Chapter 18 THE SECOND LAW OF THERMODYNAMICS REVERSIBLE

Chapter 18 THE SECOND LAW OF THERMODYNAMICS

REVERSIBLE PROCESSES Most laws of physics are reversible in time: Newton’s Laws, optics, electromagnetism, relativity, … Being reversible means if you record some physical processes, and play the tape backward, the action observed in the video still obeys the same law. Examples: • A ball rolling up and down a frictionless bowl. • A ball bouncing perfectly elastically off a wall. • A beam of light passing through a lens. • A planet orbiting the sun.

IRREVERSIBLE PROCESSES There are, however, many processes that only goes one way. When you play such processes backward, something is “unnatural” about the reversed processes. These are call irreversible processes. Examples: • Heat flowing from a hot object to a cold object. • Ice melting. • A glass breaking after falling on the ground. • An explosion. • A gas expanding from a small box to a big box. This is curious, because the laws that governs the details of these processes are reversible, so somehow reversible laws can combine to give irreversible processes.

THE SECOND LAW AND ENTROPY S Entropy (unit: J/K) is an indication of the “disorder” of the system. Any system if left alone, tends to become more disordered. This is essentially the idea behind the Second Law of Thermodynamics: The entropy of the universe can never decrease. In other words, the universe tends to become more and more “messy”.

HEAT ENGINES It turns out the Second Law forbids one from building a heat engine that is 100% efficient. This is one of the key result of this chapter, so we will devote some time in studying heat engines. A heat engine is any device that uses heat as a source of energy to do mechanical work. It always involves at least three parts: 1. Hot reservoir (sources of energy) 2. Cold reservoir (absorbs waste heat) 3. Engine (generates work) Note that QC is negative because it represents heat going out of the engine.

HEAT ENGINE AND CONSERVATION OF ENERGY Note that QC is negative because it represents heat going out of the engine.
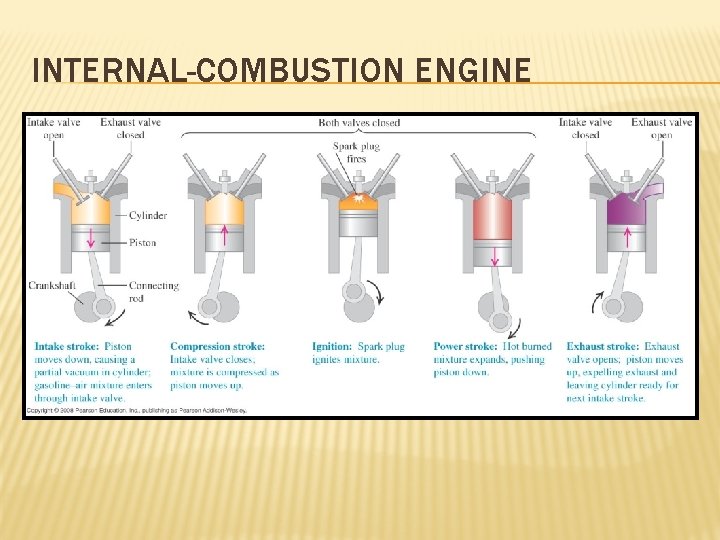
INTERNAL-COMBUSTION ENGINE

OTHER FORMS OF THE SECOND LAW Two other equivalent statements of the Second Law: • It is impossible for any process to have as its sole result the transfer of heat from a cooler object to a hotter object. • It is impossible for any system to undergo a process in which it absorbs heat from a reservoir at a single temperature and converts the heat completely into mechanical work, with the system ending in the same state in which it began.
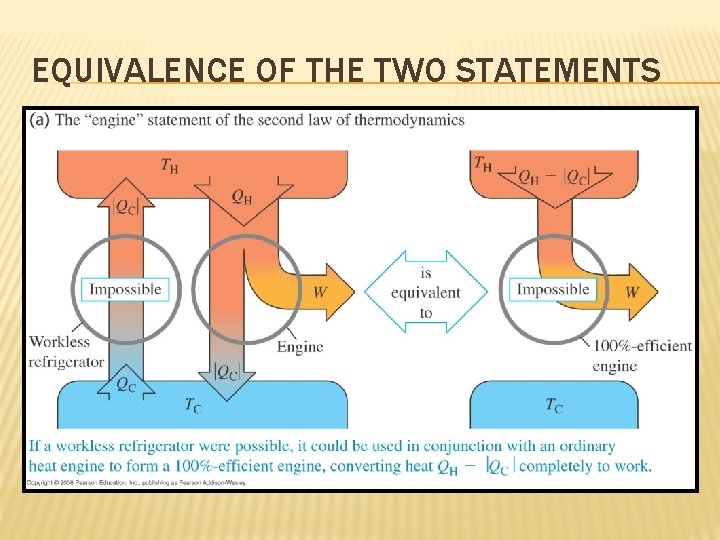
EQUIVALENCE OF THE TWO STATEMENTS
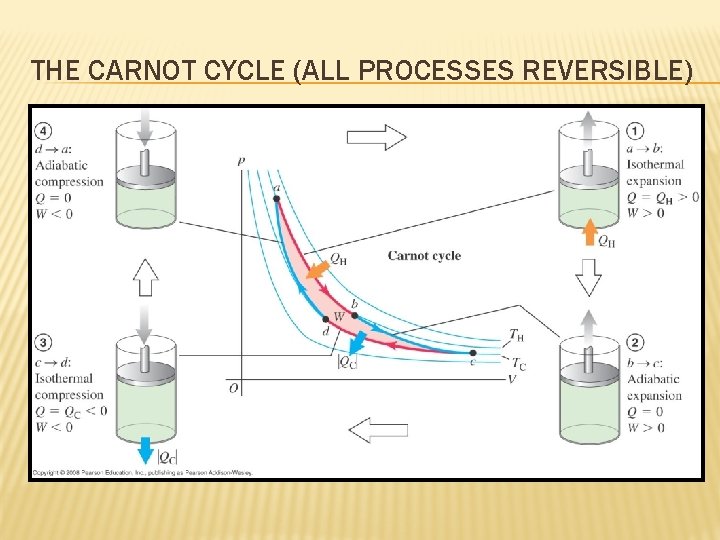
THE CARNOT CYCLE (ALL PROCESSES REVERSIBLE)

THE EFFICIENCY OF THE CARNOT CYCLE

CARNOT ENGINE IS A REVERSIBLE ENGINE Carnot engine is an example of a reversible engine because every step can be done in the reversed direction. • The efficiency of a Carnot engine depends only on the temperatures of the reservoirs. • This is the highest efficiency allowed by the laws of thermodynamics. • The efficiency of all reversible engines are the same.

SUMMARY OF EQUATIONS
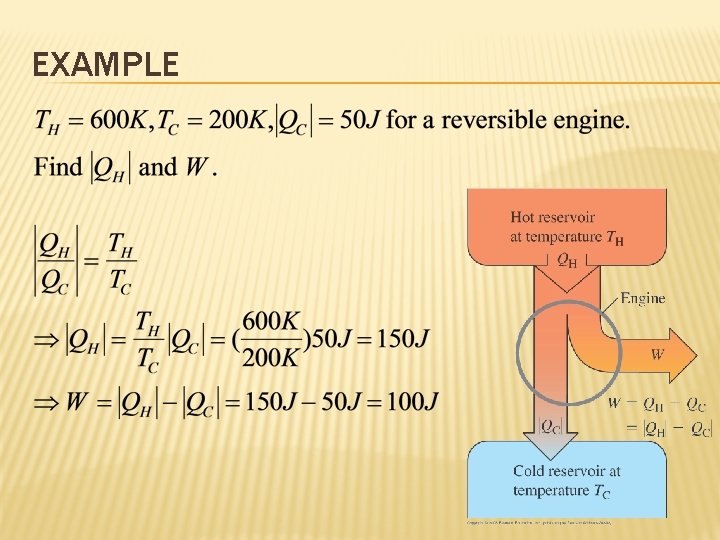
EXAMPLE

W VERSUS J Watts is the unit for power (rate of energy consumption). 1 W=1 J/s. For example, a light bulb of 100 W = 100 J/s consumes 100 J of energy per second. QC, QH and W are normally given in J. In Web. Assign, many questions gives the units in W. For example, when the question states QH=20 W, it just means that the engine runs at a rate that takes in 20 J of QH per second. If you use this to calculate QC for example, your answer will be in W also.
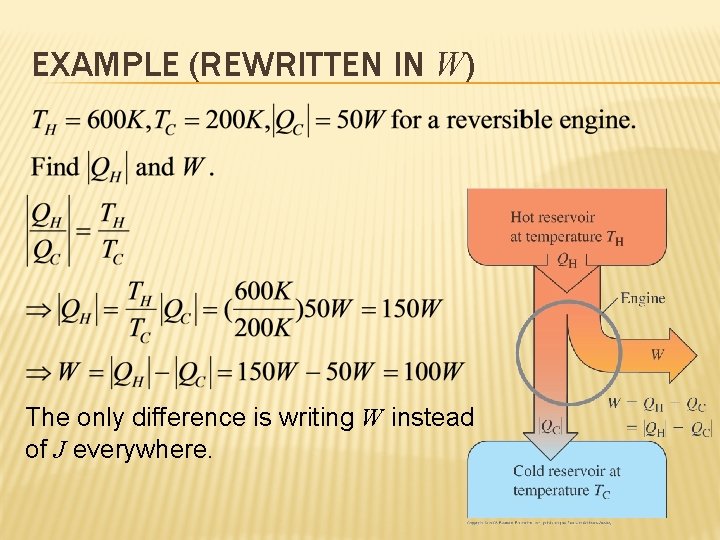
EXAMPLE (REWRITTEN IN W) The only difference is writing W instead of J everywhere.
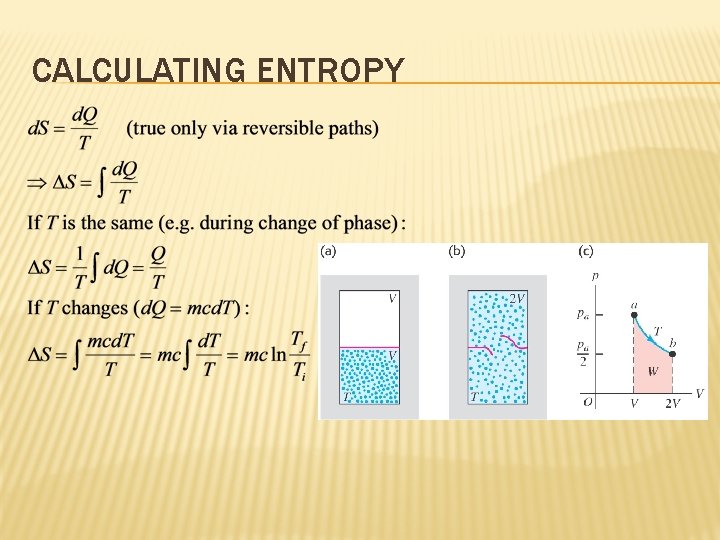
CALCULATING ENTROPY

EXAMPLE: ENTROPY CHANGE IN WATER 1 kg of liquid water is heated from 70°C to steam at 120°C. Find the change in entropy. Change of phase involved, need to be careful!
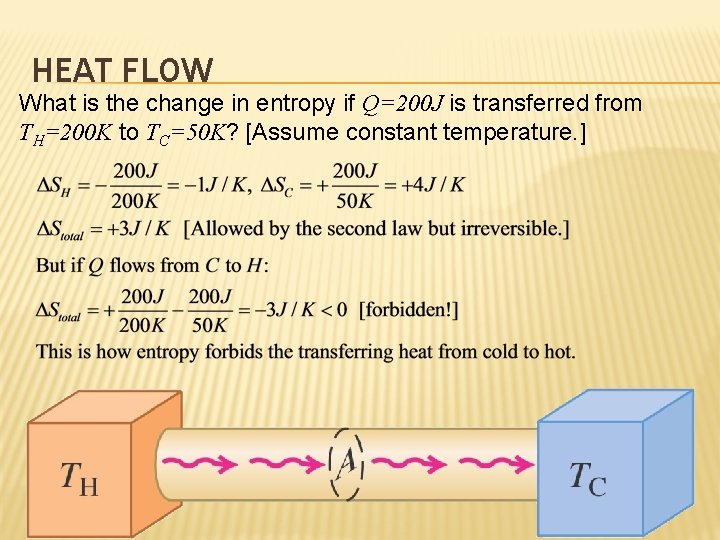
HEAT FLOW What is the change in entropy if Q=200 J is transferred from TH=200 K to TC=50 K? [Assume constant temperature. ]

THE BEST ENGINE?

SUMMARY OF EQUATIONS

CONCEPTUAL QUESTION 1 Consider a hypothetical device that takes 1000 J of heat from a hot reservoir at 300 K, ejects 200 J of heat to a cold reservoir at 100 K, and produces 800 J of work. Does this device violate the first law of thermodynamics ? This device doesn't violate the first law of thermodynamics because no energy is being created nor destroyed. All the energy is conserved.

CONCEPTUAL QUESTION 2 Consider a hypothetical device that takes 1000 J of heat from a hot reservoir at 300 K, ejects 200 J of heat to a cold reservoir at 100 K, and produces 800 J of work. Does this device violate the second law of thermodynamics ?

THE MICROSCOPIC ORIGIN OF ENTROPY

COUNTING STATES How many ways are there to get a 7 from two dices? How many ways are there to get an 8? How many ways are there to get an 2? Macroscopic systems are dominated by the most probable states How many words can you make out of the following letters? aabbbbcdddddddeeee

MAXWELL’S DEMON
RATCHET AND PAWL (BROWNIAN MOTOR)
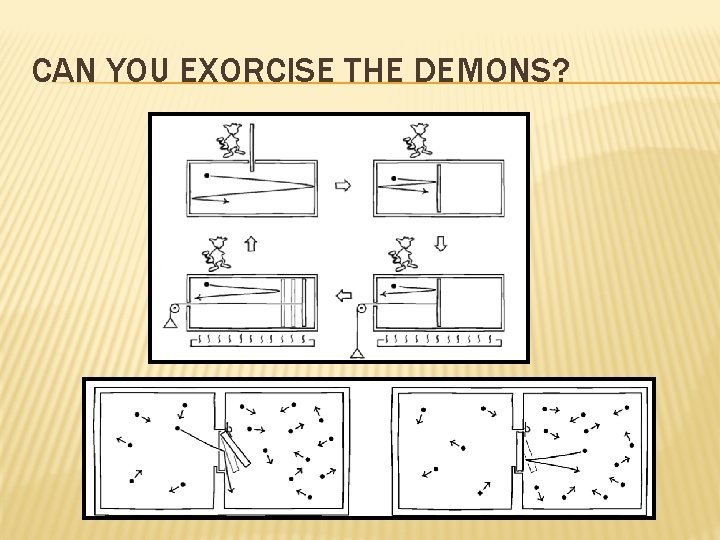
CAN YOU EXORCISE THE DEMONS?

INFORMATION THEORY Coin flip: (Head, Tail, Tail) can be encoded as (1001000). Interesting, if the coin is biased, so that Head is more likely to appear, then the number of bits required would decrease. Shannon Entropy: Indicates the minimal number of bits per letter required to send (or encode) a message. [1/0 is called one bit of information. ]

LAWS OF BLACK HOLES AND THERMODYNAMICS Zeroth Thermodynamics Black Holes T constant on a body in thermal equalibrium Surface gravity κ constant on the horizon of a black hole T=0 cannot be reached κ=0 cannot be reached First Second Third

- Slides: 31