Chapter 19 Porphyrins and Hemoglobin By Louann W

Chapter 19: Porphyrins and Hemoglobin By Louann W. Lawrence, Larry A. Broussard Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Introduction • Porphyrins, hemoglobin, & myoglobin all contain porphyrin ring: 4 pyrrole groups bonded by methene bridges. • Porphyrins can chelate metals to form functional groups that participate in oxidative metabolism. • Hemoglobin designed to bind, deliver, and release oxygen. • Porphyrias: disturbances heme synthesis • Hemoglobinopathies: disorders of qualitative defects in hemoglobin molecule (example: Sickle Cell Anemia) • Thalassemias: disorders of quantitative defects in production of normal hemoglobin molecules (examples: alpha and beta thalassemia) Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins
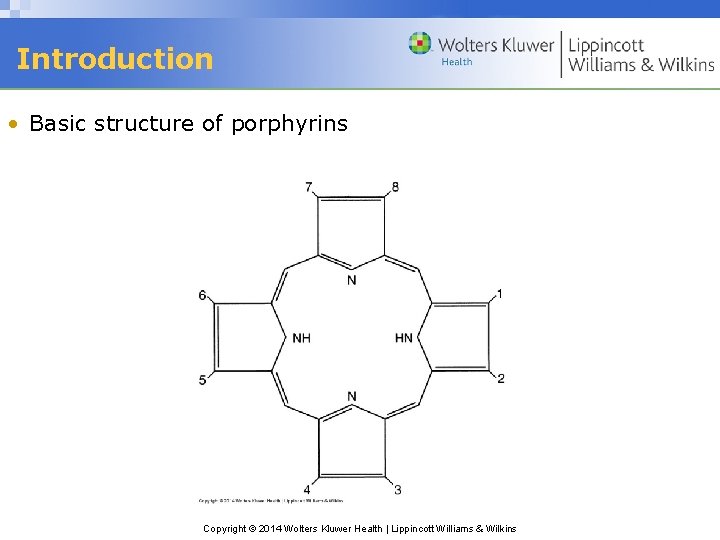
Introduction • Basic structure of porphyrins Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins
Introduction Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Porphyrins (in the body): – Act as chemical intermediates in synthesis of hemoglobin, myoglobin, & other respiratory pigments called cytochromes – Part of peroxidase & catalase enzymes, which contribute to efficiency of internal respiration – Chelate iron to form heme – Used to diagnose porphyrias (result from heme disturbances) Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Heme Synthesis �Main synthesis sites are the bone marrow and liver �Synthesis of heme: 1. Delta-aminolevulinic acid 2. Porphobilinogen 3. Uroporphyrinogen 4. Coproporphyrinogen 5. Protoporphyrin 6. Heme Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Chemistry of Porphyrins – Porphyrins are organic compounds found in nature. • Pigment Chlorophyl is a magnesium porphyrin – Four basic isomers may exist for every porphyrin compound (I-IV) however, only types I & III, which differ in side chain arrangement, occur in nature. – Only Type III isomers form heme – Porphyrins are stable & red-violet to red-brown and fluoresce red when excited by light near 400 nm. – Porphyrinogens: reduced form of of porphyrins, functional forms that must be used in heme synthesis; unstable, colorless, do not fluoresce Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • 3 porphyrin compounds clinically significant in humans: – Protoporphyrin (PROTO) – primarily excreted in feces – Uroporphyrin (URO) – primarily excreted in urine – Coprophorphyrin (COPRO) – can be both • Excess of the 3 in biological fluids is a sign of abnormal heme synthesis. • Again – porphyrinogens are the functional form of the compound that MUST be used in heme synthesis. Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Porphyrin Synthesis – All cells contain hemoproteins & can synthesize heme, but bone marrow & liver are main sites. – Rate of heme synthesis in cells of liver is achieved largely through regulation of enzyme -aminolevulinic acid (ALA) synthase. • Increase in heme causes decrease in ALA synthase. • Decrease in heme causes increase in ALA synthase. – Rate of heme synthesis is flexible & can change rapidly in response to many external stimuli. Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Clinical Significance and Disease Correlation – Porphyrias: inherited or acquired enzyme deficiencies that result in overproduction of heme precursors in bone marrow or liver – Diagnosis is made by combination of history & physical & laboratory findings. – Types associated with neuropsychiatric symptoms (these 3 conditions are associated with excess of early precursors (ALA, phorphobilinogen or both) • ALA dehydratase (ALAD) • Deficiency porphyria (ADP) • Acute intermittent porphyria (AIP) Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Clinical Significance and Disease Correlation – Types associated with cutaneous symptoms (photosensitivity, blisters, excess facial hair, and hyperpigmentation). The following are due to excess later intermediates: • Porphyria cutanea tarda (PCT) exposed skin **increased fragility to light- • Hepatoerythropoietic porphyria (HEP) • Erythropoietic porphyria (EP) • Congenital erythropoietic porphyria (CEP) – Types associated with neurocutaneous symptoms – due to excess in both early and later intermediates. • Hereditary coproporphyria (HCP) • Variegate porphyria (VP) Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins
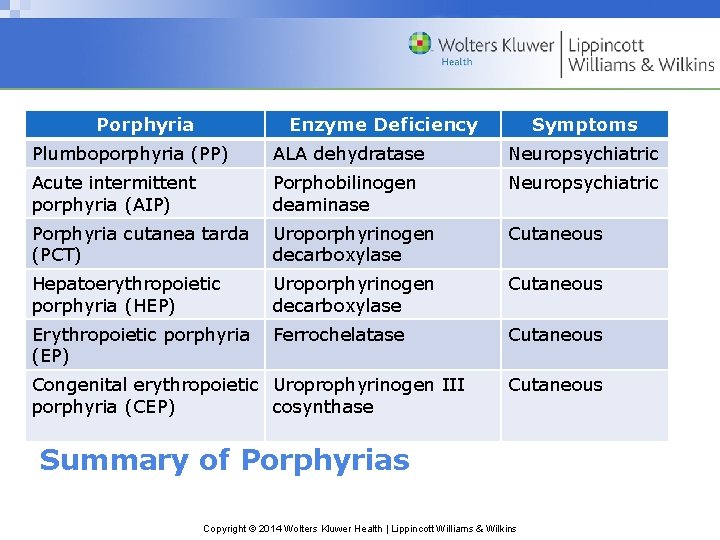
Porphyria Enzyme Deficiency Symptoms Plumboporphyria (PP) ALA dehydratase Neuropsychiatric Acute intermittent porphyria (AIP) Porphobilinogen deaminase Neuropsychiatric Porphyria cutanea tarda (PCT) Uroporphyrinogen decarboxylase Cutaneous Hepatoerythropoietic porphyria (HEP) Uroporphyrinogen decarboxylase Cutaneous Erythropoietic porphyria (EP) Ferrochelatase Cutaneous Congenital erythropoietic Uroprophyrinogen III porphyria (CEP) cosynthase Cutaneous Summary of Porphyrias Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Porphyrins • Methods of Analyzing Porphyrins – Tests for urinary PBG (porphobilinogen) & ALA • Watson-Schwartz • Hoesch sometimes used to confirm results of Watson-Schwarts due to Hoesch not having interference with uronbilinogen • Both of above use Ehrlich’s reagent. Ehrlich’s – form a red-orange color. When PBGs mixed with – Tests for porphyrins • Enhanced fluorescence of compounds in acidic solution • Chromatic separation & quantitation with spectrophotometry or fluorometry • Molecular diagnostic techniques Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

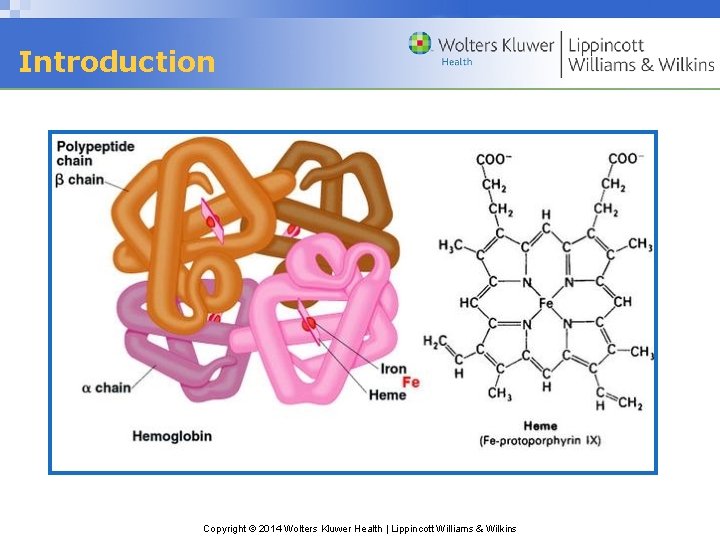
Hemoglobin • Role in Body: transports oxygen to tissue & CO 2 to lungs • Structure – A large, spherical, complex protein molecule (molecular weight is 64, 000) – Comprises heme (3%) & globin proteins (97%) – Contains 4 heme groups attached to 4 globin chains – Each globin chain consists of 141 or more amino acids & has 4 fold structure. – Majority of hemoglobin in normal adults is hemoglobin A (or A 1). – Hemoglobinopathies: diseases related to defects in hemoglobin structure Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins
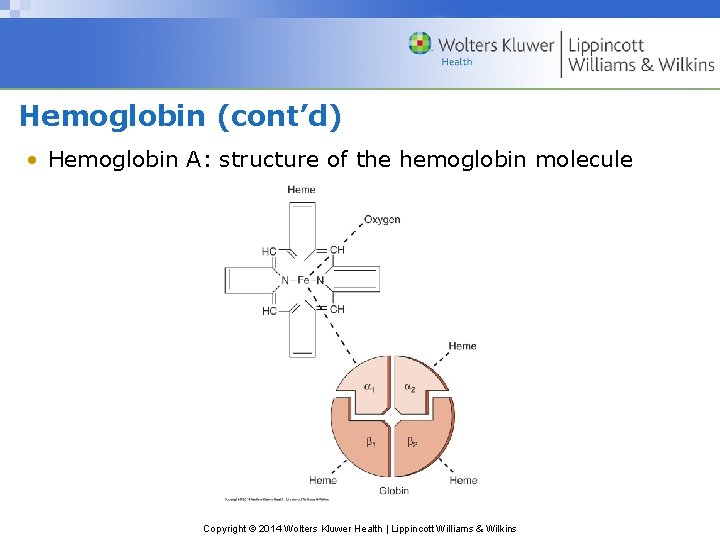
Hemoglobin (cont’d) • Hemoglobin A: structure of the hemoglobin molecule Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Synthesis and Degradation of Hemoglobin – Synthesis occurs in immature RBCs in bone marrow: • 65% in nucleated cells • 35% in reticulocytes – Normal synthesis depends on adequate iron supply & heme & protein synthesis to form globin portion. – Two pathways degrade hemoglobin: • Extravascular (80– 90%): occurs outside circulatory system within phagocytic cells of spleen, liver, & bone marrow • Intravascular (10– 20%): released directly into bloodstream & dissociated into & dimers Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Clinical Significance and Disease Correlation – Hemoglobin Qualitative Defects: The Hemoglobinopathies • Hemoglobin S • Amino acid defect is at 6 th position on -chain – glutamic acid substituted by valine. • Most common hemoglobinopathy in U. S. • Hb. AS (heterozygous = trait) and Hb. SS (homozygous = disease) • Heterozygous state offers protection from parasites (plasmodium falciparum) • Hemoglobin C • Glutamic acid in 6 th position of -chain is replaced by lysine, resulting in net positive charge. • Found in West Africa (North Ghana) in 17– 28% of population & in 2– 3% of African Americans Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Clinical Significance and Disease Correlation – Hemoglobin Qualitative Defects: The Hemoglobinopathies • Hemoglobin SC • Most common mixed hemoglobinopathy • -genes code for -S chains & -C chains, leaving no normal chains to produce hemoglobin A. • Less severe than Hb. SS disease but still has similar clinical symptoms to Hb. SS disease. • Hemoglobin E • An amino acid substitution of lysine for glutamic acid in 26 th position of chain, resulting in net positive charge • Found in Asia in about 20 million people, 80% of whom live in Southeast Asia Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Clinical Significance and Disease Correlation – Hemoglobin Qualitative Defects: The Hemoglobinopathies • Hemoglobin D • Variant with electrophoretic mobility on cellulose acetate, like that of hemoglobin S but with negative solubility test – Hemoglobin Quantitative Defects: The Thalassemias • Thalassemias: group of diseases in which defect causes reduced synthesis of 1 or more of hemoglobin chains • -Thalassemias – defective production of alpha chains • -Thalassemias – defective production of beta chains Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin -Thalassemias 2 alpha genes located on each chromosome 16, yielding a total of 4 genes. – Hydrops fetalis – (4 gene deletion) total absence of alpha chain synthesis • Make hemoglobin Barts (four gamma chains) • Die before birth or upon birth – Hemoglobin H disease (3 gene deletion) • Hgb H = beta chain tetramers (due to excess beta chains and lack of alpha chains). – -Thalassemia minor (2 gene deletion) • Mild, microcytic, hypochromic anemia. – Silent carrier (1 gene deletion) – sufficient alpha chains for normal amounts of hemoglobin • Often discovered by expression of 1 to 2% Hgb Barts Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin -Thalassemias (point mutations vs. gene deletions) • Many possible expressions of this condition/disease • -Thalassemia Major – aka Cooley’s anemia (severe anemia present) • In -Thalassemia, there is only one gene inherited from each parent – however – there can be a gene expression – which correlates to varying degree of severity – 0 = no beta chain production – + = decreased beta chain production – = normal beta chain production • Clinical findings of Beta Thalassemia – Increased Hb F (2 alpha/2 gamma) – Varied increases in Hb. A 2 (2 alpha/2 delta) Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Methodology – Solubility test (screening test for sickling hemoglobins) • Based on principle that sickling hemoglobin, in deoxygenated state, is relatively insoluble & forms precipitate in solution • If sickling hemoglobin present – can’t see through. • If No sickling hemoglobin present – see through. – Cellulose acetate (alkaline) hemoglobin electrophoresis • Fresh hemolysate made from packed RBC sample is applied to a cellulose acetate plate using a buffer of alkaline p. H (8. 4– 8. 6); electrophoresis is performed. – Citrate agar (acidic) electrophoresis • Performed at acid p. H (6. 0– 6. 2) after abnormal hemoglobin is detected on cellulose-acetate electrophoresis Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin • Methodology – Hemoglobin A 2 quantitation (2 alpha / 2 delta) • Best accomplished by microcolumn chromatography or highperformance liquid chromatography (HPLC) – Acid elution stain for hemoglobin F (2 alpha / 2 gamma) • Distinguishes erythrocytes containing increased amount of hemoglobin F from normal adult cells – Hemoglobin F quantitation • Based on principle that fetal hemoglobin is resistant to alkali denaturation in 1. 25 mol/L Na. OH for 2 minutes Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Hemoglobin (cont’d) • DNA Technology – Definitive diagnosis of some hemoglobinopathies & thalassemias that involve combinations of genetic defects may require DNA analysis. – DNA sequence of interest may be easily analyzed from whole blood or spots of dried blood on filter paper. – Advantages: provides definitive info on genotype of individuals tested & sometimes direct detection of molecular lesions – Disadvantages: higher cost & lack of availability in many labs – Especially useful in prenatal diagnosis of thalassemia major & sickle cell anemia Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Myoglobin • Structure and Role in the Body – A simple heme protein found in skeletal & cardiac muscle – Can reversibly bind oxygen, similar to hemoglobin molecule – Unable to release oxygen, except under low oxygen tension – Acts as oxygen carrier in cytoplasm of muscle cell – Serves as an extra reserve of oxygen to sustain activity in exercising muscle Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Myoglobin • Clinical Significance – Elevated levels in serum & urine often indicate muscle damage. – Combination of high serum myoglobin & low clearance rate indicates high risk for acute renal failure. – Primary use of serum myoglobin is investigation of chest pain to rule out acute myocardial infarction. – Also has been investigated to aid in diagnosis & differentiation of types of hereditary progressive muscular dystrophy Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins

Myoglobin • Methodology – Several immunoassay methods are used to measure & identify myoglobin (fluorescence, chemiluminescence, immunochromic). – Procedures incorporate binding of specific antibodies to myoglobin. – Resulting chemical or physical change can be measured & correlated to myoglobin concentration. – Methods have been adapted to point-of-care devices for rapid assessment of chest pain. Copyright © 2014 Wolters Kluwer Health | Lippincott Williams & Wilkins
- Slides: 28