Chapter 19 Chemical Bonds Chemical Compounds Chemical compounds

Chapter 19 Chemical Bonds

Chemical Compounds Chemical compounds are formed when two or more atoms combine chemically to form a new product with a new identity

Chemical Formula o Chemical formula = used to tell what elements a compound contains and the exact number of atoms of each element o It’s like a recipe for the compound n Ex: Ca. F 2 1 Ca atom to 2 F atoms

Atoms are joined by CHEMICAL BONDS A CHEMICAL BOND is a FORCE that joins atoms together to form a compound or molecule n Forms when atoms gain, lose, or share electrons n Follows the octet rule n 8 electrons in valence shell = stability
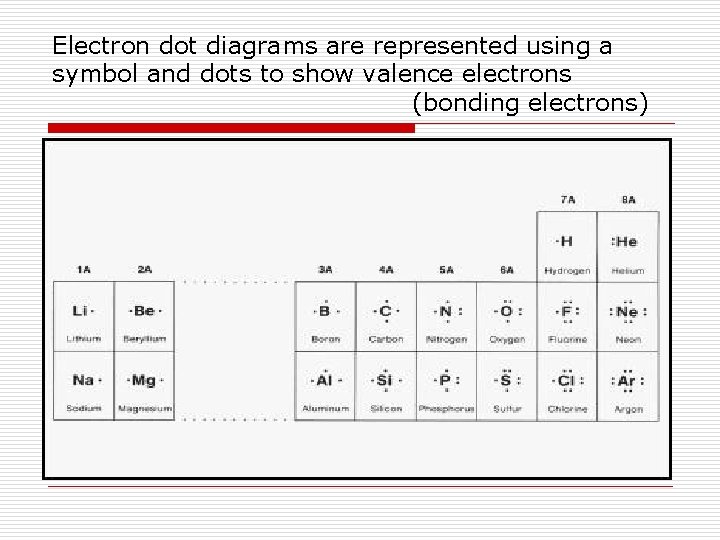
Electron dot diagrams are represented using a symbol and dots to show valence electrons (bonding electrons)

There are 2 exceptions to the octet rule: Hydrogen and Helium are stable with 2 electrons in their valence shell

Each group is assigned an oxidation number based on the number of electrons in the valence shell Group 1 A: +1 Group 2 A: +2 Group 3 A: +3 Group 4 A: +/- 4 Group 5 A: -3 Group 6 A: -2 Group 7 A: -1

Types of Chemical Bonds There are 3 types of chemical bonds o Metallic Bonds o Ionic Bonds o Covalent Bonds § Polar § Nonpolar

METALLIC BONDS Formed when metallic ions (charged atoms) are held together by their attraction to a common pool of electrons

Metallic (continued) Electron pool makes metals malleable, ductile, and lustrous in conjunction with being highly conductive. Mixture of metals formed from metallic bonds are called ALLOYS o o Sterling silver: (Ag and Cu) Brass: (Zn and Cu) Filling in teeth: (Ag, Cu, Sn, Hg) Stainless Steel: (Fe, Cr, Ni, C)

IONIC BONDS Occur between a metal and a nonmetal Na+ and Cl- form Na. Cl n Cation and anion pulled together by electrostatic attraction n Creates an Ionic Compound n Generally have a high melting point n Solids are usually crystalline n Salts conduct electricity when in solution § Separate into ions when in solution
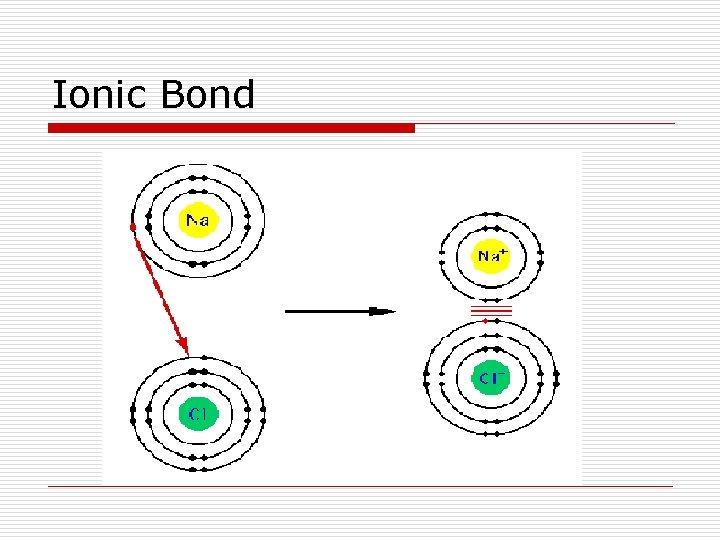
Ionic Bond

Forming Binary Compounds Atoms combine to form binary compounds using valence shell electrons: n There must be a + and – charged ion o An ION is a charged atom that has gained or lost electrons n + is cation, - is anion n The ions must appear in the order +, n The total + and – must add to equal zero n Numbers of ions used are represented using subscripts

Naming Binary Compounds o When naming binary compounds start with the cation name (ex. Sodium) o Write the name of the anion and change the ending of the word to –IDE (ex. Chloride) o If a transition metal is used, use roman numerals to indicate the correct oxidation number o When using polyatomic ions, do not change the name of the polyatomic ion o Naming covalent molecules involves using prefixes to indicate numbers of atoms involved.

COVALENT BONDS Form as a result of electrons being shared between atoms of nonmetals n n Form molecules Do not separate in solution Do not conduct electricity May be multiple bonds n Each pair of electrons represents a bond § 1 pair = single bond § 2 pairs= double bond § 3 pairs = triple bonds

Some compounds have water A hydrate is a compound with water chemically attached Hydrate = water We represent the presence of water as follows: Mg 3(PO 4)2. 4 H 20 This is read, Magnesium phosphate tetrahydrate Compounds without water are called anhydrous
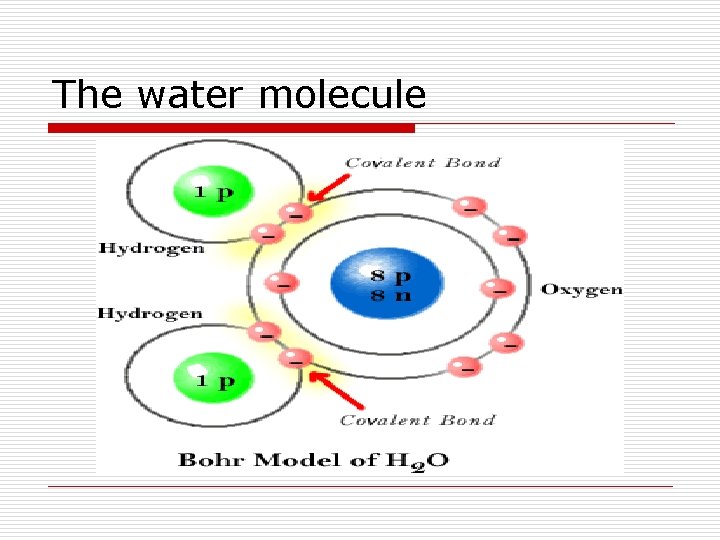
The water molecule

Special nonmetals bond to themselves Certain nonmetals cannot be alone and therefore bond to themselves when alone Called DIATOMICS Br 2 H 2 O 2 N 2 Cl 2 I 2 F 2 Bonding to themselves satisfies the octet rule and keeps them stable

Polar and Nonpolar Covalent Bonds n Electrons are shared equally by atoms
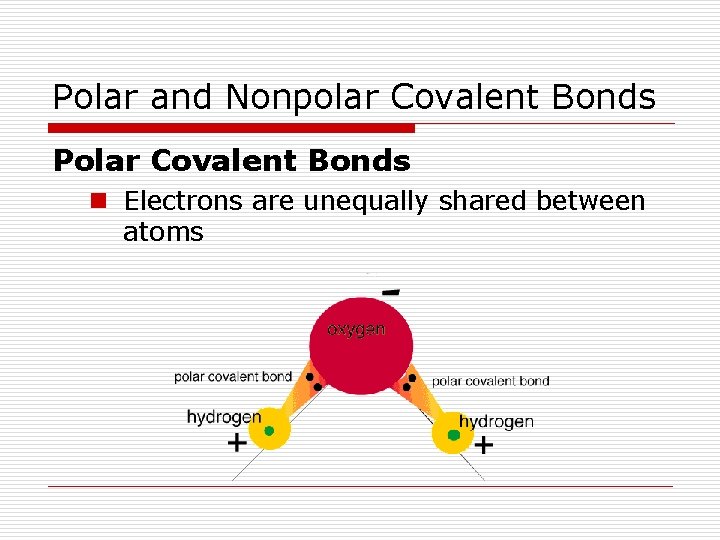
Polar and Nonpolar Covalent Bonds Polar Covalent Bonds n Electrons are unequally shared between atoms

Naming binary covalent compounds Greek prefixes are used to indicate how many atoms of each element are in a binary covalent compound 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa. Prefixes are also used to name hydrates
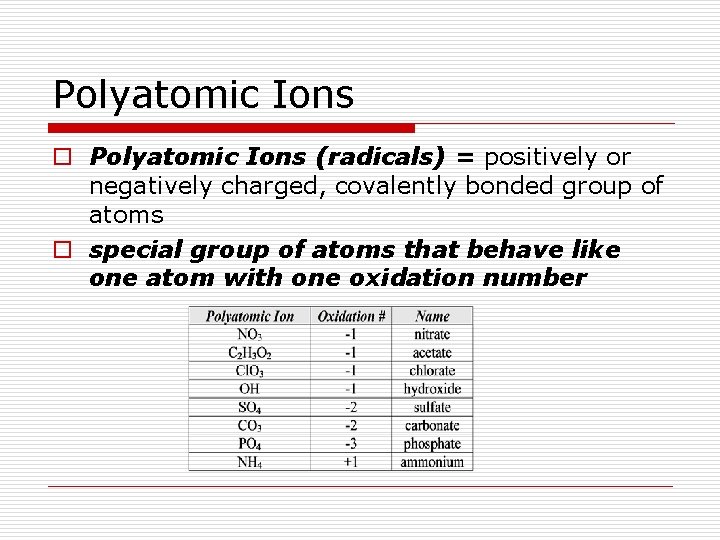
Polyatomic Ions o Polyatomic Ions (radicals) = positively or negatively charged, covalently bonded group of atoms o special group of atoms that behave like one atom with one oxidation number

Mass of a formula may be calculated In order to calculate the mass of a formula, several steps are taken 1. List the elements in the compound 2. Calculate the number of atoms of each element in the compound 3. Multiply the number of atoms by the atomic mass of each element 4. Add the atomic masses and label the sum amu
- Slides: 23