Chapter 18 Electrochemistry Section 18 4 Cell Potential












- Slides: 13

Chapter 18 Electrochemistry

Section 18. 4 Cell Potential, Electrical Work, and Free Energy Work § Work is never the maximum possible if any current is flowing. § In any real, spontaneous process some energy is always wasted – the actual work realized is always less than the calculated maximum. Copyright © Cengage Learning. All rights reserved 2

Section 18. 4 Cell Potential, Electrical Work, and Free Energy Maximum Cell Potential § Directly related to the free energy difference between the reactants and the products in the cell. § ΔG° = –n. FE° Ø F = 96, 485 C/mol e– Copyright © Cengage Learning. All rights reserved 3

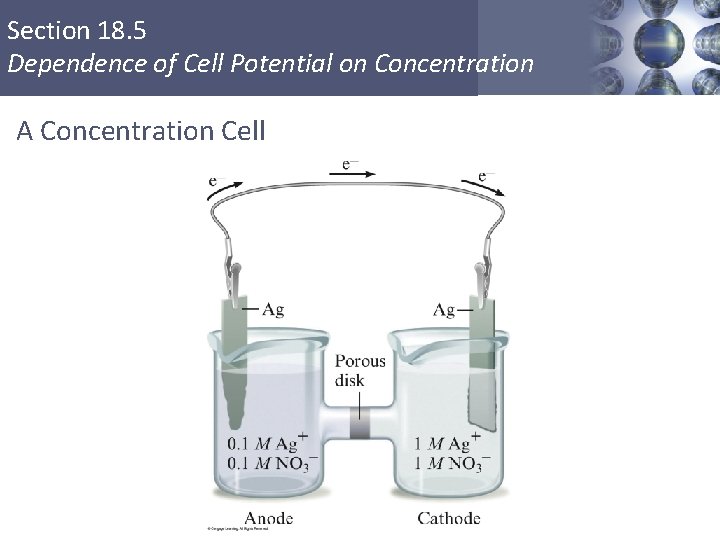
Section 18. 5 Dependence of Cell Potential on Concentration A Concentration Cell Copyright © Cengage Learning. All rights reserved 4

Section 18. 5 Dependence of Cell Potential on Concentration Nernst Equation § The relationship between cell potential and concentrations of cell components § At 25°C: or (at equilibrium) Copyright © Cengage Learning. All rights reserved 5

Section 18. 5 Dependence of Cell Potential on Concentration CONCEPT CHECK! Explain the difference between E and E°. When is E equal to zero? When is E° equal to zero? Copyright © Cengage Learning. All rights reserved 6

Section 18. 5 Dependence of Cell Potential on Concentration EXERCISE! A concentration cell is constructed using two nickel electrodes with Ni 2+ concentrations of 1. 0 M and 1. 00 × 10 -4 M in the two half-cells. Calculate the potential of this cell at 25°C. Copyright © Cengage Learning. All rights reserved 7

Section 18. 5 Dependence of Cell Potential on Concentration CONCEPT CHECK! You make a galvanic cell at 25°C containing: § A nickel electrode in 1. 0 M Ni 2+(aq) § A silver electrode in 1. 0 M Ag+(aq) Sketch this cell, labeling the anode and cathode, showing the direction of the electron flow, and calculate the cell potential. Copyright © Cengage Learning. All rights reserved 8

Section 18. 8 Electrolysis § Forcing a current through a cell to produce a chemical change for which the cell potential is negative. Copyright © Cengage Learning. All rights reserved 9

Section 18. 8 Electrolysis Stoichiometry of Electrolysis § How much chemical change occurs with the flow of a given current for a specified time? current and time �quantity of charge � moles of electrons �moles of analyte � grams of analyte Copyright © Cengage Learning. All rights reserved 10

Section 18. 8 Electrolysis Stoichiometry of Electrolysis § current and time �quantity of charge Coulombs of charge = amps (C/s) × seconds (s) § quantity of charge �moles of electrons Copyright © Cengage Learning. All rights reserved 11

Section 18. 8 Electrolysis CONCEPT CHECK! An unknown metal (M) is electrolyzed. It took 52. 8 sec for a current of 2. 00 amp to plate 0. 0719 g of the metal from a solution containing M(NO 3)3. What is the metal? Copyright © Cengage Learning. All rights reserved 12

Section 18. 8 Electrolysis CONCEPT CHECK! Consider a solution containing 0. 10 M of each of the following: Pb 2+, Cu 2+, Sn 2+, Ni 2+, and Zn 2+. Predict the order in which the metals plate out as the voltage is turned up from zero. Do the metals form on the cathode or the anode? Explain. Copyright © Cengage Learning. All rights reserved 13