Chapter 19 Acids Bases Properties of Acids Bases

Chapter 19: Acids & Bases

Properties of Acids & Bases • Acid: substance dissociates to form H+ in solution • • • Tastes sour Feels sticky Conductor of electricity Turns blue litmus red Example: HCl • Base: substance dissociates to form OH- (hydroxide) in solution • • • Tastes bitter Feels slippery Conductor of electricity Turns red litmus blue Example: Na. OH

Arrhenius Model • Arrhenius model: • Traditional definition for acids and bases • States that an acid is a substance that contains hydrogen and ionizes to produce hydrogen ions in aqueous solution, and a base is a substance that contains a hydroxide group and dissociates to produce a hydroxide ion in solution. – HCl ionizes to produce H+ ions. HCl(g) → H+(aq) + Cl–(aq) – Na. OH dissociates to produce OH– ions. Na. OH(s) → Na+(aq) + OH–(aq) * Some solutions produce hydroxide ions even though they do not contain a hydroxide group.
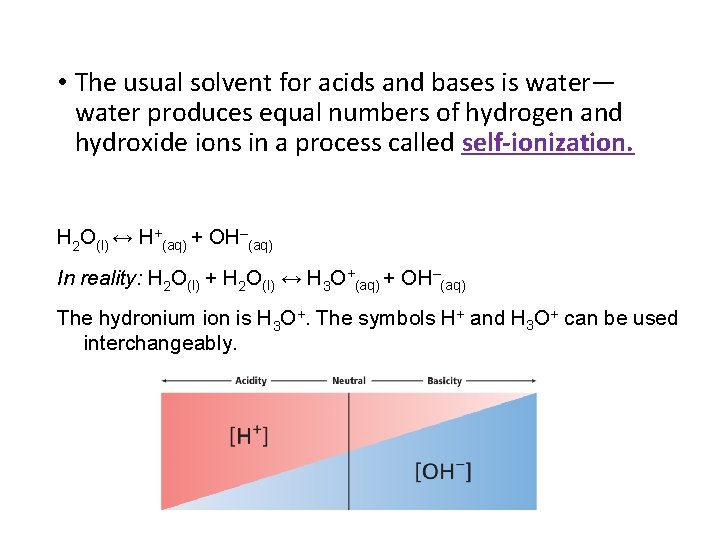
• The usual solvent for acids and bases is water— water produces equal numbers of hydrogen and hydroxide ions in a process called self-ionization. H 2 O(l) ↔ H+(aq) + OH–(aq) In reality: H 2 O(l) + H 2 O(l) ↔ H 3 O+(aq) + OH–(aq) The hydronium ion is H 3 O+. The symbols H+ and H 3 O+ can be used interchangeably.

Brønsted-Lowry Model • Brønsted-Lowry Model: • More general definition for acids and bases • States that an acid is a hydrogen ion donor and a base is a hydrogen ion acceptor ** NH 3 is a Brønsted-Lowry base, but not an Arrhenius base NH 3 (aq) + H 2 O (l) NH 4+ (aq) + OH- (aq) - H 2 O (acting as an acid) donates a proton to ammonia - NH 3 (acting as a base) accepts a proton from water • Conjugate acid: the species produced when a base accepts a hydrogen ion • (NH 4+ in the above reaction) • Conjugate base: the species produced when an acid donates a hydrogen ion • (OH- in example) • Conjugate acid-base pair: consists of two substances related to each other by donating and accepting a single hydrogen ion
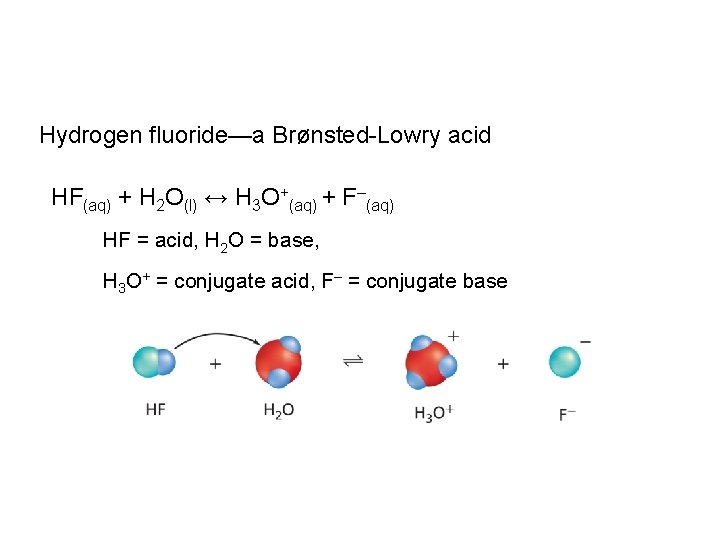
Hydrogen fluoride—a Brønsted-Lowry acid HF(aq) + H 2 O(l) ↔ H 3 O+(aq) + F–(aq) HF = acid, H 2 O = base, H 3 O+ = conjugate acid, F– = conjugate base
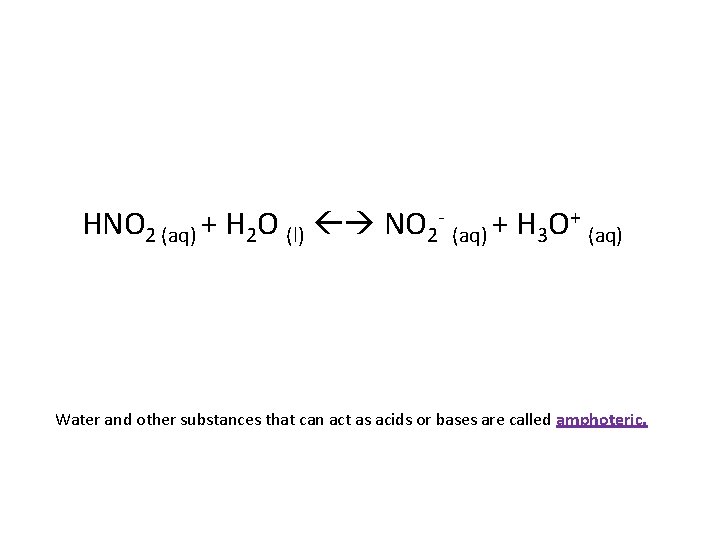
HNO 2 (aq) + H 2 O (l) NO 2 - (aq) + H 3 O+ (aq) Water and other substances that can act as acids or bases are called amphoteric.

Monoprotic and Polyprotic Acids • Monoprotic Acid: an acid that can donate only one hydrogen ion • Only ionizable hydrogen atoms can be donated • Polyprotic Acid: an acid that can donate more than one hydrogen ion
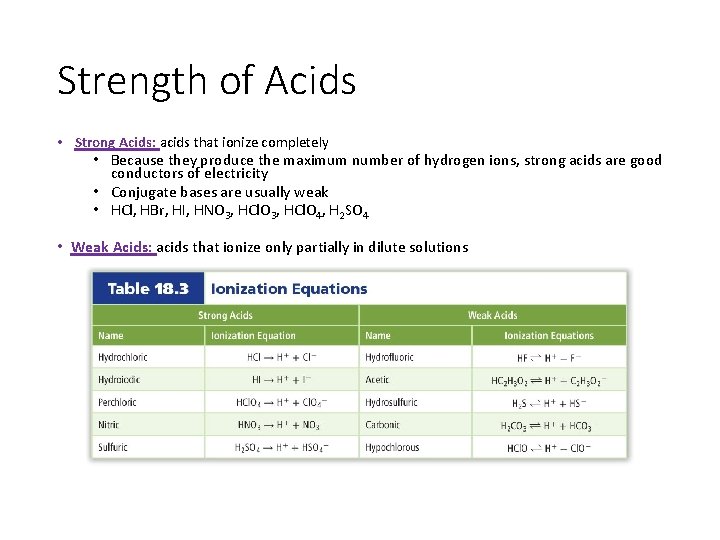
Strength of Acids • Strong Acids: acids that ionize completely • Because they produce the maximum number of hydrogen ions, strong acids are good conductors of electricity • Conjugate bases are usually weak • HCl, HBr, HI, HNO 3, HCl. O 4, H 2 SO 4 • Weak Acids: acids that ionize only partially in dilute solutions

Strength of Bases • Strong Base: a base that dissociates completely into metal ions and hydroxide ions • Alkali metals or heavier alkaline earth metals • Weak base: ionizes only partially in dilute aqueous solution

p. H scale: measure of the acidity of an aqueous solution - Concentrations of H+ ions are often small numbers expressed in exponential notation - p. H is the negative logarithm of the hydrogen ion concentration of a solution. Changing [H+] by a factor of 10 causes the p. H to change by 1 unit p. H = -log [H+] - Litmus paper and a p. H meter with electrodes can determine the p. H of a solution.
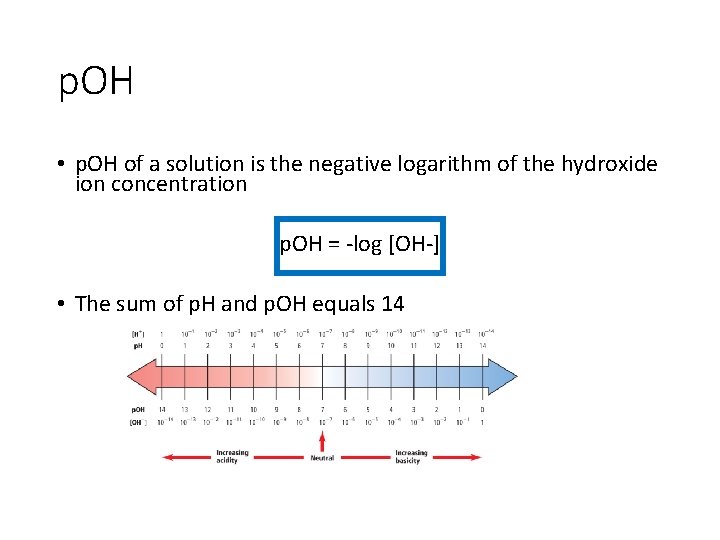
p. OH • p. OH of a solution is the negative logarithm of the hydroxide ion concentration p. OH = -log [OH-] • The sum of p. H and p. OH equals 14
![Practice • Calculate the [H+] and [OH-] when p. H = 7. 40 • Practice • Calculate the [H+] and [OH-] when p. H = 7. 40 •](http://slidetodoc.com/presentation_image_h2/69f400302b5e2cfb04128346e0e3848e/image-14.jpg)
Practice • Calculate the [H+] and [OH-] when p. H = 7. 40 • Calculate the p. H and p. OH when [OH-] = 4. 0 x 10 -3 M

Neutralization • A neutralization reaction is a reaction in which an acid and a base in an aqueous solution react to produce a salt and water. • A salt is an ionic compound made up of a cation from a base and an anion from an acid. • Neutralization is a double-replacement reaction.

Titrations • Titration is a method for determining the concentration of a solution by reacting a known volume of that solution with a solution of known concentration. • In a titration procedure, a measured volume of an acid or base of unknown concentration is placed in a beaker, and initial p. H recorded. • A buret is filled with the titrating solution of known concentration, called a titrant.
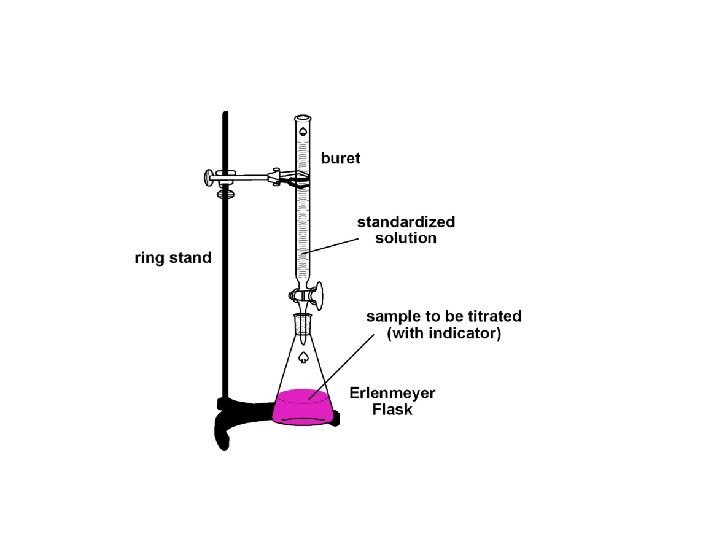

• Measured volumes of the standard solution are added slowly and mixed into the solution in the beaker, and the p. H is read and recorded after each addition. The process continues until the reaction reaches the equivalence point, which is the point at which moles of H+ ion from the acid equals moles of OH– ion from the base. • An abrupt change in p. H occurs at the equivalence point.
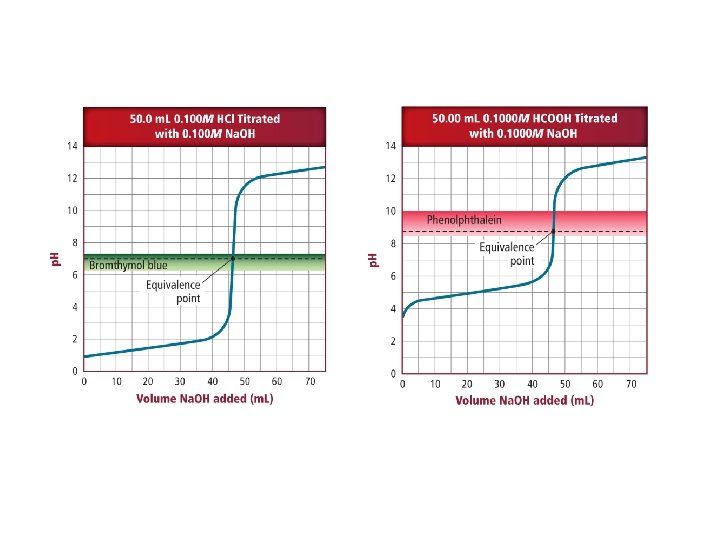
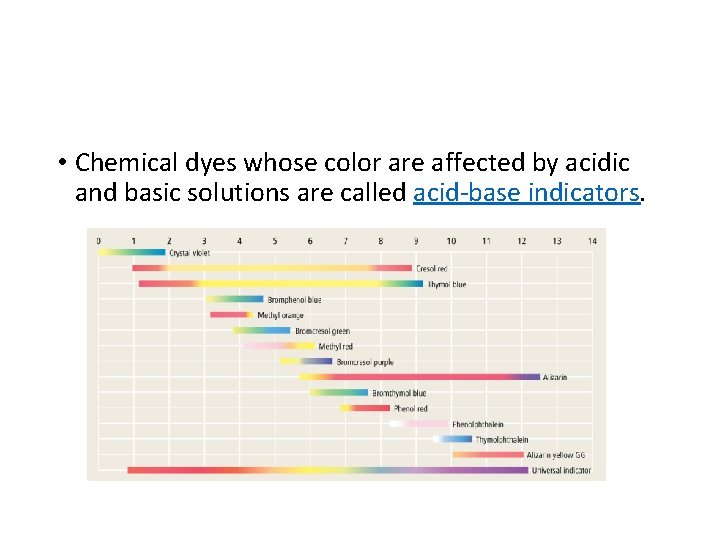
• Chemical dyes whose color are affected by acidic and basic solutions are called acid-base indicators.

• An end point is the point at which an indicator used in a titration changes color. • An indicator will change color at the equivalence point.

Molarity from Titration • ** Must have a balanced equation** • Use stoichiometry A volume of 18. 28 m. L of a standard solution of 0. 1000 M Na. OH was required to neutralize 25. 00 m. L of a solution of methanoic acid (HCOOH). What is the molarity of the acid solution

• What is the molarity of nitric acid solution is 43. 33 m. L of 0. 1000 M KOH solution is needed to neutralize 20. 00 m. L of the acid solution.

• How many milliliters of 0. 500 M Na. OH would neutralize 25. 00 m. L of 0. 100 M H 3 PO 4.
- Slides: 24