Chapter 19 Oxidation Reduction Reactions 19 1 Oxidation











- Slides: 17

Chapter 19 Oxidation - Reduction Reactions 19. 1 Oxidation and Reduction

Oxidation – Reduction • Rules for assigning oxidation numbers 1. Most substances have the same oxidation number as their individual charge (the more electronegative element 1 st) 2. All oxidation numbers in a compound must add up to equal the total charge on the compound 3. All single elements have a oxidation number of zero 4. All single ions have the same oxidation number as their charge 5. Oxygen has a charge of -2, except with F (+2), or peroxides (-1) 6. Hydrogen is usually +1, unless with a metal (-1)

Oxidation Half. Reaction Fe → Fe+2 + 2 e- • Reactions in which the atoms or ions of an element experience an increase in oxidation state • A species whose oxidation number increases is oxidized

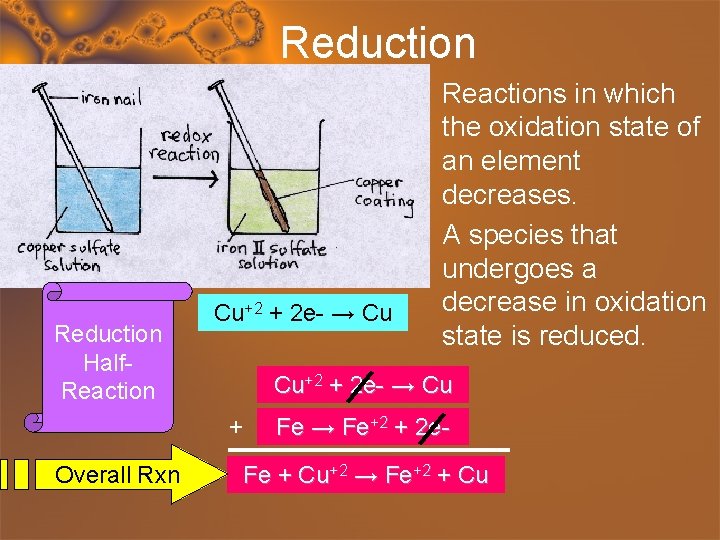
Reduction Half. Reaction • Reactions in which the oxidation state of an element decreases. • A species that undergoes a decrease in oxidation Cu+2 + 2 e- → Cu state is reduced. Cu+2 + 2 e- → Cu + Overall Rxn Fe → Fe+2 + 2 e. Fe + Cu+2 → Fe+2 + Cu

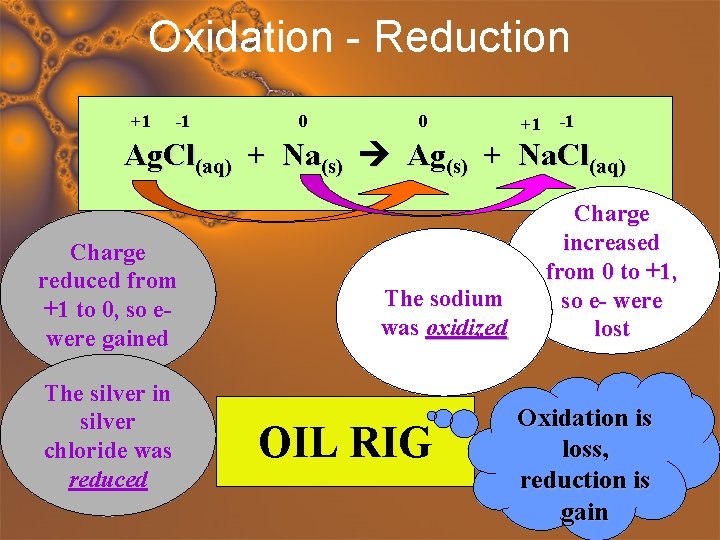
Oxidation - Reduction +1 -1 0 0 +1 -1 Ag. Cl(aq) + Na(s) Ag(s) + Na. Cl(aq) Charge reduced from +1 to 0, so ewere gained The silver in silver chloride was reduced The sodium was oxidized OIL RIG Charge increased from 0 to +1, so e- were lost Oxidation is loss, reduction is gain

Leo the Lion goes Ger Lose Electrons Oxidation Gain Electrons Reduction Electron Loss Means Oxidation

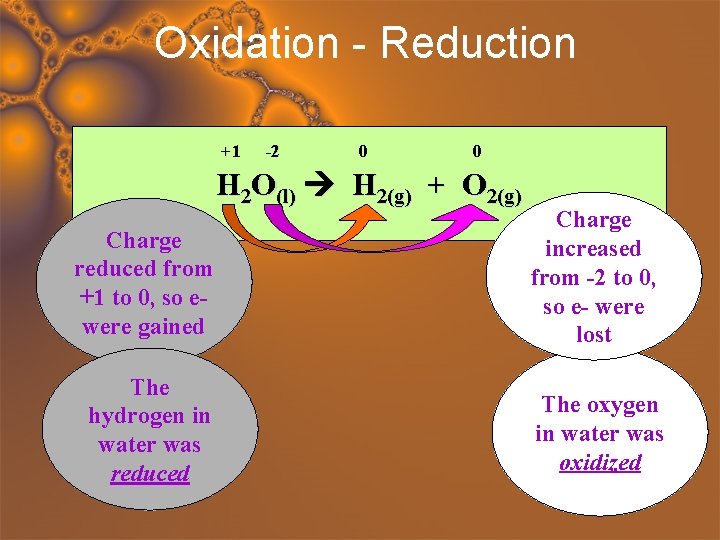
Oxidation - Reduction +1 -2 0 0 H 2 O(l) H 2(g) + O 2(g) Charge reduced from +1 to 0, so ewere gained Charge increased from -2 to 0, so e- were lost The hydrogen in water was reduced The oxygen in water was oxidized

Example • Household Bleach removes stains through a redox reaction: Stain molecules (s) + OCl- (aq) → colorless molecules (s) + Cl- (aq) • Determine the oxidation numbers of oxygen & chlorine in OCl-2 +1

Chapter 19 Oxidation - Reduction Reactions 19. 2 Balancing Redox Equations

Example • Balance the following reaction: I- + Mn. O 4 - + H+ → Mn. O 2 + I 2 + H 2 O 2 I- + Mn. O 4 - + 4 H+ → Mn. O 2 + I 2 + 2 H 2 O But this reaction is balanced for mass not charge! A half-reaction system has to be used to balance for charge.

Half-Reaction Method 1. Write the formula equation then ionic equation 2. Assign oxidation numbers. Exclude anything with an ox. # of zero, or that doesn’t change ox. # 3. Write the ½ rxn for oxidation 4. Balance the atoms 5. Balance the charge (w/ electrons) 6. Write the ½ rxn for reduction 7. Balance the atoms 8. Balance the charge (w/ electrons) 9. Use coefficients to ensure the # of e- lost in ox. equals the # of e- gained in red. 10. Combine both ½ rxns and cancel (like Hess’s Law) 11. Combine ions to form initial compounds.

Example Now try to balance the following reaction: I- + Mn. O 4 - + H+ → Mn. O 2 + I 2 + H 2 O Half-Reactions: 2 Mn. O 4 - + 8 H+ + 6 e- → 2 Mn. O 2 + 4 H 2 O 6 I- → 3 I 2 + 6 e. Overall Balanced Equation: 6 KI + 2 KMn. O 4 + 8 HCl → 2 Mn. O 2 + 3 I 2 + 4 H 2 O + 8 KCl

Chapter 19 Oxidation - Reduction Reactions 19. 3 Oxidizing and Reducing Agents

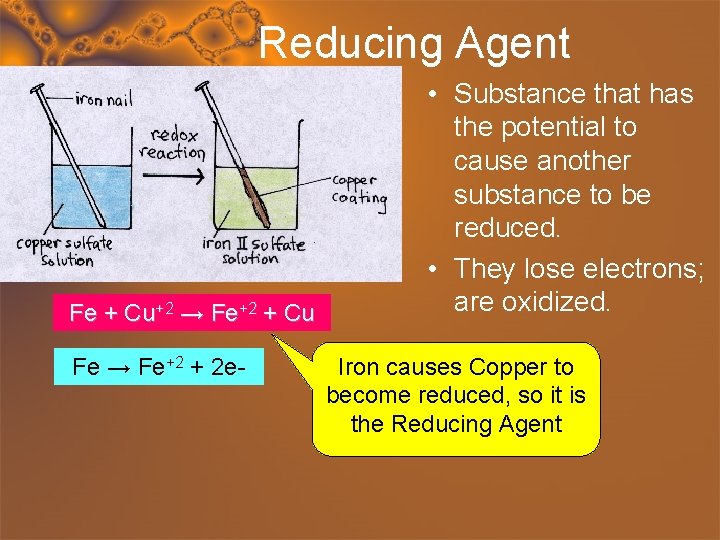
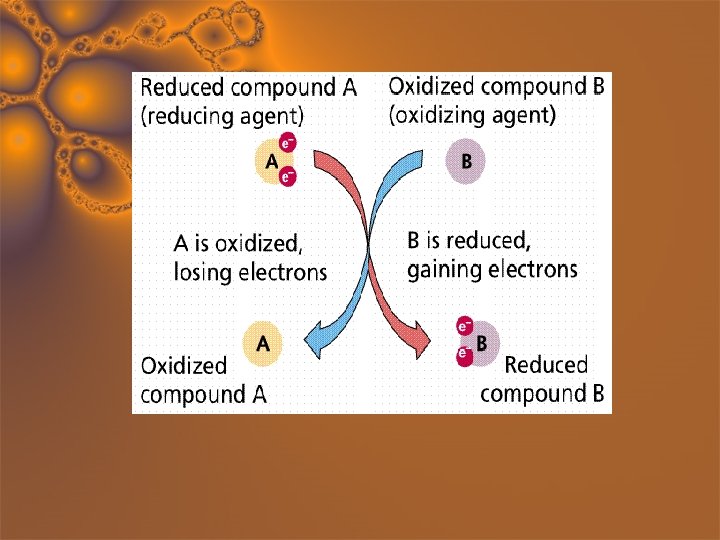
Reducing Agent Fe + Cu+2 → Fe+2 + Cu Fe → Fe+2 + 2 e- • Substance that has the potential to cause another substance to be reduced. • They lose electrons; are oxidized. Iron causes Copper to become reduced, so it is the Reducing Agent

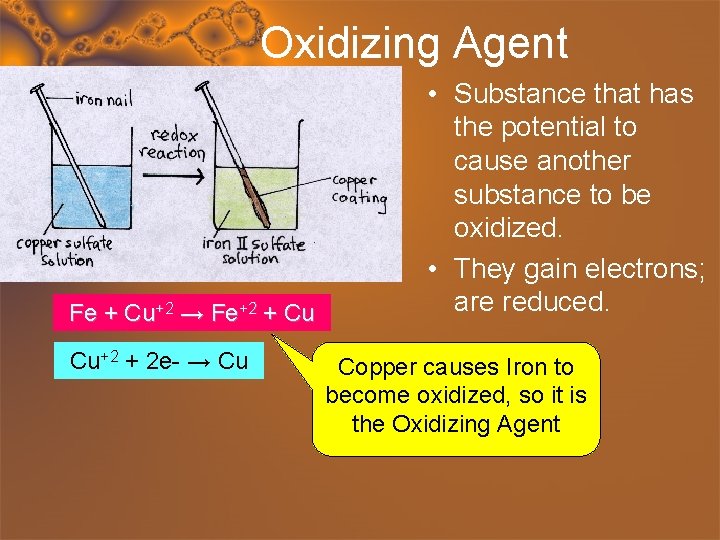
Oxidizing Agent Fe + Cu+2 → Fe+2 + Cu Cu+2 + 2 e- → Cu • Substance that has the potential to cause another substance to be oxidized. • They gain electrons; are reduced. Copper causes Iron to become oxidized, so it is the Oxidizing Agent

Disproportionation/ Autooxidation • A process by which a substance acts as both an oxidizing and reducing agent
