Chapter 2 Alkaline Earth Metals Beryllium Table of

Chapter 2 Alkaline Earth Metals Beryllium Table of Contents Introduction 1. Occurrence Magnesium 2. Chemical Properties 3. Compounds Uses Strontium Radium Barium Calcium

Chapter 2 Alkaline Earth Metals Warm Up • Why were these elements called “alkaline earth metals”? • What do you think about the uses of these elements commonly?
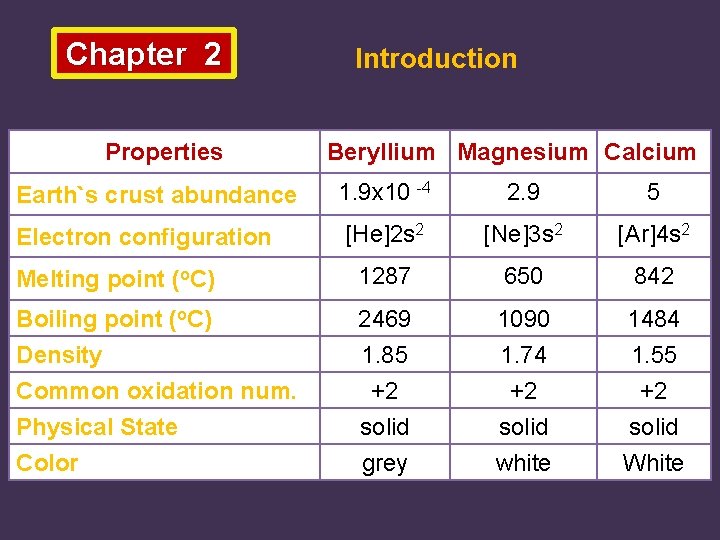
Chapter 2 Properties Introduction Beryllium Magnesium Calcium 1. 9 x 10 -4 2. 9 5 [He]2 s 2 [Ne]3 s 2 [Ar]4 s 2 Melting point (o. C) 1287 650 842 Boiling point (o. C) Density Common oxidation num. Physical State Color 2469 1. 85 +2 solid grey 1090 1. 74 +2 solid white 1484 1. 55 +2 solid White Earth`s crust abundance Electron configuration

Chapter 2 Introduction • The word “earth” was used for the oxides of group 2 A by the ancient scientists. • Alkaline earth metals, after the alkali metals, are secondary metals with strong metallic properties. • The group 2 A elements are less active than those of 1 A, but more active than those of group 3 A. • Except Be, all form ionic compounds. • Be forms mostly covalent compounds.

Chapter 2 Introduction • The atomic radius increases from top to bottom. • The melting point, boiling point and the hardness of the elements decrease top to bottom. • Beryllium is the hardest alkaline earth metal • Barium is the softest alkaline earth metal. • Compounds of CO 3 -2 and O-2 are insoluble in water. • Malleable and ductile. • Excess Mg+2 and Ca+2 in water means that it is hard.

Chapter 2 Introduction Characteristic Flame Colors of Alkaline Earth Metals Calcium Strontium Barium Magnesium

Chapter 2 1. Occurrence Minerals of Some Alkaline Earth Metals Beryllium Calcium Barium Beryl, Be 3 Al 2(Si. O 3)6 Limestone (marble), Ca. CO 3 Barytes, Ba. SO 4 Magnesium Gypsum, Ca. SO 4. 2 H 2 O Witherite, Ba. CO 3 Dolomite, Ca. CO 3. Mg. CO 3 Fluorite, Ca. F 2 Radium Carnallite, KCI. Mg. CI 2. 6 H 2 O Strontium All isotopes are radioactive Epsom salt, Mg. SO 4. 7 H 2 O Celestite, Sr. SO 4 Strontianite, Sr. CO 3

Chapter 2 2. Chemical Properties • They are Covered by their oxides when they are exposed to air. • Burn in air with bright flame. • Alkaline earth metals form MSO 4, MCO 3, MO, M(OH)2, compounds where M alkaline earth metal. • Activity of the metals increases from Be to Ba. • Reactions of Ca, Sr and Ba are easy with water. • Reactions of Be and Mg with water are difficult.

Chapter 2 2. Chemical Properties • Ca(s) + 2 H 2 O(l) → Ca+2(aq) + 2 OH-(aq) + H 2(g) • Mg(s) + 2 H 2 O(l) → Mg(OH)2(s) + H 2(g) • 2 Mg(s) + O 2(g) → 2 Mg. O(s) • Mg (s) + H 2(g) → Mg. H 2(s) • Ca (s) + 2 HCI(aq) → Ca. Cl 2 + H 2(g) • Be (s) + 2 HCI(aq) → Be. CI 2 + H 2(g) • All oxides and hydroxides of alkaline earth metals show basic properties. • Oxides and its hydroxides of beryllium show amphoteric properties.

Chapter 2 2. Chemical Properties

Chapter 2 2. Chemical Properties

Chapter 2 2. Chemical Properties

Chapter 2 2. Chemical Properties

Chapter 2 3. Compounds • Ionic • Have high melting and boiling points. • Thermally stable. • Less soluble in water than those of 1 A metals. • Solubility of salts increase from Be to Ba.

Chapter 2 3. Compounds Calcium Oxide, Ca. O • Obtained by heating calcium carbonate (limestone) at 900°C. • Ca. CO 3(s) → Ca. O(s) + CO 2(g) Calcium Carbonate, Ca. CO 3 • It forms by evolving CO 2 gas from Ca(OH)2 solution.

Chapter 2 3. Compounds Calcium Carbide, Ca. C 2 • Small gray solid pieces • Used widely in industry • Prepared by the reaction of calcium oxide (lime) and coke. Ca. O(s) + 3 C(s) ↔ Ca. C 2(s) + CO (g) Ca. C 2(s) + 2 H 2 O(l) → C 2 H 2(g) + Ca(OH)2(s)

Chapter 2 3. Compounds Calcium Sulfate, Ca. SO 4 • Ca. SO 4 : Gypsum Ca. SO 4. 2 H 2 O : Alabaster Ca. SO 4. 1/2 H 2 O : Plaster of Paris • Gypsum is a white substance and used for fabrication into wallboard.

Chapter 2 Uses Beryllium • Very light element • To prepare some special alloys. • 2% Be in Cu alloy in springs. • Making glass for X-ray tubes. • In the aerospace industry.

Chapter 2 Uses Magnesium • Light metal • Production of planes, missiles • Some light household items. • Flash in photography

Chapter 2 Uses

Chapter 2 Uses Magnesium • To increase the hardness, durability and resistance to corrosion of alloys. • Additive in rocket fuels and signal rockets. • Mg(OH)2 is used as a stomach antacid.

Chapter 2 Uses Calcium Compounds • Construction materials, bleaching agents. • Ca. O in the production of some chemicals, steel, glass, paper and sugar. • Ca. C 2 is used to produce acetylene gas for welding. • In fertilizers and toothpaste

Chapter 2 Uses Barium and Its Compounds • Ba is used as a gas absorbent in vacuum tubes. • The alloys of Ba with Ni donate electrons so used in vacuum tubes and in ignitors and spark plugs. • Barium nitrate and barium chlorate are used to obtain green light in fireworks.

Chapter 2 Uses
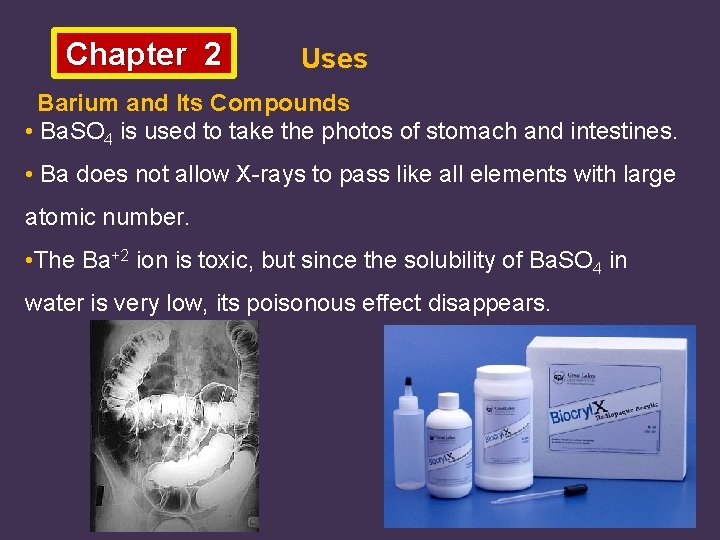
Chapter 2 Uses Barium and Its Compounds • Ba. SO 4 is used to take the photos of stomach and intestines. • Ba does not allow X-rays to pass like all elements with large atomic number. • The Ba+2 ion is toxic, but since the solubility of Ba. SO 4 in water is very low, its poisonous effect disappears.

Chapter 2 Uses Strontium • The salts of strontium in signal rockets and fireworks to produce red light. • Nuclear batteries.
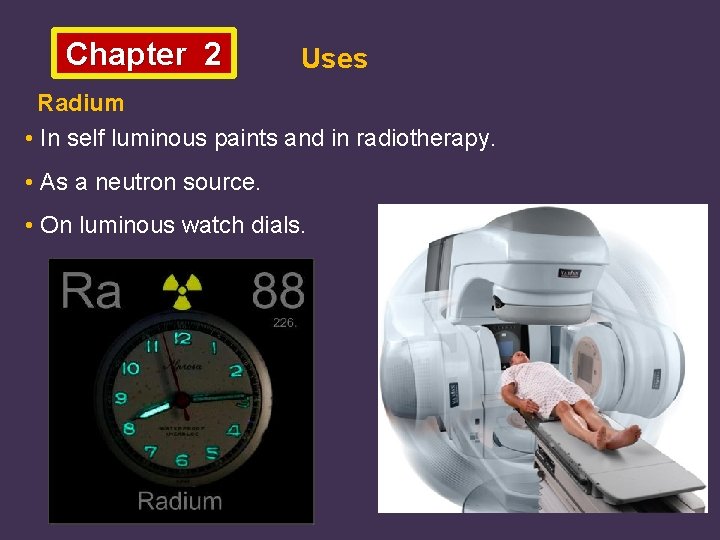
Chapter 2 Uses Radium • In self luminous paints and in radiotherapy. • As a neutron source. • On luminous watch dials.

Chapter 2 Chapter Review 1. Match the formulas of calcium compounds and their technical names. Names Lime water Chalk Alabaster Lime Marble Quicklime Gypsum Calcium carbide Limestone Formula Ca. O Ca(OH)2 Ca. CO 3 Ca. SO 4. 1/2 H 2 O Ca. SO 4. 2 H 2 O

Chapter 2 Chapter Review 2. Choose from the following: Na. OH, HNO 3, Fe. O, H 2 O, SO 3, Cu. Cl 2 substances that can react with A. Calcium oxide B. Barium oxide

Chapter 2 Chapter Review 3. Choose from the following: H 2 O, Cl 2, S, Mg. O, CO 2, H 3 PO 4, Cu(OH)2, Fe(NO 3)2, Na. Cl substances that can react with solutions of: A. Potassium hydride B. Barium hydroxide Write down the equations in the molecular and ionic form.

Chapter 2 Chapter Review 4. Complete the equations, and name the products: A. Ca + O 2 B. Ca + H 2 O C. Ca + HCl D. Ca + KOH E. Ca. O + CO 2 F. Ca. O + HNO 3 G. Ca(OH)2 + Cu(NO 3)2 H. Ca. O + P 2 O 5

Chapter 2 Chapter Review 5. Write down the possible reaction equations in the molecular and ionic form: A. Ca. CO 3 + HNO 3 B. Ca. CO 3 + Na. OH C. Ca(NO 3)2 + Ba. Cl 2 D. Ca. Cl 2 + Ba 3(PO 4)2 E. Mg. O + HCl F. Mg(OH)2 + HNO 3 G. Mg(OH)2 + Ba. Cl 2 H. Mg. CO 3 + HCl

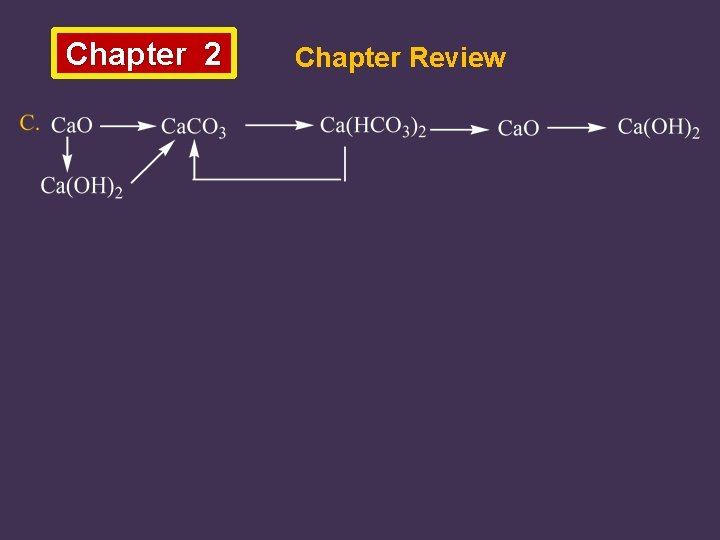
Chapter 2 Chapter Review 6. Write down the equations for the following transformations:

Chapter 2 Chapter Review B. Ca Ca. Cl 2 Ca. CO 3 Ca. O Ca(OH)2 Ca(Cl. O)2
Chapter 2 Chapter Review

Chapter 2 Chapter Review D. Mg. CO 3 Mg. O Mg. Cl 2 Mg(OH)2 Mg. SO 4

Chapter 2 Chapter Review 7. Which water is called "hard"? How many types of hardness do you know? Explain what "carbonated" hardness means. How can it be removed? Write down the equations of carbonated hardness removal.

Chapter 2 Chapter Review 8. Gypsum (Ca. SO 4. 2 H 2 O) is formed by addition of water to alabaster (Ca. SO 4. 1/2 H 2 O). Calculate the mass of water necessary to transform alabaster into 43. 5 kg gypsum.

Chapter 2 Chapter Review 9. A solution containing 15. 9 g of sodium carbonate was added to a solution containing 11. 1 g of calcium chloride. Determine the mass of oxide formed.

Chapter 2 Chapter Review 10. 4. 8 g magnesium was dissolved in an excess hydrochloric acid. Calculate: A. Volume of the formed gas. B. Mass of accumulated salt.

Chapter 2 Chapter Review 11. Calculate the volume of carbon dioxide and mass of lime that can be obtained from chalk with a mass of 500 kg, if it contains 10% impurities.

Chapter 2 Chapter Review 12. Calculate the mass of carbon dioxide that can be absorbed by calcium hydroxide with a mass of 50 g and it contains 8% impurities.

End of the chapter 2
- Slides: 43