl, isobutyl, isopropyl, methyl, and propyl. 4 -isopropyl-5 -tert-butylnonane 5 -tert-butyl-4 -isopropylnonane 53 Copyright© 1999, Michael J. Wovkulich. All rights reserved.

Ignore the prefixes di-, tri-, etc. when arranging alkyl group names alphabetically. 2, 3 -dimethyl-4 -ethylheptane 4 -ethyl-2, 3 -dimethylheptane 54 Copyright© 1999, Michael J. Wovkulich. All rights reserved.

Additional points to keep in mind. . . n When both directions lead to the same lowest number for one of the substituents, the direction is chosen that gives the lowest possible number to one of the remaining substituents. 55
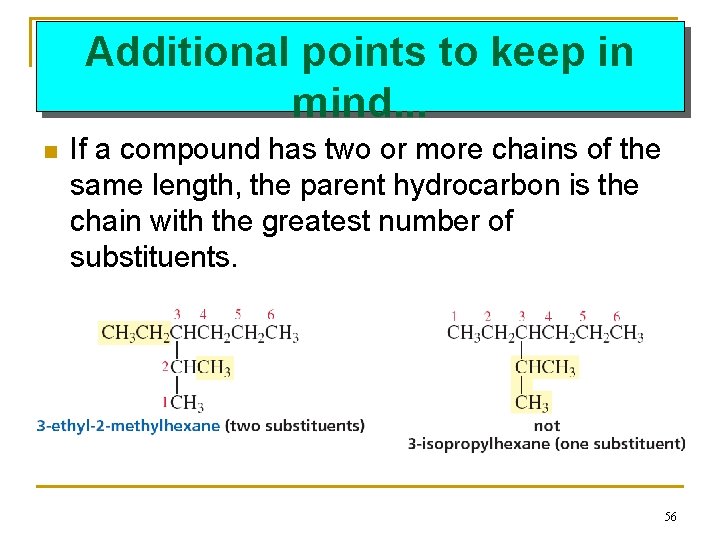
Additional points to keep in mind. . . n If a compound has two or more chains of the same length, the parent hydrocarbon is the chain with the greatest number of substituents. 56
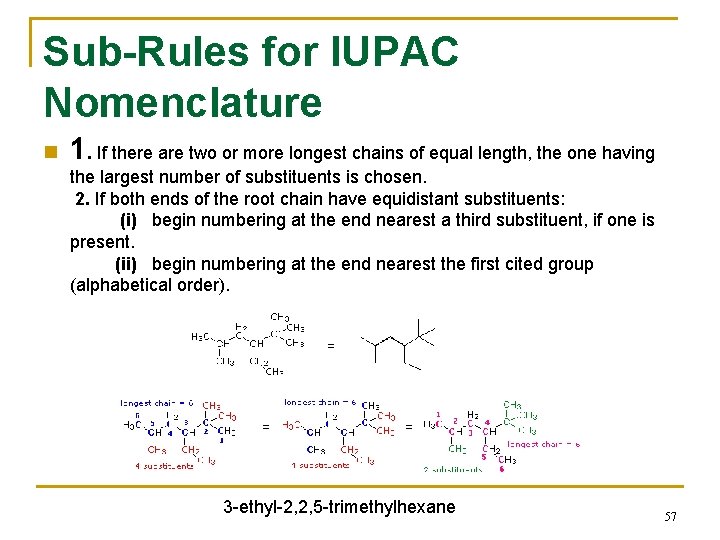
Sub-Rules for IUPAC Nomenclature n 1. If there are two or more longest chains of equal length, the one having the largest number of substituents is chosen. 2. If both ends of the root chain have equidistant substituents: (i) begin numbering at the end nearest a third substituent, if one is present. (ii) begin numbering at the end nearest the first cited group (alphabetical order). 3 -ethyl-2, 2, 5 -trimethylhexane 57
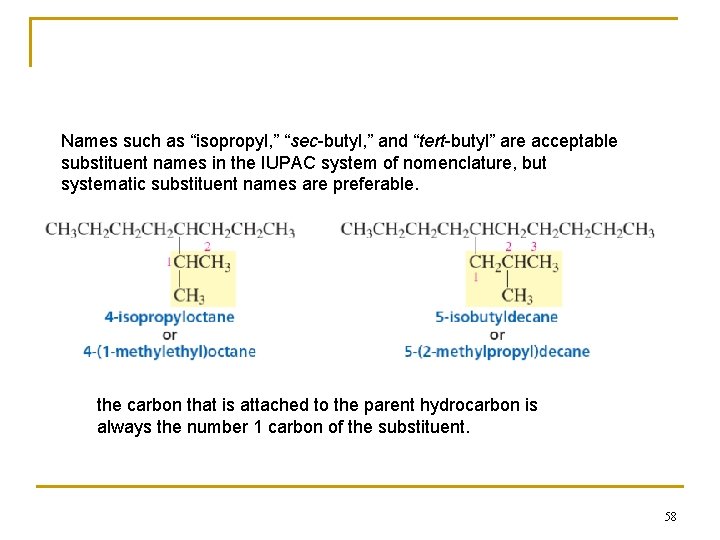
Names such as “isopropyl, ” “sec-butyl, ” and “tert-butyl” are acceptable substituent names in the IUPAC system of nomenclature, but systematic substituent names are preferable. the carbon that is attached to the parent hydrocarbon is always the number 1 carbon of the substituent. 58

Some substituents have only a systematic name. 59
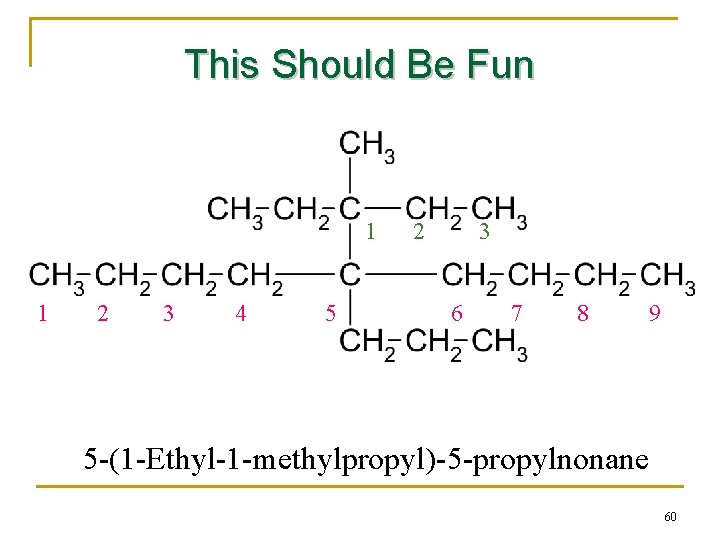
This Should Be Fun 1 1 2 3 4 5 2 3 6 7 8 9 5 -(1 -Ethyl-1 -methylpropyl)-5 -propylnonane 60
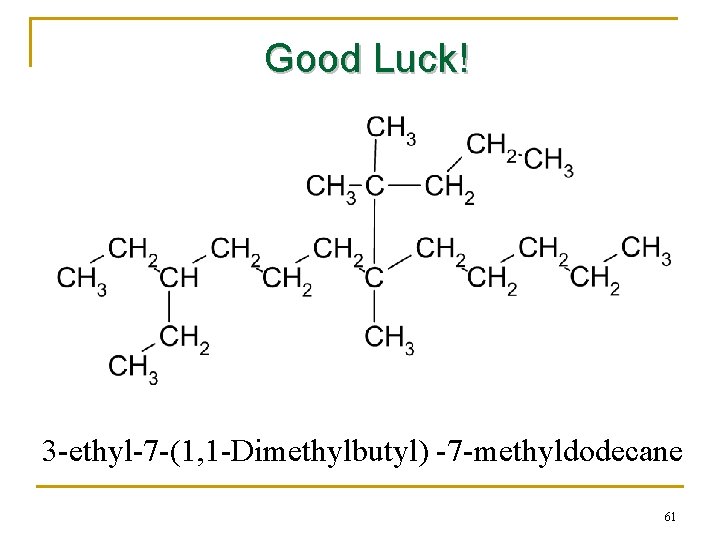
Good Luck! 3 -ethyl-7 -(1, 1 -Dimethylbutyl) -7 -methyldodecane 61
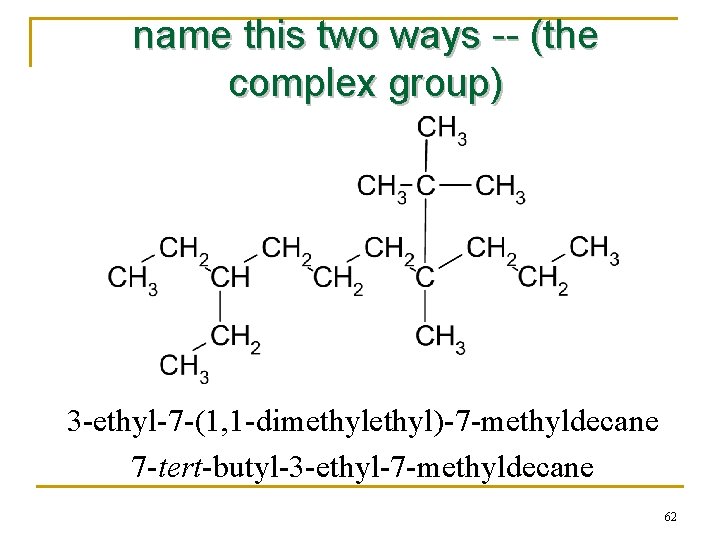
name this two ways -- (the complex group) 3 -ethyl-7 -(1, 1 -dimethyl)-7 -methyldecane 7 -tert-butyl-3 -ethyl-7 -methyldecane 62
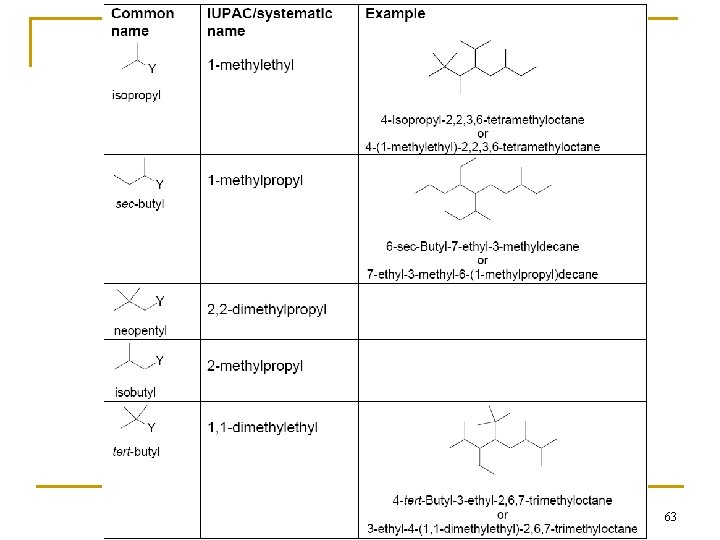
63

2. 5 Properties of Alkanes n n Called paraffins (low affinity compounds) because they are relatively unreactive They will burn in a flame, producing carbon dioxide, water, and heat CH 4 + 2 O 2 → CO 2 + 2 H 2 O + heat ΔH° = – 803 k. J mol– 1 64

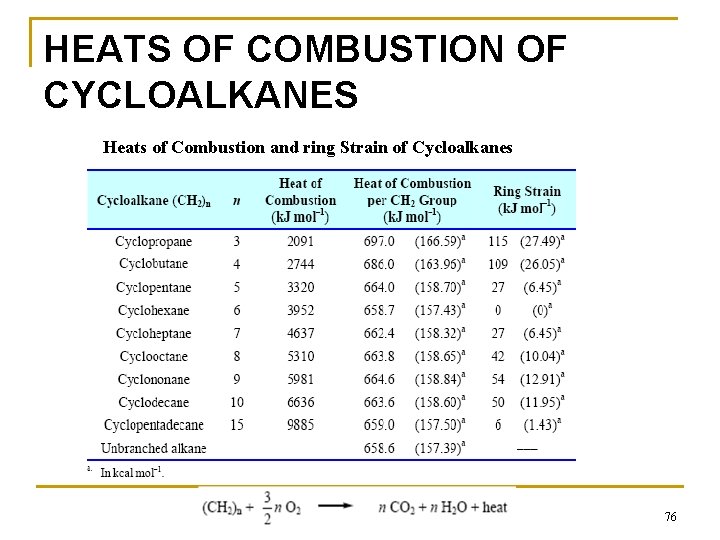
HEATS OF COMBUSTION Heat of combustion: the enthalpy change for the complete oxidation of a compound n ⇒ for a hydrocarbon means converting it to CO 2 and water. 1) For methane, the heat of combustion is – 803 k. J mol– 1 (– 191. 9 kcal mol– 1): n CH 4 + 2 O 2 → CO 2 + 2 H 2 O + heat 2) Heat of combustion can be used to measure relative stability of isomers. 65
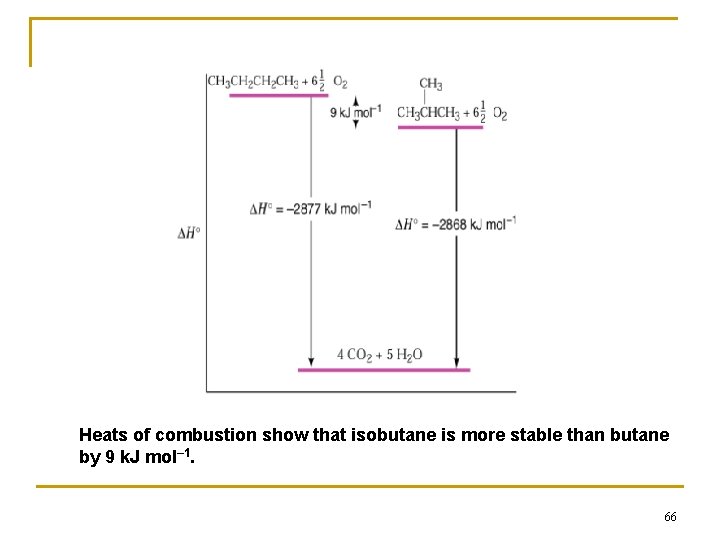
Heats of combustion show that isobutane is more stable than butane by 9 k. J mol– 1. 66
67

2. 5 Properties of Alkanes n Solubility of Alkanes q n Densities of Alkanes q n alkanes are nonpolar and are hydrophobic “water hating” because they do not dissolve in water. alkanes have densities around 0. 7 g/m. L Therefore a mixture of an alkane (such as gasoline or oil) and water quickly separates into two phases, with the alkane on top. 68

2. 5 Properties of Alkanes n They react with Cl 2 in the presence of light to replace H’s with Cl’s (not easily controlled) 69
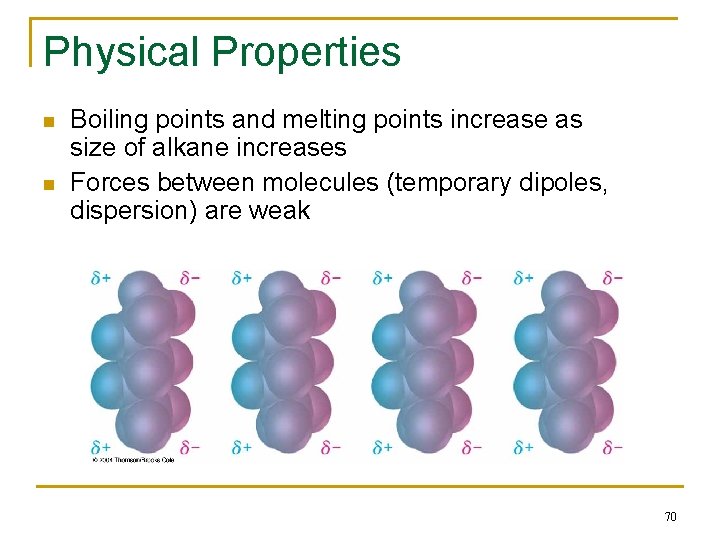
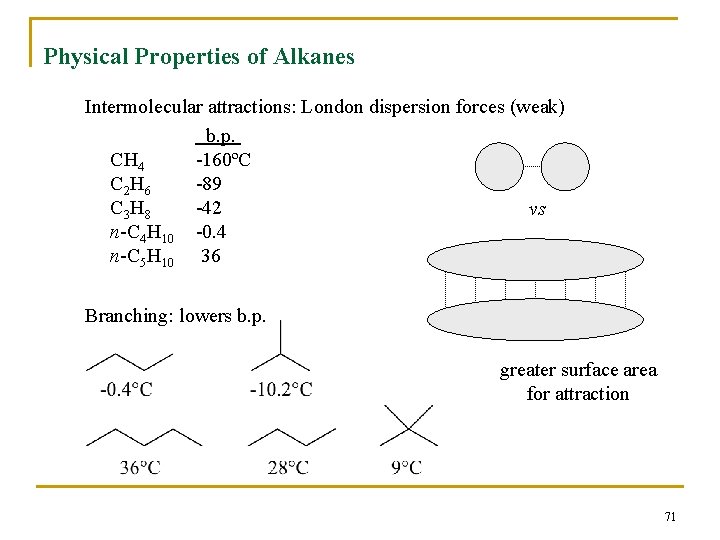
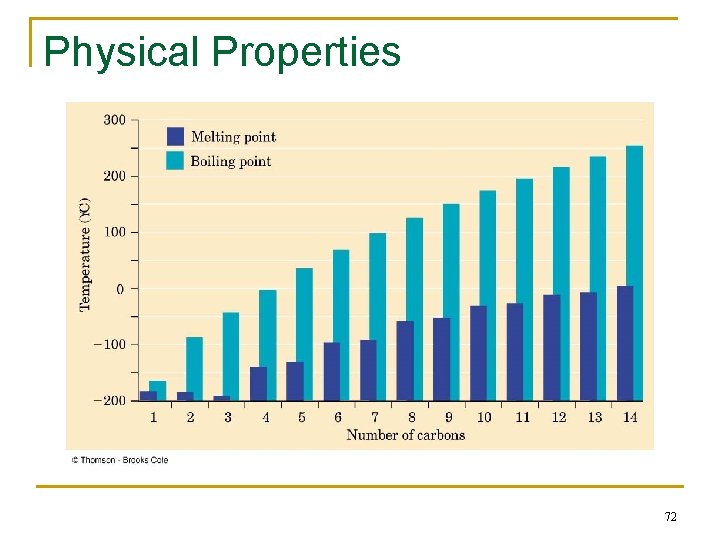
Physical Properties n n Boiling points and melting points increase as size of alkane increases Forces between molecules (temporary dipoles, dispersion) are weak 70
Physical Properties of Alkanes Intermolecular attractions: London dispersion forces (weak) b. p. CH 4 -160ºC C 2 H 6 -89 C 3 H 8 -42 vs n-C 4 H 10 -0. 4 n-C 5 H 10 36 Branching: lowers b. p. greater surface area for attraction 71
Physical Properties 72

2. 6 Cycloalkanes n n n Cycloalkanes are alkanes that have carbon atoms that form a ring (called alicyclic compounds) Simple cycloalkanes are rings of CH 2 units, (CH 2)n, or Cn. H 2 n Structure is shown as a regular polygon with the number of vertices equal to the number of C’s (a projection of the actual structure) 73
2. 6 Cycloalkanes 74
22_499 C No "head-on" overlap of atomic orbitals 109. 5° 60° C (a) C (b) 75
HEATS OF COMBUSTION OF CYCLOALKANES Heats of Combustion and ring Strain of Cycloalkanes 76

Complex Cycloalkanes n Naturally occurring materials contain cycloalkane structures n Examples: q chrysanthemic acid (cyclopropane), q prostaglandins (cyclopentane), q steroids (cyclohexanes and cyclopentane) 77
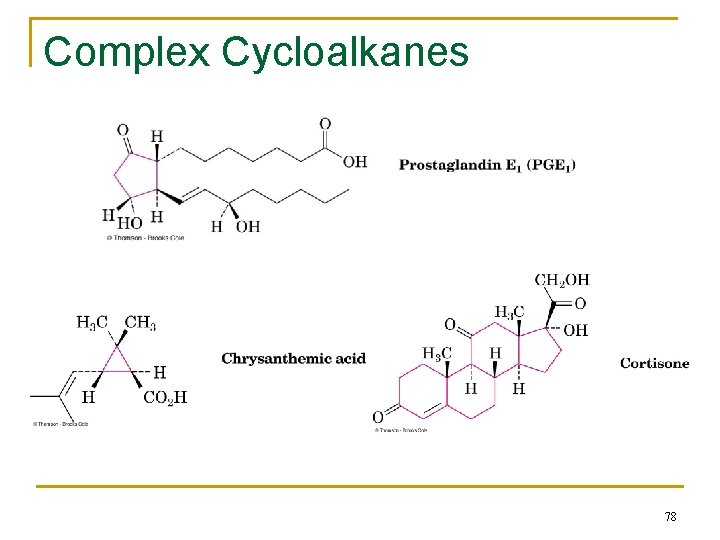
Complex Cycloalkanes 78
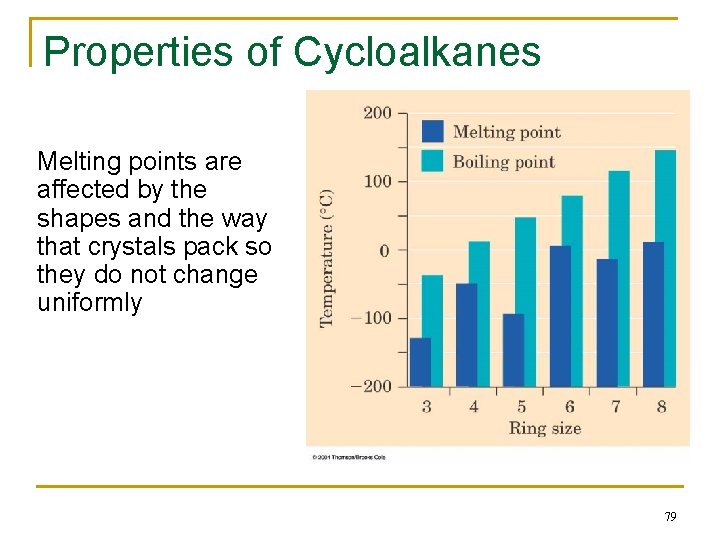
Properties of Cycloalkanes Melting points are affected by the shapes and the way that crystals pack so they do not change uniformly 79

1. Cycloalkanes with only one ring: n n n 1) Substituted cycloalkanes: alkylcycloalkanes, halocycloalkanes, alkylcycloalkanols 2) Number the ring beginning with the substituent first in the alphabet, and number in the direction that gives the next substituent the lower number possible. 3) When three or more substituents are present, begin at the substituent that leads to the lowest set of locants. 80
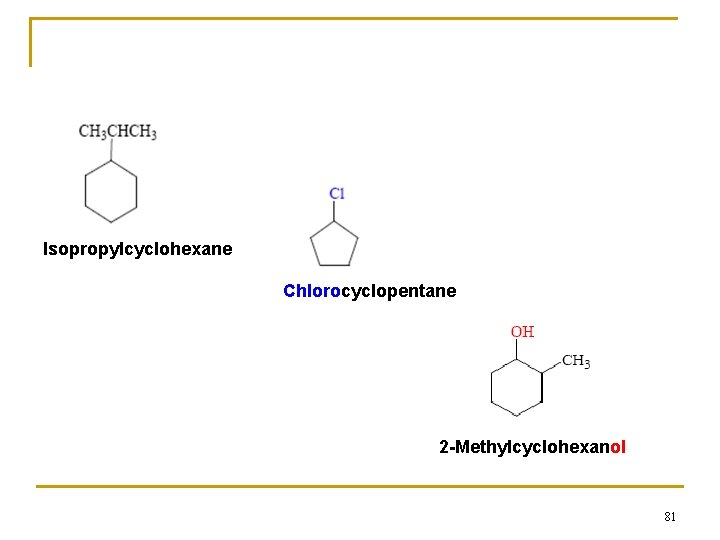
Isopropylcyclohexane Chlorocyclopentane 2 -Methylcyclohexanol 81
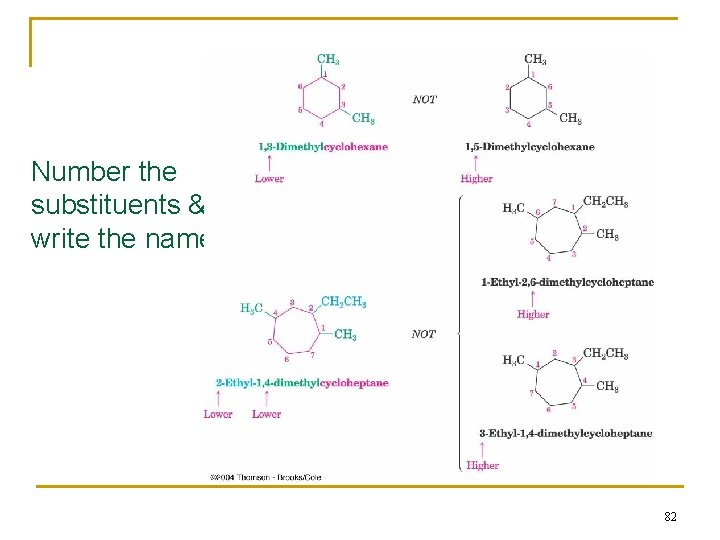
Number the substituents & write the name: 82
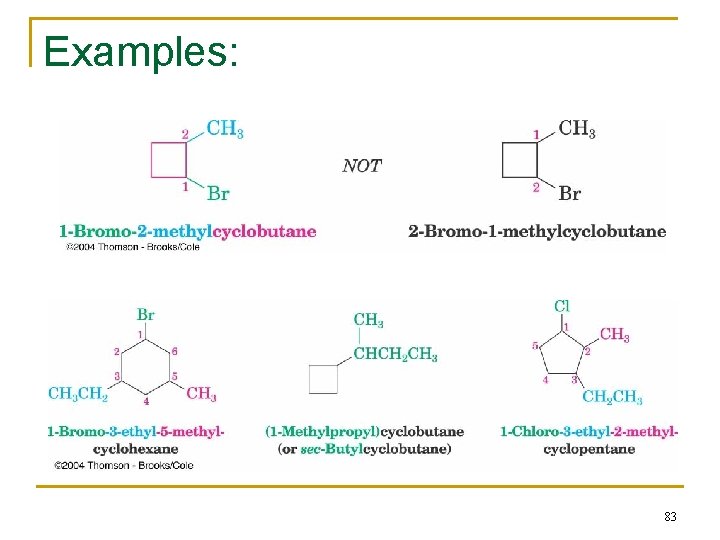
Examples: 83

2. 7 Naming Cycloalkanes n Count the number of carbon atoms in the ring and the number in the largest substituent chain. If the number of carbon atoms in the ring is equal to or greater than the number in the substituent, the compound is named as an alkyl-substituted cycloalkane 84
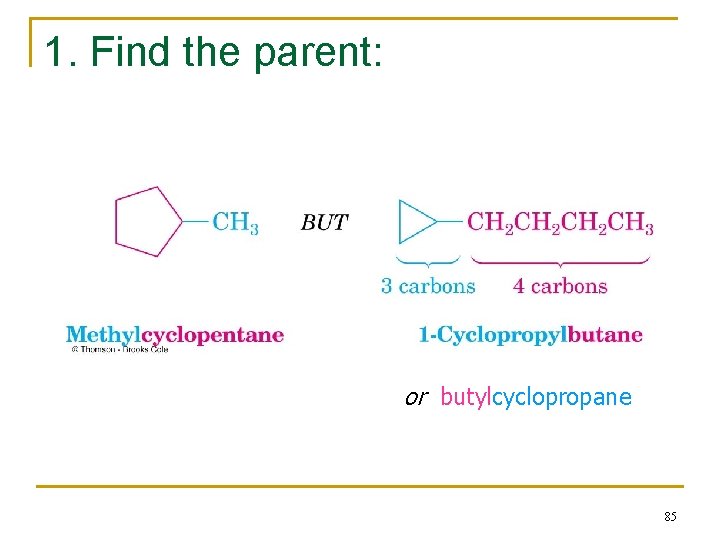
1. Find the parent: or butylcyclopropane 85
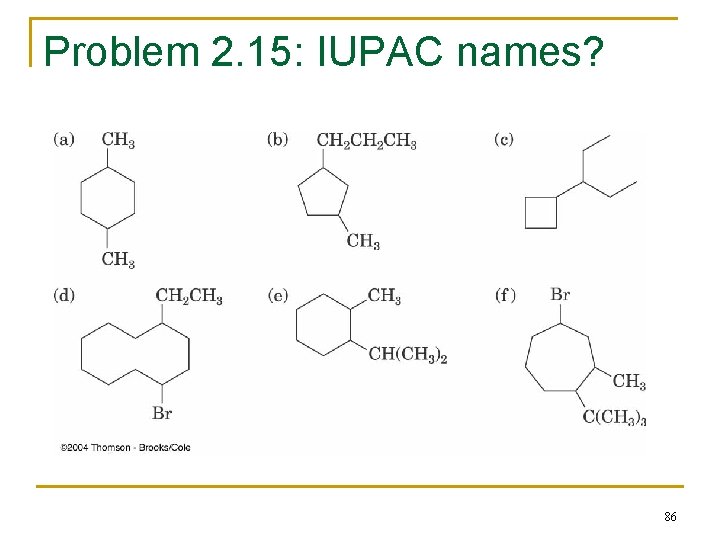
Problem 2. 15: IUPAC names? 86

Stereoisomers n Compounds with atoms connected in the same order but which differ in three-dimensional orientation, are stereoisomers. آﻨﻬﺎﺳﺖ (configuration) ﺑﻨﺪی پیکﺮ ﺩﺭ ﻓﻀﺎیی ﻫﺎی ﺍیﺰﻭﻣﺮ ﺗﻔﺎﻭﺕ n n n The terms “cis” and “trans” should be used to specify stereoisomeric ring structures. Recall that constitutional isomers have atoms connected in different order. 87

2. 8 Cis-Trans Isomerism in Cycloalkanes n n n Rotation about C-C bonds in cycloalkanes is limited by the ring structure Rings have two “faces” and substituents are labeled as to their relative facial positions There are two different 1, 2 -dimethyl-cyclopropane isomers, one with the two methyls on the same side (cis) of the ring and one with the methyls on opposite sides (trans) 88
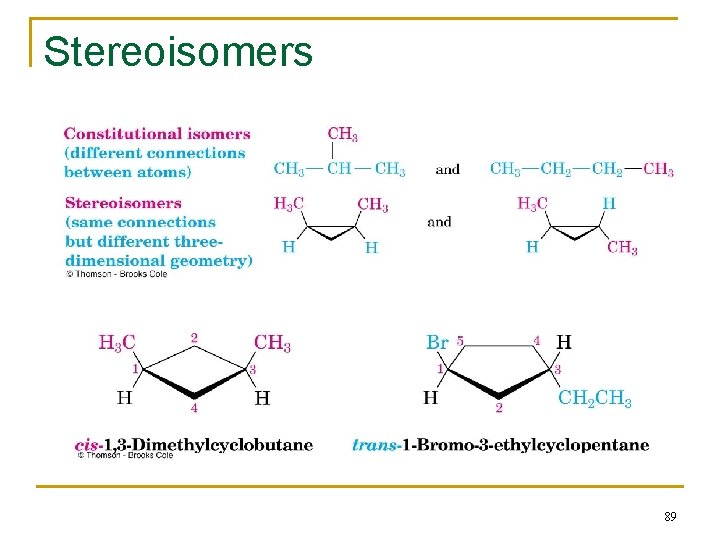
Stereoisomers 89
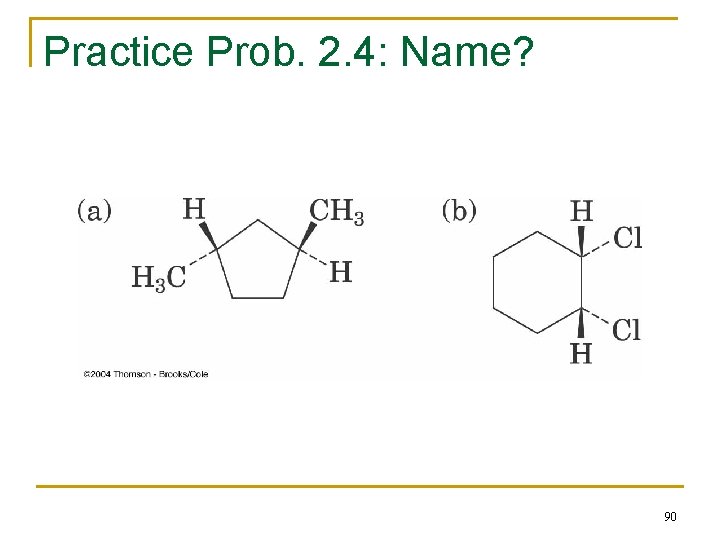
Practice Prob. 2. 4: Name? 90
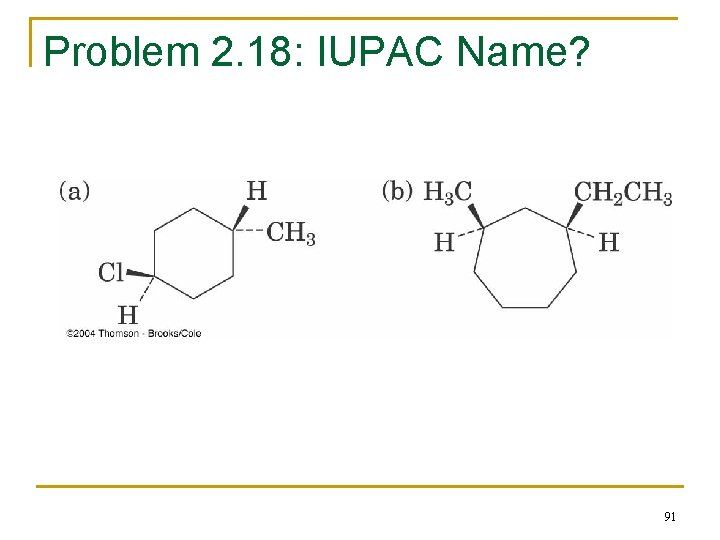
Problem 2. 18: IUPAC Name? 91


n Octane rating: the quality of the gasoline q percent 2, 2, 4 -trimethylpentane (isooctane) in a mixture of isooctane and heptane that has equivalent antiknock properties Typical "octane booster" gasoline additives include MTBE (methyl tertiary butyl ether), ETBE, isooctane and toluene. Lead in the form of tetra-ethyl lead was once a common additive, but since the 1970 s, its use in most of the industrialised world has been restricted, and its use is currently limited mostly to aviation gasoline. 93
- Slides: 93