Chapter 2 2 2 Properties of Water Polar









- Slides: 10

Chapter 2 2. 2 – Properties of Water

Polar covalent • In the water molecule, the electrons of the polar covalent bonds spend more time near the oxygen than the hydrogen • The water molecule is thus a polar molecule: the overall charge is unevenly distributed • Polarity allows water molecules to form hydrogen bonds with each other

2. 2 – Properties of Water • Water (H 2 O) is a polar molecule – Polar – Unequal distribution of charge • Hydrogen side is slightly + • Oxygen side is slightly – – Hydrogen Bonding – the force of attraction between a hydrogen with a slightly + charge to another atom with a slightly - charge • Water’s unequal charge allows it to form hydrogen bonds and gives water its unique properties

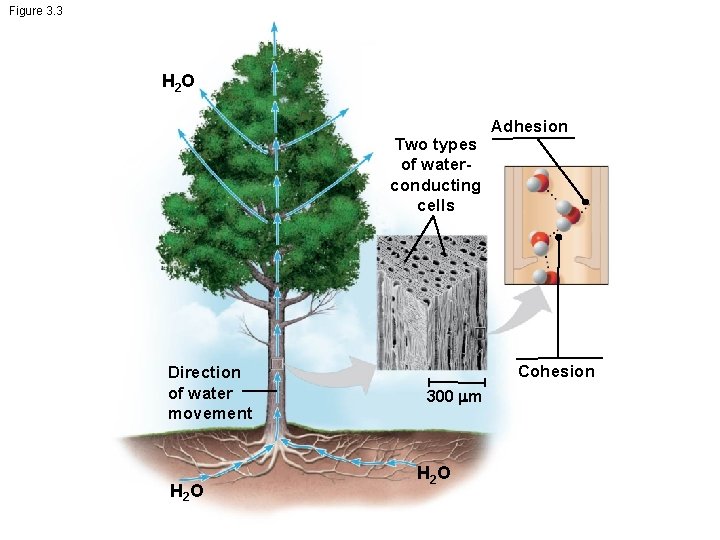
2. 2 – Properties of Water • Properties of Water – Cohesion – attraction between like molecules • Hydrogen bonds between water molecules produces surface tension • Biological membranes – Adhesion – attraction between unlike molecules • Hydrogen bonds between water and other atoms allows water to dissolve many different substances • Water = the solvent of life • Adhesion and cohesion of water lead to capillary action – Heat Capacity – ability to resist a change in temperature • Hydrogen bonding between water molecules requires more energy than normal to cause a change in water temperature

Figure 3. 3 H 2 O Two types of waterconducting cells Direction of water movement H 2 O Adhesion Cohesion 300 m H 2 O

Water: The Solvent of Life • A solution is a liquid that is a completely homogeneous mixture of substances • A solvent is the dissolving agent of a solution • The solute is the substance that is dissolved • An aqueous solution is one in which water is the solvent

Water: The Solvent of Life • Water is a versatile solvent due to its polarity • When an ionic compound is dissolved in water, each ion is surrounded by a sphere of water molecules called a hydration shell

Hydrophilic and Hydrophobic • A hydrophilic substance is one that has an affinity for water • A hydrophobic substance is one that does not have an affinity for water • Oil molecules are hydrophobic because they have relatively nonpolar bonds • Hydrophobic molecules related to oils are the major ingredients of cell membranes

2. 2 – Properties of Water • Acids, Bases and p. H – In some cases, certain substances dissolve in water and give off reactive ions • Acid – a substance, that when dissolved in water, gives off hydrogen ions (H+) • Base – a substance, that when dissolved in water, gives off hydroxide ions (OH-)

2. 2 – Properties of Water • Acids, Bases and p. H (cont. ) – The strength of an acid or base depends on how much H+ or OH- is given off – Measured using the p. H scale • p. H 7 = neutral • p. H from 0 -6. 9 = acidic • p. H from 7. 1 -14 = basic (alkaline) – Buffer maintain homeostasis in organisms. • weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in p. H.