Chapter 18 Electrochemistry Section 18 1 Balancing OxidationReduction

Chapter 18 Electrochemistry

Section 18. 1 Balancing Oxidation-Reduction Equations POP QUIZ § What is electrochemistry? § What is a voltaic or galvanic cell? § List 2 types of voltaic (galvanic cell)?

Section 18. 1 Balancing Oxidation-Reduction Equations Review of Terms § Electrochemistry – the study of the interchange of chemical and electrical energy § Oxidation–reduction (redox) reaction – involves a transfer of electrons from the reducing agent to the oxidizing agent § Oxidation – loss of electrons § Reduction – gain of electrons § Reducing agent – electron donor § Oxidizing agent – electron acceptor Copyright © Cengage Learning. All rights reserved 3

Electrochemical processes are oxidation-reduction reactions in which: • the energy released by a spontaneous reaction is converted to electricity or • electrical energy is used to cause a nonspontaneous reaction to occur 0 0 2 Mg (s) + O 2 (g) 2 Mg O 2 + 4 e- 2+ 2 - 2 Mg. O (s) 2 Mg 2+ + 4 e- Oxidation half-reaction (lose e-) 2 O 2 - Reduction half-reaction (gain e-) 4

Oxidation number The charge the atom would have in a molecule (or an ionic compound) if electrons were completely transferred. 1. Free elements (uncombined state) have an oxidation number of zero. Na, Be, K, Pb, H 2, O 2, P 4 = 0 2. In monatomic ions, the oxidation number is equal to the charge on the ion. Li+, Li = +1; Fe 3+, Fe = +3; O 2 -, O = -2 3. The oxidation number of oxygen is usually – 2. In H 2 O 2 and O 22 - it is – 1. 5

4. The oxidation number of hydrogen is +1 except when it is bonded to metals in binary compounds. In these cases, its oxidation number is – 1. 5. Group IA metals are +1, IIA metals are +2 and fluorine is always – 1. 6. The sum of the oxidation numbers of all the atoms in a molecule or ion is equal to the charge on the molecule or ion. HCO 3− Identify the oxidation numbers of all the atoms in HCO 3− ? O = − 2 H = +1 3(− 2) + 1 + x = − 1 C = +4 6

Balancing Redox Equations The oxidation of Fe 2+ to Fe 3+ by Cr 2 O 72 - in acid solution? 1. Write the unbalanced equation for the reaction in ionic form. Fe 2+ + Cr 2 O 72 - Fe 3+ + Cr 3+ 2. Separate the equation into two half-reactions. +2 Fe 2+ Oxidation: Reduction: +3 +6 Cr 2 O 7 Fe 3+ 2 - +3 Cr 3+ 3. Balance the atoms other than O and H in each half-reaction. Cr 2 O 72 - 2 Cr 3+ 7

Balancing Redox Equations 4. For reactions in acid, add H 2 O to balance O atoms and H+ to balance H atoms. Cr 2 O 7214 H+ + Cr 2 O 72 - 2 Cr 3+ + 7 H 2 O 5. Add electrons to one side of each half-reaction to balance the charges on the half-reaction. Fe 2+ 6 e- + 14 H+ + Cr 2 O 72 - Fe 3+ + 1 e 2 Cr 3+ + 7 H 2 O 6. If necessary, equalize the number of electrons in the two halfreactions by multiplying the half-reactions by appropriate coefficients. 6 Fe 2+ 6 Fe 3+ + 6 e 6 e- + 14 H+ + Cr 2 O 72 - 2 Cr 3+ + 7 H 2 O 8

Balancing Redox Equations 7. Add the two half-reactions together and balance the final equation by inspection. The number of electrons on both sides must cancel. Oxidation: 6 Fe 2+ Reduction: 6 e- + 14 H+ + Cr 2 O 72 - + 6 Fe 2+ 6 Fe 3+ + 6 e 2 Cr 3+ + 7 H 2 O 6 Fe 3+ + 2 Cr 3+ + 7 H 2 O 8. Verify that the number of atoms and the charges are balanced. 14 x 1 – 2 + 6 x 2 = 24 = 6 x 3 + 2 x 3 9. For reactions in basic solutions, add OH- to both sides of the equation for every H+ that appears in the final equation. 9

Section 18. 1 Balancing Oxidation-Reduction Equations Half–Reactions § The overall reaction is split into two half–reactions, one involving oxidation and one involving reduction. 8 H+ + Mn. O 4– + 5 Fe 2+ Mn 2+ + 5 Fe 3+ + 4 H 2 O Reduction: 8 H+ + Mn. O 4– + 5 e– Mn 2+ + 4 H 2 O Oxidation: 5 Fe 2+ 5 Fe 3+ + 5 e– Copyright © Cengage Learning. All rights reserved 10

Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Acidic Solution 1. Write separate equations for the oxidation and reduction half–reactions. 2. For each half–reaction: A. Balance all the elements except H and O. B. Balance O using H 2 O. C. Balance H using H+. D. Balance the charge using electrons. Copyright © Cengage Learning. All rights reserved 11

Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Acidic Solution 3. If necessary, multiply one or both balanced half–reactions by an integer to equalize the number of electrons transferred in the two half–reactions. 4. Add the half–reactions, and cancel identical species. 5. Check that the elements and charges are balanced. Copyright © Cengage Learning. All rights reserved 12

Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Acidic Solution Copyright © Cengage Learning. All rights reserved 13

Section 18. 1 Balancing Oxidation-Reduction Equations Cr 2 O 72 -(aq) + SO 32 -(aq) Cr 3+(aq) + SO 42 -(aq) § How can we balance this equation? § First Steps: § Separate into half-reactions. § Balance elements except H and O. Copyright © Cengage Learning. All rights reserved 14

Section 18. 1 Balancing Oxidation-Reduction Equations Method of Half Reactions § Cr 2 O 72 -(aq) 2 Cr 3+(aq) § SO 32 -(aq) SO 42 -(aq) § How many electrons are involved in each half reaction? Copyright © Cengage Learning. All rights reserved 15

Section 18. 1 Balancing Oxidation-Reduction Equations Method of Half Reactions (continued) § 6 e- + Cr 2 O 72 -(aq) 2 Cr 3+(aq) § SO 32 -(aq) + SO 42 -(aq) + 2 e§ How can we balance the oxygen atoms? Copyright © Cengage Learning. All rights reserved 16

Section 18. 1 Balancing Oxidation-Reduction Equations Method of Half Reactions (continued) § 6 e- + Cr 2 O 72 -(aq) Cr 3+(aq) + 7 H 2 O § H 2 O +SO 32 -(aq) + SO 42 -(aq) + 2 e§ How can we balance the hydrogen atoms? Copyright © Cengage Learning. All rights reserved 17

Section 18. 1 Balancing Oxidation-Reduction Equations Method of Half Reactions (continued) § § This reaction occurs in an acidic solution. 14 H+ + 6 e- + Cr 2 O 72 - 2 Cr 3+ + 7 H 2 O +SO 32 - SO 42 - + 2 e- + 2 H+ How can we balance the electrons? Copyright © Cengage Learning. All rights reserved 18

Section 18. 1 Balancing Oxidation-Reduction Equations Method of Half Reactions (continued) § 14 H+ + 6 e- + Cr 2 O 72 - 2 Cr 3+ + 7 H 2 O § 3[H 2 O +SO 32 - SO 42 - + 2 e- + 2 H+] § Final Balanced Equation: Cr 2 O 72 - + 3 SO 32 - + 8 H+ 2 Cr 3+ + 3 SO 42 - + 4 H 2 O Copyright © Cengage Learning. All rights reserved 19

Section 18. 1 Balancing Oxidation-Reduction Equations EXERCISE! Balance the following oxidation–reduction reaction that occurs in acidic solution. Br–(aq) + Mn. O 4–(aq) Br 2(l)+ Mn 2+(aq) 10 Br–(aq) + 16 H+(aq) + 2 Mn. O 4–(aq) 5 Br 2(l)+ 2 Mn 2+(aq) + 8 H 2 O(l) Copyright © Cengage Learning. All rights reserved 20

Example 18. 1 Write a balanced ionic equation to represent the oxidation of iodide ion (I-) by permanganate ion ( ) in basic solution to yield molecular iodine (I 2) and manganese(IV) oxide (Mn. O 2). 21

Example 18. 1 Strategy We follow the preceding procedure for balancing redox equations. Note that the reaction takes place in a basic medium. Solution Step 1: The unbalanced equation is + I- Mn. O 2 + I 2 22

Example 18. 1 Step 2: The two half-reactions are -1 Oxidation: I- 0 I 2 +7 +4 Reduction: Mn. O 2 Step 3: We balance each half-reaction for number and type of atoms and charges. Oxidation half-reaction: We first balance the I atoms: 2 I- I 2 23

Example 18. 1 To balance charges, we add two electrons to the right-hand side of the equation: 2 I- I 2 + 2 e- Reduction half-reaction: To balance the O atoms, we add two H 2 O molecules on the right: Mn. O 2 + 2 H 2 O To balance the H atoms, we add four H+ ions on the left: + 4 H+ Mn. O 2 + 2 H 2 O 24

Example 18. 1 There are three net positive charges on the left, so we add three electrons to the same side to balance the charges: + 4 H+ + 3 e- Mn. O 2 + 2 H 2 O Step 4: We now add the oxidation and reduction half reactions to give the overall reaction. In order to equalize the number of electrons, we need to multiply the oxidation half-reaction by 3 and the reduction half-reaction by 2 as follows: 3(2 II 2 + 2 e-) 2( + 4 H+ + 3 e. Mn. O 2 + 2 H 2 O) ____________________ 6 I- + + 8 H+ + 6 e 3 I 2 + 2 Mn. O 2 + 4 H 2 O + 6 e- 25

Example 18. 1 The electrons on both sides cancel, and we are left with the balanced net ionic equation: 6 I- + 2 + 8 H+ 3 I 2 + 2 Mn. O 2 + 4 H 2 O This is the balanced equation in an acidic medium. However, because the reaction is carried out in a basic medium, for every H+ ion we need to add equal number of OH- ions to both sides of the equation: 6 I- + 2 + 8 H+ + 8 OH- 3 I 2 + 2 Mn. O 2 + 4 H 2 O + 8 OH 26

Example 18. 1 Finally, combining the H+ and OH- ions to form water, we obtain 6 I- + 2 + 4 H 2 O 3 I 2 + 2 Mn. O 2 + 8 OH- Step 5: A final check shows that the equation is balanced in terms of both atoms and charges. 27

Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Basic Solution 1. Use the half–reaction method as specified for acidic solutions to obtain the final balanced equation as if H+ ions were present. 2. To both sides of the equation obtained above, add a number of OH– ions that is equal to the number of H+ ions. (We want to eliminate H+ by forming H 2 O. ) Copyright © Cengage Learning. All rights reserved 28

Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Basic Solution 3. Form H 2 O on the side containing both H+ and OH– ions, and eliminate the number of H 2 O molecules that appear on both sides of the equation. 4. Check that elements and charges are balanced. Copyright © Cengage Learning. All rights reserved 29
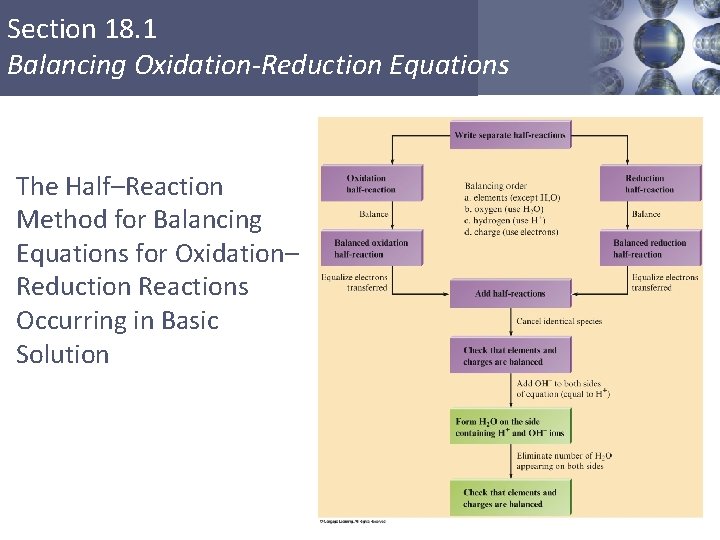
Section 18. 1 Balancing Oxidation-Reduction Equations The Half–Reaction Method for Balancing Equations for Oxidation– Reduction Reactions Occurring in Basic Solution Copyright © Cengage Learning. All rights reserved 30

The next several topics describe battery cells or voltaic cells (galvanic cells). An electrochemical cell is a system consisting of electrodes that dip into an electrolyte and in which a chemical reaction either uses or generates an electric current.

Section 18. 2 Galvanic Cells Galvanic Cell § Device in which chemical energy is changed to electrical energy. § Uses a spontaneous redox reaction to produce a current that can be used to do work. Copyright © Cengage Learning. All rights reserved 32
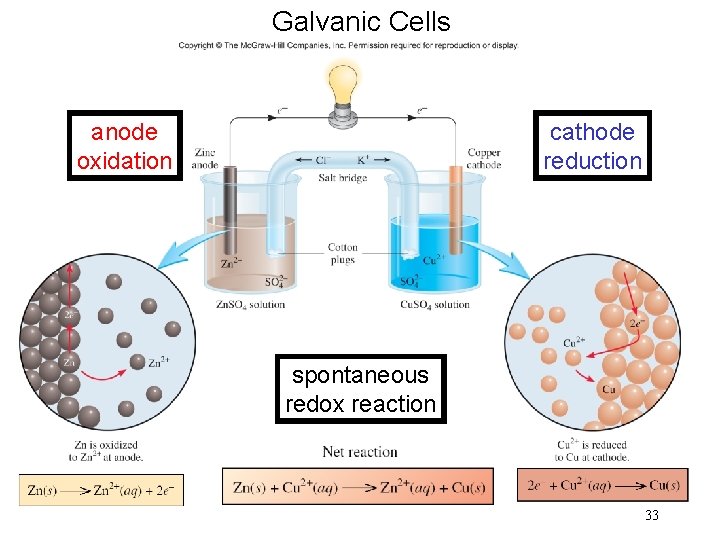
Galvanic Cells anode oxidation cathode reduction spontaneous redox reaction 33
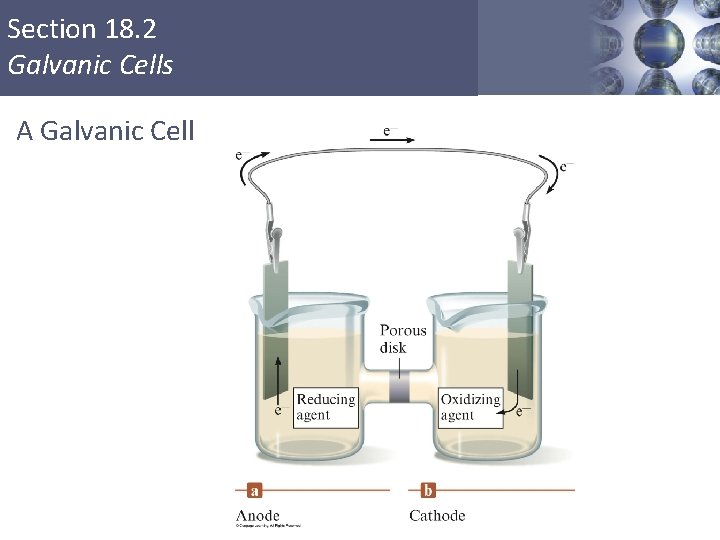
Section 18. 2 Galvanic Cells A Galvanic Cell Copyright © Cengage Learning. All rights reserved 34

Section 18. 2 Galvanic Cells Galvanic Cell § Oxidation occurs at the anode. § Reduction occurs at the cathode. § Salt bridge or porous disk – devices that allow ions to flow without extensive mixing of the solutions. § Salt bridge – contains a strong electrolyte held in a Jello–like matrix. § Porous disk – contains tiny passages that allow hindered flow of ions. Copyright © Cengage Learning. All rights reserved 35

Section 18. 2 Galvanic Cells Cell Potential § A galvanic cell consists of an oxidizing agent in one compartment that pulls electrons through a wire from a reducing agent in the other compartment. § The “pull”, or driving force, on the electrons is called the cell potential ( ), or the electromotive force (emf) of the cell. § Unit of electrical potential is the volt (V). Ø 1 joule of work per coulomb of charge transferred. Copyright © Cengage Learning. All rights reserved 36
Section 18. 2 Galvanic Cells Voltaic Cell: Cathode Reaction To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 37
Section 18. 2 Galvanic Cells Voltaic Cell: Anode Reaction To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 38

Section 18. 3 Standard Reduction Potentials Galvanic Cell § § All half-reactions are given as reduction processes in standard tables. § Table 18. 1 § 1 M, 1 atm, 25°C When a half-reaction is reversed, the sign of E° is reversed. When a half-reaction is multiplied by an integer, E° remains the same. A galvanic cell runs spontaneously in the direction that gives a positive value for E°cell. Copyright © Cengage Learning. All rights reserved 39

• E 0 is for the reaction as written • The more positive E 0 the greater the tendency for the substance to be reduced • The half-cell reactions are reversible • The sign of E 0 changes when the reaction is reversed • Changing the stoichiometric coefficients of a half-cell reaction does not change the value of E 0 40

Comparing Oxidizing Strengths The oxidizing agent is itself reduced and is the species on the left of the reduction half-reaction. Consequently, the strongest oxidizing agent is the product of the half-reaction with the largest (most positive) E° value.

Comparing Reducing Strengths The reducing agent is itself oxidized and is the species on the right of the reduction half-reaction. Consequently, the strongest reducing agent is the reactant in the half-reaction with the smallest (most negative) E° value.

Section 18. 3 Standard Reduction Potentials Example: Fe 3+(aq) + Cu(s) → Cu 2+(aq) + Fe 2+(aq) § § § Half-Reactions: § Fe 3+ + e– → Fe 2+ E° = 0. 77 V § Cu 2+ + 2 e– → Cu E° = 0. 34 V To balance the cell reaction and calculate the cell potential, we must reverse reaction 2. § Cu → Cu 2+ + 2 e– – E° = – 0. 34 V Each Cu atom produces two electrons but each Fe 3+ ion accepts only one electron, therefore reaction 1 must be multiplied by 2. § 2 Fe 3+ + 2 e– → 2 Fe 2+ E° = 0. 77 V Copyright © Cengage Learning. All rights reserved 43

Section 18. 3 Standard Reduction Potentials Overall Balanced Cell Reaction 2 Fe 3+ + 2 e– → 2 Fe 2+ E° = 0. 77 V (cathode) Cu → Cu 2+ + 2 e– – E° = – 0. 34 V (anode) § Balanced Cell Reaction: Cu + 2 Fe 3+ → Cu 2+ + 2 Fe 2+ § Cell Potential: E°cell = E°(cathode) – E°(anode) E°cell = 0. 77 V – 0. 34 V = 0. 43 V Copyright © Cengage Learning. All rights reserved 44

Section 18. 3 Standard Reduction Potentials CONCEPT CHECK! Order the following from strongest to weakest oxidizing agent and justify. Of those you cannot order, explain why. Fe Na F- Na+ Cl 2

Section 18. 3 Standard Reduction Potentials Line Notation Used to describe electrochemical cells. Anode components are listed on the left. Cathode components are listed on the right. Separated by double vertical lines which indicated salt bridge or porous disk. § The concentration of aqueous solutions should be specified in the notation when known. § Example: Mg(s)|Mg 2+(aq)||Al 3+(aq)|Al(s) § Mg → Mg 2+ + 2 e– (anode) § Al 3+ + 3 e– → Al (cathode) § § Copyright © Cengage Learning. All rights reserved 46
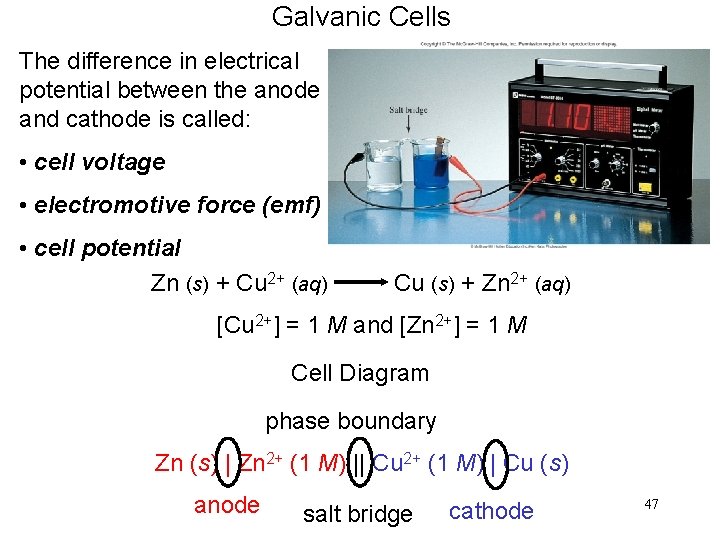
Galvanic Cells The difference in electrical potential between the anode and cathode is called: • cell voltage • electromotive force (emf) • cell potential Zn (s) + Cu 2+ (aq) Cu (s) + Zn 2+ (aq) [Cu 2+] = 1 M and [Zn 2+] = 1 M Cell Diagram phase boundary Zn (s) | Zn 2+ (1 M) || Cu 2+ (1 M) | Cu (s) anode salt bridge cathode 47

Section 18. 3 Standard Reduction Potentials Description of a Galvanic Cell § The cell potential (always positive for a galvanic cell where E°cell = E°(cathode) – E°(anode)) and the balanced cell reaction. § The direction of electron flow, obtained by inspecting the half–reactions and using the direction that gives a positive E°cell. Copyright © Cengage Learning. All rights reserved 48

Section 18. 3 Standard Reduction Potentials Description of a Galvanic Cell § Designation of the anode and cathode. § The nature of each electrode and the ions present in each compartment. A chemically inert conductor is required if none of the substances participating in the half–reaction is a conducting solid. Copyright © Cengage Learning. All rights reserved 49

When the half-reaction involves a gas, the electrode is an inert material such as platinum, Pt. It is included as a third substance in the half-cell. For example, the half-reaction of Cl 2 being reduced to Cl- is written as follows: Cl 2(g) | Cl-(aq) | Pt Because this is a reduction, the electrode appears on the far right. For the oxidation of H 2(g) to H+(aq), the notation is Pt | H 2(g) | H+(aq) In this case, the electrode appears on the far left.

Section 18. 3 Standard Reduction Potentials CONCEPT CHECK! Sketch a cell using the following solutions and electrodes. Include: § The potential of the cell § The direction of electron flow § Labels on the anode and the cathode Example 18. 4 a) Ag electrode in 1. 0 M Ag+(aq) and Fe 2+ electrode in 1. 0 M Fe 3+(aq) Copyright © Cengage Learning. All rights reserved 51

Section 18. 3 Standard Reduction Potentials CONCEPT CHECK! Sketch a cell using the following solutions and electrodes. Include: § The potential of the cell § The direction of electron flow § Labels on the anode and the cathode b) Zn electrode in 1. 0 M Zn 2+(aq) and Cu electrode in 1. 0 M Cu 2+(aq) Copyright © Cengage Learning. All rights reserved 52

Section 18. 3 Standard Reduction Potentials CONCEPT CHECK! Consider the cell from part b. What would happen to the potential if you increase the [Cu 2+]? Explain. The cell potential should increase. Copyright © Cengage Learning. All rights reserved 53

Just as work is required to pump water from one point to another, so work is required to move electrons. Water flows from areas of high pressure to areas of low pressure. Similarly, electrons flow from high electric potential to low electric potential. Electric potential can be thought of as electric pressure.

Section 18. 4 Cell Potential, Electrical Work, and Free Energy Work § Work is never the maximum possible if any current is flowing. § In any real, spontaneous process some energy is always wasted – the actual work realized is always less than the calculated maximum. Copyright © Cengage Learning. All rights reserved 55

Potential difference is the difference in electric potential (electrical pressure) between two points. Potential difference is measured in the SI unit volt (V). Electrical work = charge x potential difference The SI units for this are J=C×V

Section 18. 4 Cell Potential, Electrical Work, and Free Energy Maximum Cell Potential § Directly related to the free energy difference between the reactants and the products in the cell. § ΔG° = –n. FE° Ø F = 96, 485 C/mol e– Copyright © Cengage Learning. All rights reserved 57
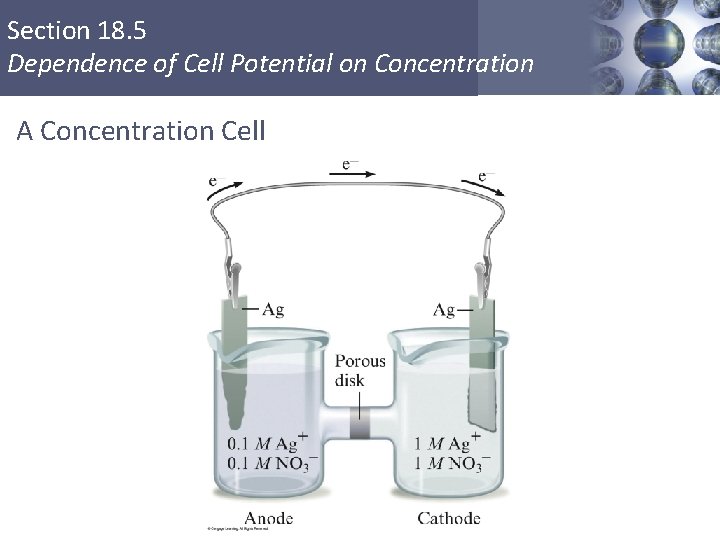
Section 18. 5 Dependence of Cell Potential on Concentration A Concentration Cell Copyright © Cengage Learning. All rights reserved 58

The Effect of Concentration on Cell Emf DG = DG 0 + RT ln Q DG = -n. FE DG 0 = -n. FE 0 -n. FE = -n. FE 0 + RT ln Q Nernst equation E = E 0 - RT ln Q n. F At 298 E = E 0 - 0. 0257 V ln Q n E = E 0 - 0. 0592 V log Q n 19. 5

Section 18. 5 Dependence of Cell Potential on Concentration Nernst Equation § The relationship between cell potential and concentrations of cell components § At 25°C: or (at equilibrium) Copyright © Cengage Learning. All rights reserved 60

Section 18. 5 Dependence of Cell Potential on Concentration CONCEPT CHECK! Explain the difference between E and E°. When is E equal to zero? When the cell is in equilibrium ("dead" battery). When is E° equal to zero? E is equal to zero for a concentration cell. Copyright © Cengage Learning. All rights reserved 61

Section 18. 5 Dependence of Cell Potential on Concentration EXERCISE! A concentration cell is constructed using two nickel electrodes with Ni 2+ concentrations of 1. 0 M and 1. 00 × 10 -4 M in the two half-cells. Calculate the potential of this cell at 25°C. 0. 118 V Copyright © Cengage Learning. All rights reserved 62

Section 18. 5 Dependence of Cell Potential on Concentration CONCEPT CHECK! You make a galvanic cell at 25°C containing: § A nickel electrode in 1. 0 M Ni 2+(aq) § A silver electrode in 1. 0 M Ag+(aq) Sketch this cell, labeling the anode and cathode, showing the direction of the electron flow, and calculate the cell potential. 1. 03 V Copyright © Cengage Learning. All rights reserved 63
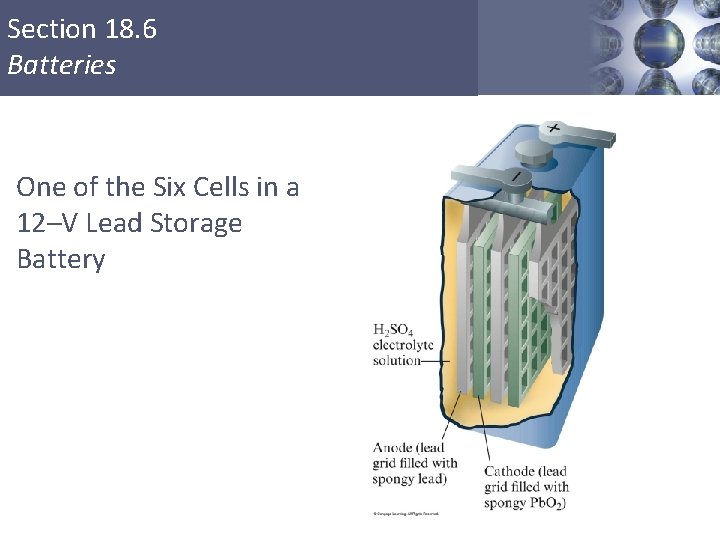
Section 18. 6 Batteries One of the Six Cells in a 12–V Lead Storage Battery Copyright © Cengage Learning. All rights reserved 64

Lead Storage Cell The electrodes are lead alloy grids: one is packed with a spongy lead to form the anode, and the other is packed with lead dioxide to form the cathode. Both electrodes are in an aqueous solution of H 2 SO 4. Anode: Cathode: Pb(s) + HSO 4 -(aq) Pb. SO 4(s) + H+(aq) + 2 e. Pb. O 2(s) + 3 H+(aq) + HSO 4 -(aq) + 2 e Pb. SO 4(s) + 2 H 2 O(l) Unlike dry cells, after discharge, lead storage cells can be recharged. Copyright © Cengage Learning. All rights reserved. 19 | 65

Each of the six cells in the lead storage cell generates 2 V, yielding 12 V. During discharge, white Pb. SO 4(s) coats each electrode. To recharge the cell, an external current is used, reversing the previous reactions. Some water decomposes into hydrogen and oxygen gas, so more water may need to be added. Newer batteries use electrodes with calcium in the lead, which resists decomposition by water. These versions are maintenance free. Copyright © Cengage Learning. All rights reserved. 19 | 66
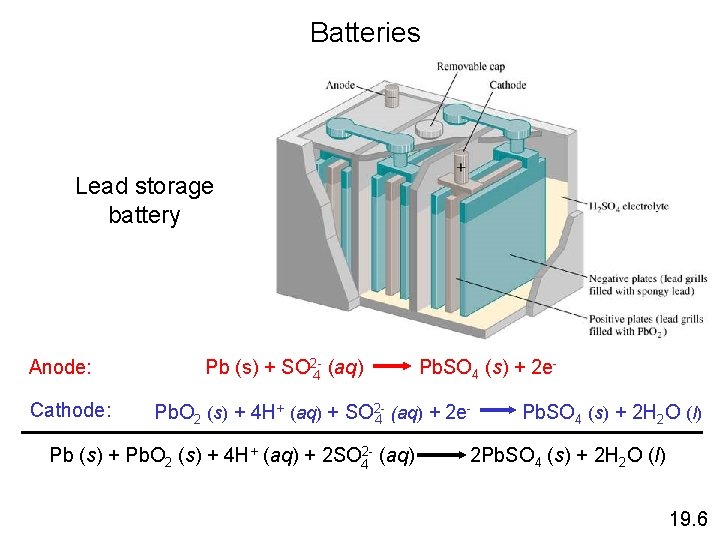
Batteries Lead storage battery Anode: Cathode: Pb (s) + SO 2 -4 (aq) Pb. SO 4 (s) + 2 e- Pb. O 2 (s) + 4 H+ (aq) + SO 24 (aq) + 2 e Pb (s) + Pb. O 2 (s) + 4 H+ (aq) + 2 SO 42 - (aq) Pb. SO 4 (s) + 2 H 2 O (l) 2 Pb. SO 4 (s) + 2 H 2 O (l) 19. 6
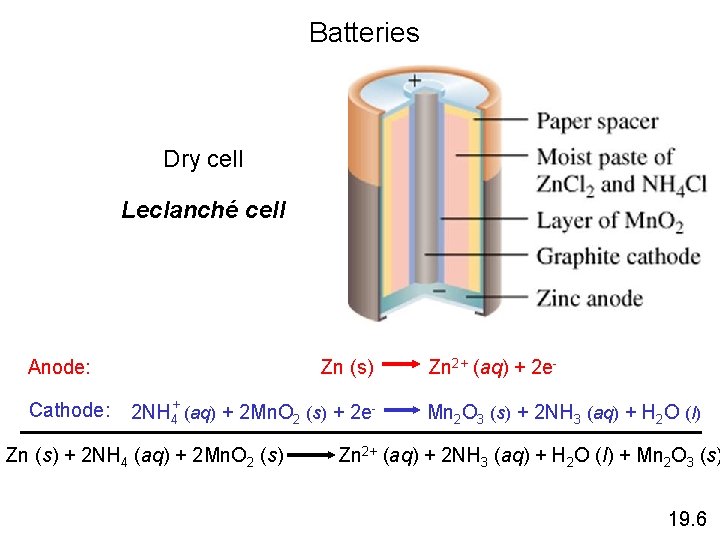
Batteries Dry cell Leclanché cell Zn (s) Anode: Cathode: 2 NH 4+ (aq) + 2 Mn. O 2 (s) + 2 e- Zn (s) + 2 NH 4 (aq) + 2 Mn. O 2 (s) Zn 2+ (aq) + 2 e. Mn 2 O 3 (s) + 2 NH 3 (aq) + H 2 O (l) Zn 2+ (aq) + 2 NH 3 (aq) + H 2 O (l) + Mn 2 O 3 (s) 19. 6

Zinc–Carbon Dry Cell: Leclanché Anode: Zn(s) Zn 2+(aq) + 2 e. Cathode: 2 NH 4+(aq) + 2 Mn. O 2(s) + 2 e Mn 2 O 3(s) + H 2 O(l) + 2 NH 3(aq) The initial voltage is about 1. 5 V, but decreases and deteriorates rapidly in cold weather.

Section 18. 6 Batteries A Common Dry Cell Battery
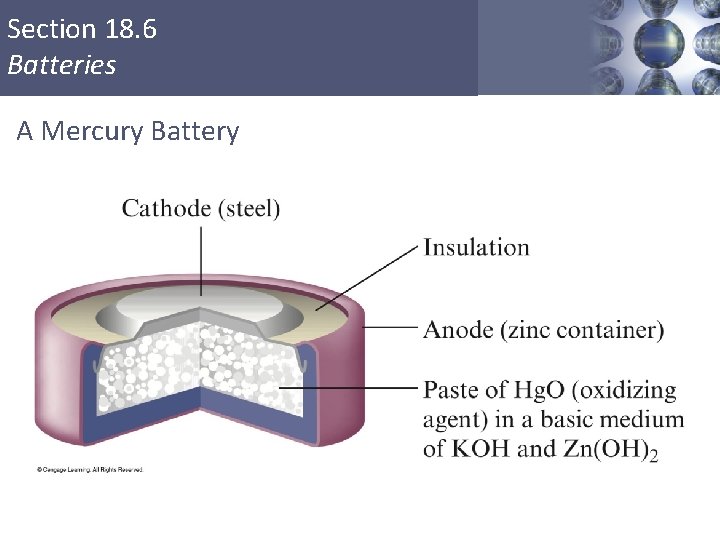
Section 18. 6 Batteries A Mercury Battery Copyright © Cengage Learning. All rights reserved 71
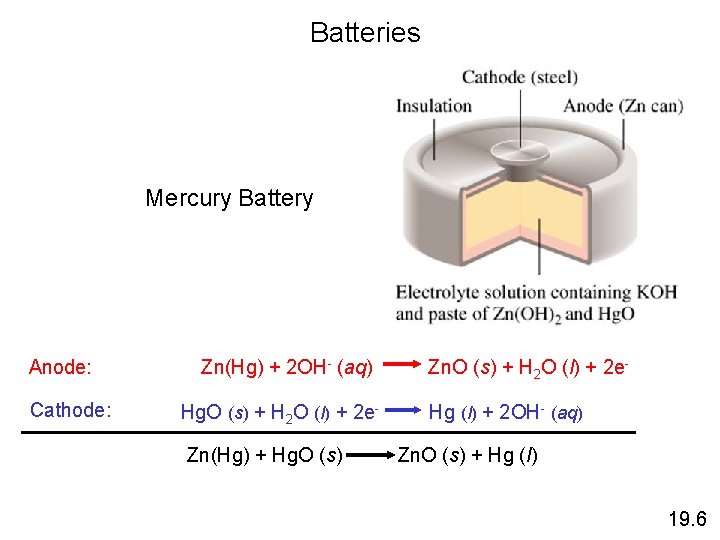
Batteries Mercury Battery Anode: Cathode: Zn(Hg) + 2 OH- (aq) Hg. O (s) + H 2 O (l) + 2 e. Zn(Hg) + Hg. O (s) Zn. O (s) + H 2 O (l) + 2 e. Hg (l) + 2 OH- (aq) Zn. O (s) + Hg (l) 19. 6
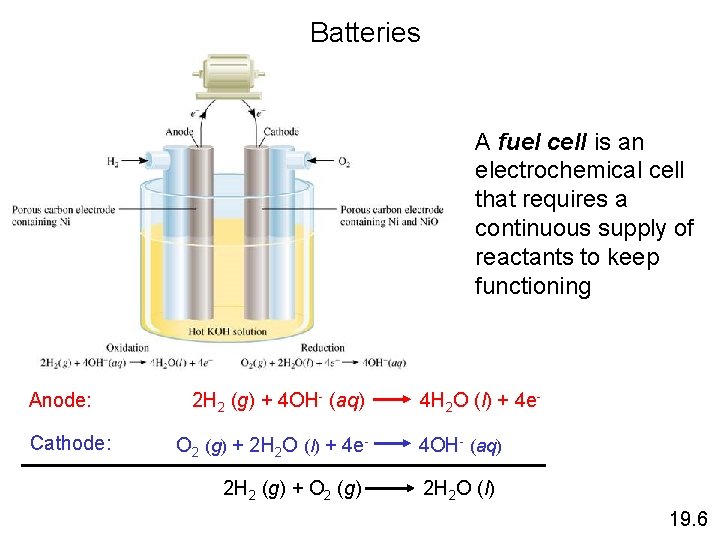
Batteries A fuel cell is an electrochemical cell that requires a continuous supply of reactants to keep functioning Anode: Cathode: 2 H 2 (g) + 4 OH- (aq) O 2 (g) + 2 H 2 O (l) + 4 e 2 H 2 (g) + O 2 (g) 4 H 2 O (l) + 4 e 4 OH- (aq) 2 H 2 O (l) 19. 6
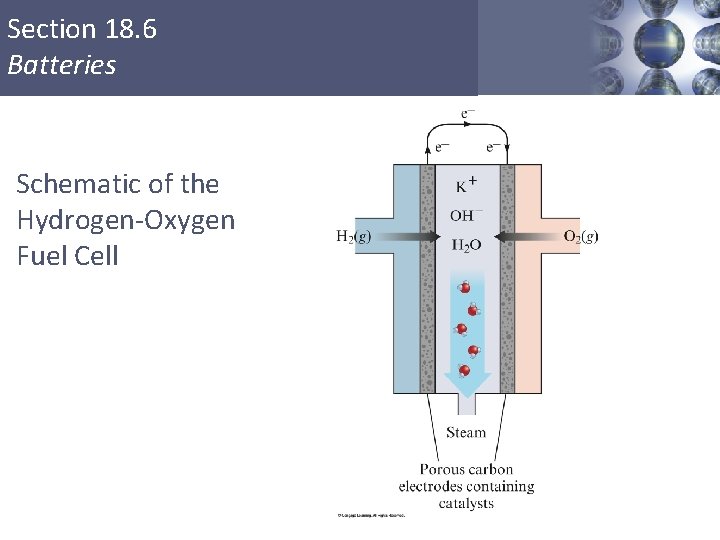
Section 18. 6 Batteries Schematic of the Hydrogen-Oxygen Fuel Cell

Section 18. 7 Corrosion § Process of returning metals to their natural state – the ores from which they were originally obtained. § Involves oxidation of the metal. Copyright © Cengage Learning. All rights reserved 75

Corrosion Control: Cathodic Protection Voltaic cells can be used to control corrosion of underground pipelines and tanks. Rusting occurs when water comes in contact with iron. The edge of the water drop, when exposed to air, becomes one pole of a voltaic cell where oxygen is reduced to hydroxide. Anode: Cathode: Fe(s) Fe 2+(aq) + 2 e. O 2(g) + 2 H 2 O(l) + 4 e- 4 OH-(aq) Copyright © Cengage Learning. All rights reserved. 19 | 76
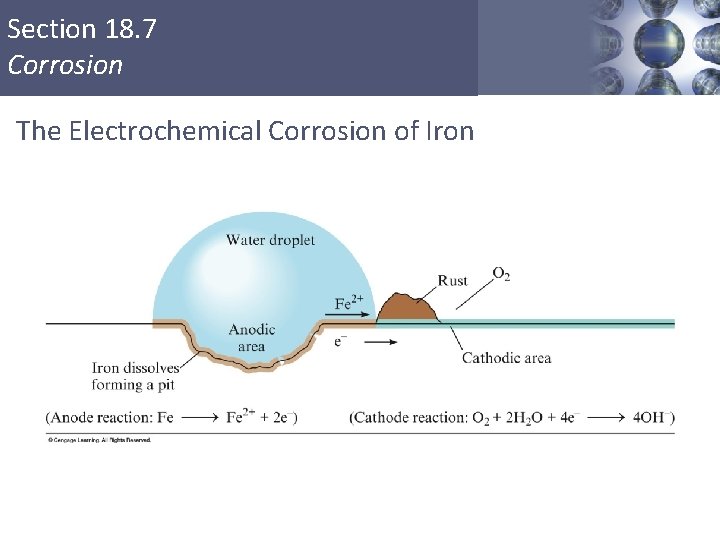
Section 18. 7 Corrosion The Electrochemical Corrosion of Iron Copyright © Cengage Learning. All rights reserved 77

Section 18. 7 Corrosion Prevention § Application of a coating (like paint or metal plating) § Galvanizing § Alloying § Cathodic Protection § Protects steel in buried fuel tanks and pipelines. Copyright © Cengage Learning. All rights reserved 78

When the buried metal is connected to a more active metal such as magnesium, the magnesium becomes the anode and the iron becomes the cathode. The iron is, therefore, protected from oxidation. This phenomenon is called cathodic protection. Copyright © Cengage Learning. All rights reserved. 19 | 79
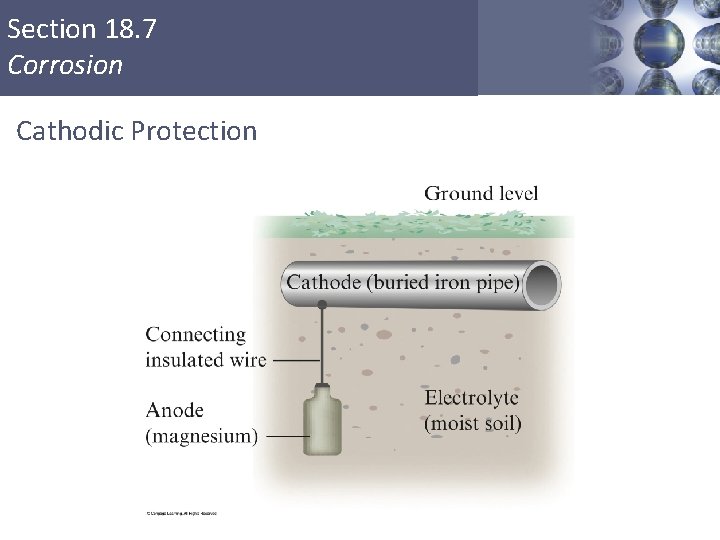
Section 18. 7 Corrosion Cathodic Protection Copyright © Cengage Learning. All rights reserved 80
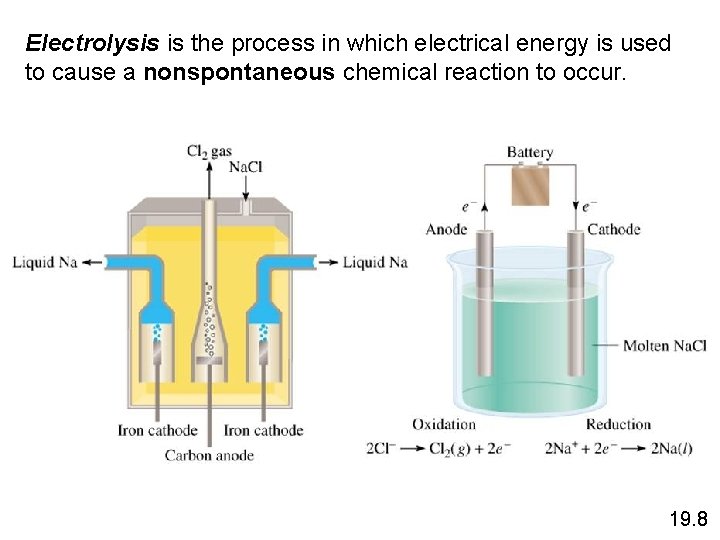
Electrolysis is the process in which electrical energy is used to cause a nonspontaneous chemical reaction to occur. 19. 8

Section 18. 8 Electrolysis § Forcing a current through a cell to produce a chemical change for which the cell potential is negative. Copyright © Cengage Learning. All rights reserved 82

Stoichiometry of Electrolysis In the 1830 s, Michael Faraday showed that the total charge that flows in a circuit is related to the amount of substance released at the electrodes. One faraday of charge is the charge on one mole of electrons and is equal to 96, 485 coulombs. Copyright © Cengage Learning. All rights reserved. 19 | 83

Section 18. 8 Electrolysis Stoichiometry of Electrolysis § How much chemical change occurs with the flow of a given current for a specified time? current and time � quantity of charge � moles of electrons � moles of analyte � grams of analyte Copyright © Cengage Learning. All rights reserved 84

Section 18. 8 Electrolysis Stoichiometry of Electrolysis § current and time � quantity of charge Coulombs of charge = amps (C/s) × seconds (s) § quantity of charge � moles of electrons Copyright © Cengage Learning. All rights reserved 85

For quantitative considerations, we also need to know the magnitude of the current and the time it has flowed. Electric charge = current × time Coulombs = amperes × seconds The ampere, A, is the base unit of current. The coulomb, C, is equal to an ampere-second. Copyright © Cengage Learning. All rights reserved. 19 | 86

What electric charge is required to plate a piece of automobile molding with 1. 00 g of chromium metal using a chromium(III) ion solution? If the electrolysis current is 2. 00 A, how long does the plating take?

First, we convert mass to moles Cr; then to moles e-; then, using current, to seconds. = 2. 78 × 103 s = 46. 4 min

A solution of nickel salt is electrolyzed to nickel metal by a current of 2. 43 A. If this current flows for 10. 0 min, how many coulombs is this? How much nickel metal is deposited in the electrolysis?

We first find the charge: = 1458 C Given the half-reaction: Ni 2+(aq) + 2 e- Ni(s) = 0. 443 g Ni

Section 18. 8 Electrolysis CONCEPT CHECK! An unknown metal (M) is electrolyzed. It took 52. 8 sec for a current of 2. 00 amp to plate 0. 0719 g of the metal from a solution containing M(NO 3)3. What is the metal? gold (Au) Copyright © Cengage Learning. All rights reserved 91

Section 18. 8 Electrolysis CONCEPT CHECK! Consider a solution containing 0. 10 M of each of the following: Pb 2+, Cu 2+, Sn 2+, Ni 2+, and Zn 2+. Predict the order in which the metals plate out as the voltage is turned up from zero. Cu 2+, Pb 2+, Sn 2+, Ni 2+, Zn 2+ Do the metals form on the cathode or the anode? Explain. Copyright © Cengage Learning. All rights reserved 92

Section 18. 9 Commercial Electrolytic Processes § § § Production of aluminum Purification of metals Metal plating Electrolysis of sodium chloride Production of chlorine and sodium hydroxide Copyright © Cengage Learning. All rights reserved 93
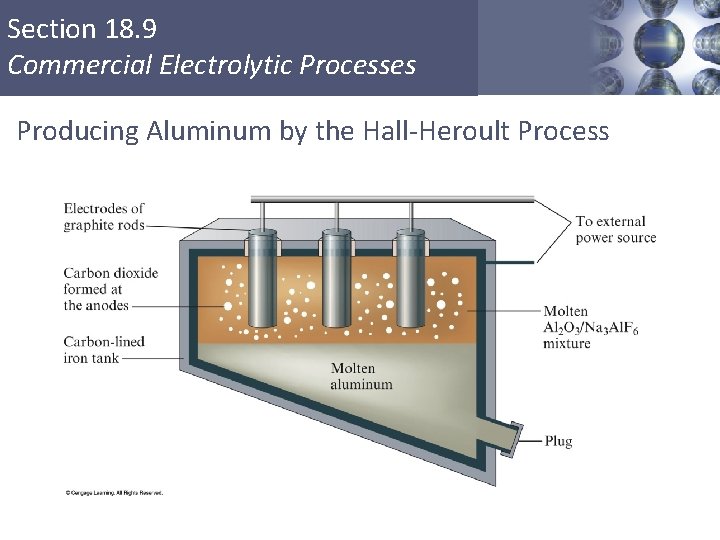
Section 18. 9 Commercial Electrolytic Processes Producing Aluminum by the Hall-Heroult Process Copyright © Cengage Learning. All rights reserved 94

Section 18. 9 Commercial Electrolytic Processes Electroplating a Spoon Copyright © Cengage Learning. All rights reserved 95

Downs Cell A Downs cell is an electrolytic cell used to obtain sodium metal by electrolysis of sodium chloride. The products must be kept separated or they would react. Anode: Cathode: Cl-(l) ½Cl 2(g) + e. Na+(l) + e- Na(l) Copyright © Cengage Learning. All rights reserved. 19 | 96
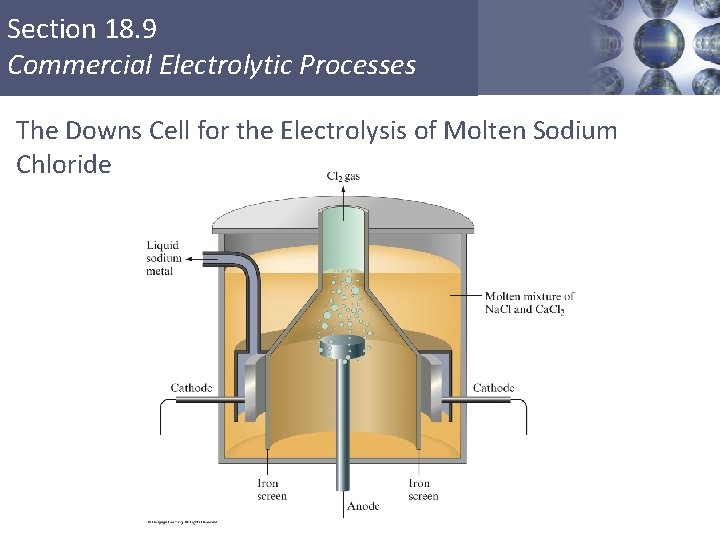
Section 18. 9 Commercial Electrolytic Processes The Downs Cell for the Electrolysis of Molten Sodium Chloride Copyright © Cengage Learning. All rights reserved 97
- Slides: 97