Chapter 18 Electrochemistry GOALS Balancing redox reactions Voltaic

Chapter 18 Electrochemistry

GOALS Balancing redox reactions Voltaic cells Electrochemical potentials Electrolysis …& the calculations!! Review: oxidation states oxidation/reduction oxidizing/reducing agent 2 ch. 17

Why Study Electrochemistry? • • • Batteries Corrosion Industrial production of chemicals such as Cl 2, Na. OH, F 2 and Al • Biological redox reactions The heme group

Electron Transfer Reactions • Electron transfer reactions are oxidation-reduction or redox reactions (i. e. changes in oxidation states). • Redox reactions can result in the generation of an electric current (battery), or, may be caused by applying an electric current (electroplating). • Therefore, this field of chemistry is often called ELECTROCHEMISTRY.

ELECTRON TRANSFER REACTIONS 0 0 1+ 1 - 2 Na(s) + Cl 2(g) 2 Na. Cl(s) 1+ 0 2 HCl(aq) + Zn(s) Zn. Cl 2(aq) + H 2(g) 0 0 Cu(s) + 2 Ag+(aq) Cu 2+(aq) + 2 Ag(s) 0 0 3+ 2 - 2 Fe(s) + x. H 2 O(l) + 1½O 2(g) Fe 2 O 3. x. H 2 O(s) 4+ 2 - 0 0 CO 2(g) + H 2 O(l) + energy (CH 2 O)n + O 2(g)

Review of Terminology for Redox Reactions • OXIDATION—loss of electron(s) by a species; increase in oxidation number ; e- to the right of arrow. Na+ + e- • REDUCTION—gain of electron(s); decrease in oxidation number; e- to left of arrow. ½Cl 2(g) + e- Cl- • OXIDIZING AGENT—electron acceptor; it is reduced: ½ Cl 2(g) + e- Cl- • REDUCING AGENT—electron donor; it is oxidized Na Na+ + e-

Electrochemical Cells • Apparatus for generating an electric current through the use of a product favored reaction (spontaneous): voltaic or galvanic cell. • An electrolytic cell is used to carry out electrolysis (an electric current is used to bring about a nonspontaneous chemical reaction). Batteries are voltaic cells

Electrochemistry Alessandro Volta, 1745 -1827, Italian scientist and inventor. Luigi Galvani, 1737 -1798, Italian scientist and inventor.

Balancing Equations for Redox Reactions Some redox reactions have equations that must be balanced by special techniques. Mn. O 4 -(aq) + 5 Fe 2+(aq) + 8 H+(aq) Fe = +2 Mn = +7 Mn 2+ (aq) + 5 Fe 3+(aq) + 4 H 2 O(liq) Mn = +2 Fe = +3

Rules for Assigning Oxidation States • rules are in order of priority 1. free elements have an oxidation state = 0 ü Na = 0 and Cl 2 = 0 in 2 Na(s) + Cl 2(g) 2. monatomic ions have an oxidation state equal to their charge ü Na = +1 and Cl = -1 in Na. Cl 3. (a) the sum of the oxidation states of all the atoms in a compound is 0 ü Na = +1 and Cl = -1 in Na. Cl, (+1) + (-1) = 0 10

Rules for Assigning Oxidation States 3. (b) the sum of the oxidation states of all the atoms in a polyatomic ion equals the charge on the ion ü N = +5 and O = -2 in NO 3–, (+5) + 3(-2) = -1 4. (a) Group I metals have an oxidation state of +1 in all their compounds ü Na = +1 in Na. Cl (b) Group II metals have an oxidation state of +2 in all their compounds ü Mg = +2 in Mg. Cl 2 11
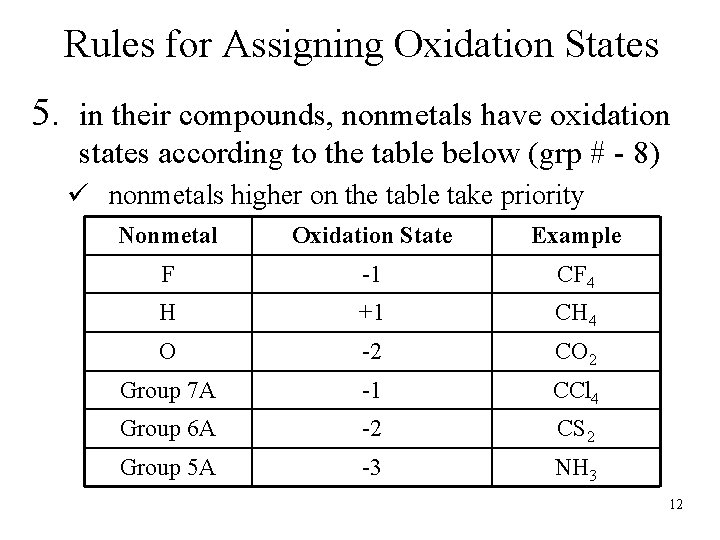
Rules for Assigning Oxidation States 5. in their compounds, nonmetals have oxidation states according to the table below (grp # - 8) ü nonmetals higher on the table take priority Nonmetal Oxidation State Example F -1 CF 4 H +1 CH 4 O -2 CO 2 Group 7 A -1 CCl 4 Group 6 A -2 CS 2 Group 5 A -3 NH 3 12
Balancing Equations Cu + Ag+ give Cu 2+ + Ag

Balancing Equations Step 1: Divide the reaction into halfreactions, one for oxidation and the other for reduction. Ox Cu 2+ Red Ag+ Ag Step 2: Balance each for mass. Already done in this case. Step 3: Balance each half-reaction for charge by adding electrons. Ox Cu 2+ + 2 e. Red Ag+ + e- Ag

Balancing Equations Step 4: Multiply each half-reaction by a factor so that the reducing agent supplies as many electrons as the oxidizing agent requires. Reducing agent Cu 2+ + 2 e. Oxidizing agent 2 Ag+ + 2 e- 2 Ag Step 5: Add half-reactions to give the overall equation. Cu + 2 Ag+ Cu 2+ + 2 Ag The equation is now balanced for both charge and mass (the 2 e- of the left are cancelled out with those on the right).

Reduction of + VO 2 with Zn

Balancing Equations Balance the following in acid solution— VO 2+ + Zn 2+ Step 1: Write the half-reactions Ox Zn 2+ Red VO 2+ Step 2: Balance each half-reaction for mass. Ox Zn 2+ + 2 e. Red VO 2+ + e- VO 2+ excess of 2+ on the right Zn lost two electrons. They are written on the right.

Balancing Equations Step 3: Balance half-reactions for charge Reaction is acidic, then we can use H+. Ox Zn 2+ + 2 e. Red e- + 2 H+ + VO 2+ + H 2 O Step 4: Multiply by an appropriate factor. Ox Zn 2+ + 2 e. Red 2 e- + 4 H+ + 2 VO 2+ + 2 H 2 O Step 5: Add balanced half-reactions. Zn + 4 H+ + 2 VO 2+ Zn 2+ + 2 VO 2+ + 2 H 2 O

Tips on Balancing Equations • Never add O 2, O atoms, or O 2 to balance oxygen. Balance O with OH- or H 2 O. • Never add H 2 or H atoms to balance hydrogen. Balance H with H+/H 2 O in acid or OH-/H 2 O in base.

Tips on Balancing Equations {Equations that include oxoanions like SO 42 -, NO 3 -, Cl. O- , Cr. O 42 -, and Mn. O 4 -, also fall into this category}. Be sure to write the correct charges on all the ions. • Check your work at the end to make sure mass and charge are balanced. • PRACTICE!!!!!!

More Practice - Balance the equations below! I (aq) + Mn. O 4 (aq) I 2(aq) + Mn. O 2(s) in basic solution An alkaline (basic) solution of hypochlorite ions reacts with solid chromium(III) hydroxide to produce chromate and chloride ions. Cl. O 3 - + Cl- Cl 2 (in acid) Cr 2 O 72 - + I- IO 3 - + Cr 3+ (in acid) Mn. O 4 - + H 2 SO 3 SO 42 - + Mn 2+ (in acid) Cr(OH)4 - + H 2 O 2 Cr. O 42 - + H 2 O (in basic soln) Zn + NO 3 - Zn(OH)4 - + NH 3 (in basic soln) 21
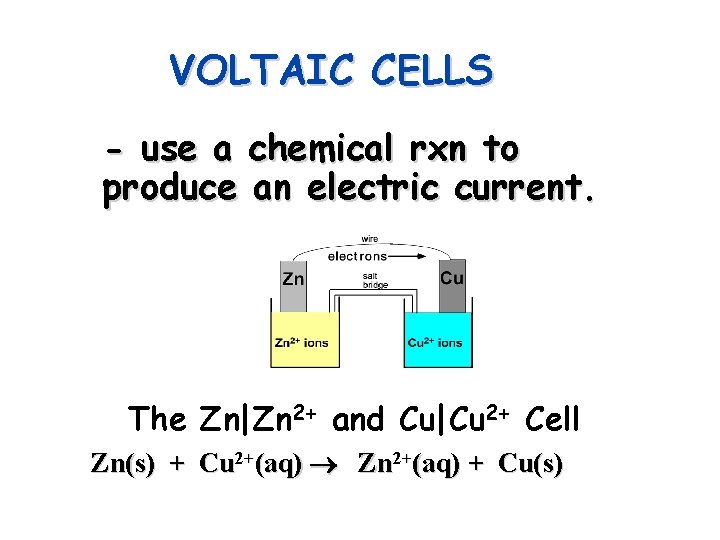
VOLTAIC CELLS - use a chemical rxn to produce an electric current. The Zn|Zn 2+ and Cu|Cu 2+ Cell Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s)
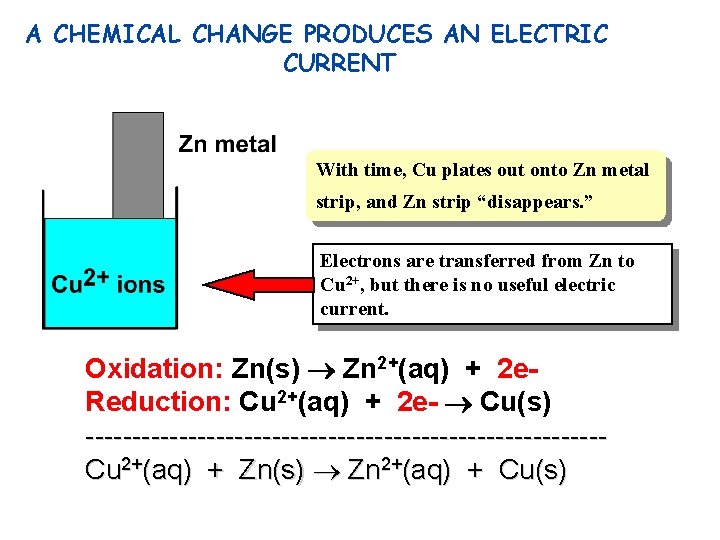
A CHEMICAL CHANGE PRODUCES AN ELECTRIC CURRENT With time, Cu plates out onto Zn metal strip, and Zn strip “disappears. ” Electrons are transferred from Zn to Cu 2+, but there is no useful electric current. Oxidation: Zn(s) Zn 2+(aq) + 2 e. Reduction: Cu 2+(aq) + 2 e- Cu(s) ----------------------------Cu 2+(aq) + Zn(s) Zn 2+(aq) + Cu(s)
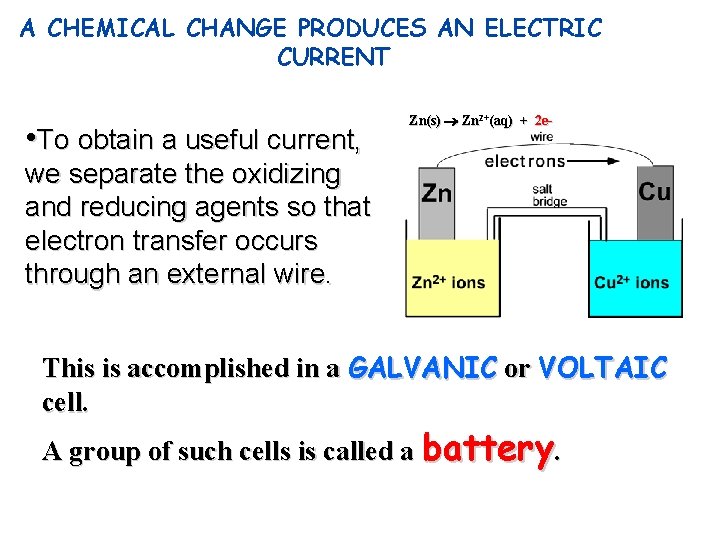
A CHEMICAL CHANGE PRODUCES AN ELECTRIC CURRENT • To obtain a useful current, Zn(s) Zn 2+(aq) + 2 e- we separate the oxidizing and reducing agents so that electron transfer occurs through an external wire. This is accomplished in a GALVANIC or VOLTAIC cell. A group of such cells is called a battery.
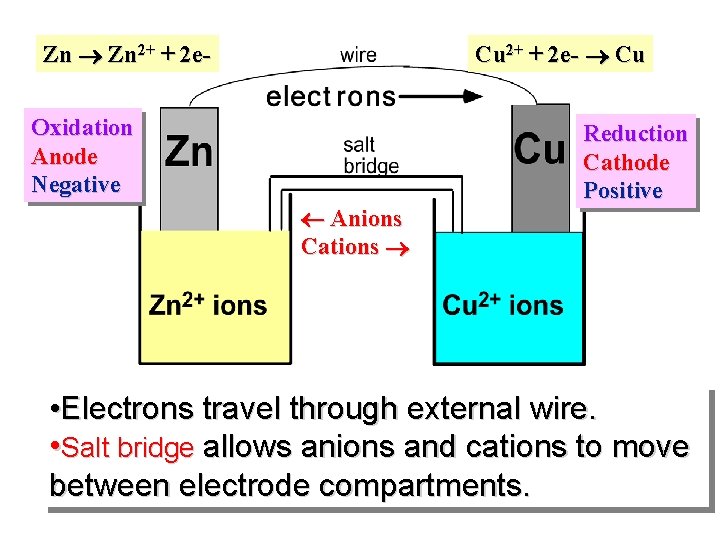
Zn Zn 2+ + 2 e- Cu 2+ + 2 e- Cu Oxidation Anode Negative Anions Cations Reduction Cathode Positive • Electrons travel through external wire. • Salt bridge allows anions and cations to move between electrode compartments.
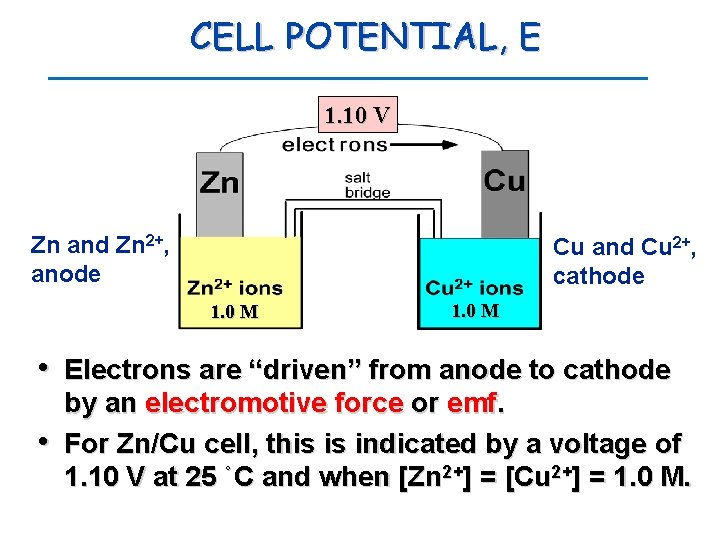
CELL POTENTIAL, E 1. 10 V Zn and Zn 2+, anode Cu and Cu 2+, cathode 1. 0 M • Electrons are “driven” from anode to cathode • by an electromotive force or emf. For Zn/Cu cell, this is indicated by a voltage of 1. 10 V at 25 ˚C and when [Zn 2+] = [Cu 2+] = 1. 0 M.

Need Calculate Cell Voltage • Balanced half-reactions can be added together to get the overall, balanced equation. Zn(s) Zn 2+(aq) + 2 e. Cu 2+(aq) + 2 e- Cu(s) ----------------------Cu 2+(aq) + Zn(s) Zn 2+(aq) + Cu(s) If we know Eo for each half-reaction, we add them to get Eo for overall reaction. -need Eo for Zn & Cu half-cells

Standard Reduction Potential • We can measure ALL other half-cell potentials relative to another half-reaction. • We select as a standard half-reaction, the reduction of H+ to H 2 under standard conditions (1 atm, 1 M @ 25 o. C) and which we assign a potential difference = 0 V üStandard Hydrogen Electrode, SHE 28
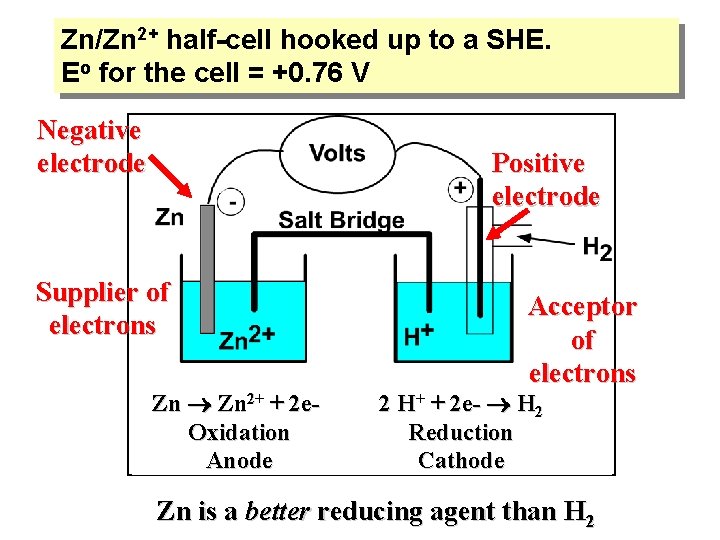
Zn/Zn 2+ half-cell hooked up to a SHE. Eo for the cell = +0. 76 V Negative electrode Positive electrode Supplier of electrons Zn 2+ + 2 e. Oxidation Anode Acceptor of electrons 2 H+ + 2 e- H 2 Reduction Cathode Zn is a better reducing agent than H 2
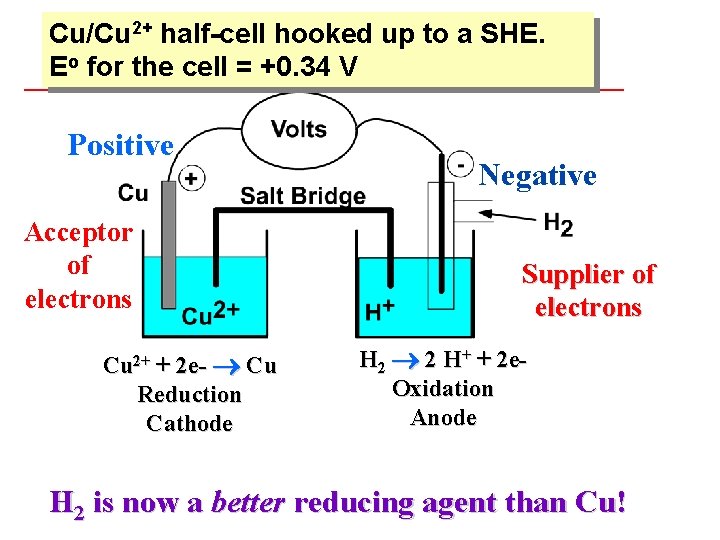
Cu/Cu 2+ half-cell hooked up to a SHE. Eo for the cell = +0. 34 V Positive Acceptor of electrons Negative Supplier of electrons + 2 e- Cu Reduction Cathode Cu 2+ H 2 2 H+ + 2 e. Oxidation Anode H 2 is now a better reducing agent than Cu!
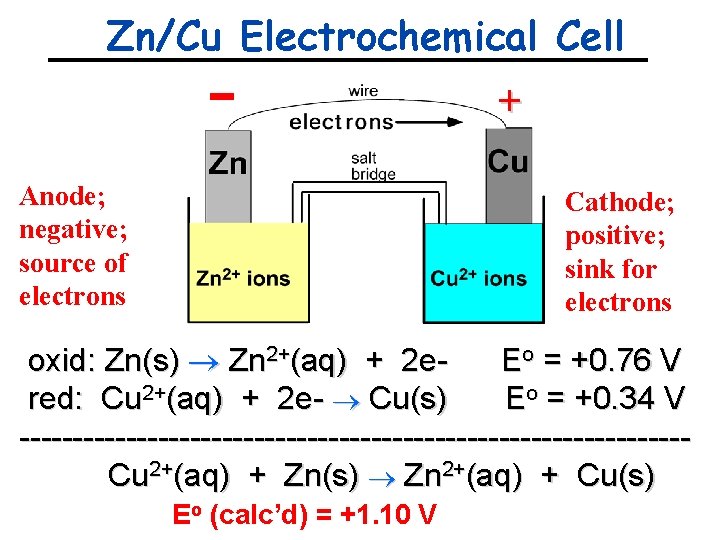
Zn/Cu Electrochemical Cell + Anode; negative; source of electrons Cathode; positive; sink for electrons oxid: Zn(s) Zn 2+(aq) + 2 e. Eo = +0. 76 V red: Cu 2+(aq) + 2 e- Cu(s) Eo = +0. 34 V -------------------------------Cu 2+(aq) + Zn(s) Zn 2+(aq) + Cu(s) Eo (calc’d) = +1. 10 V

Uses of Eo Values Organize half-reactions by relative ability to act as oxidizing/reducing agents. Half-rxns are written as reduction rxns!! Cu 2+(aq) + 2 e- Cu(s) Zn 2+(aq) + 2 e- Zn(s) Eo = +0. 34 V Eo = – 0. 76 V When a reaction is reversed, the sign of E˚ is reversed!
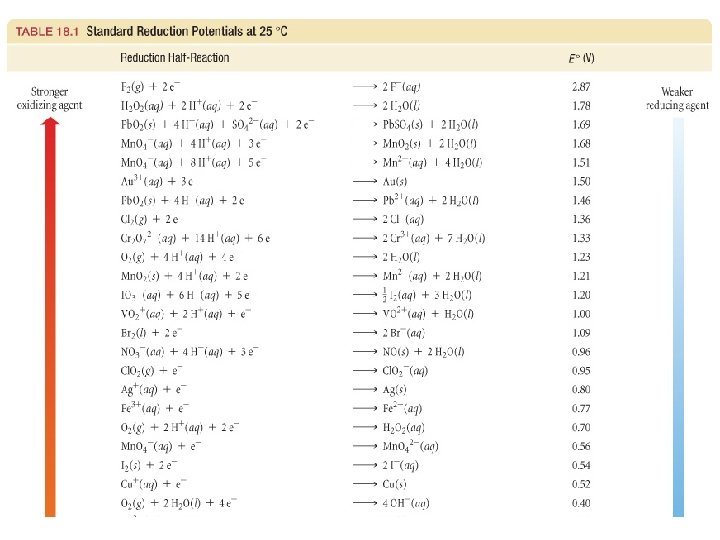
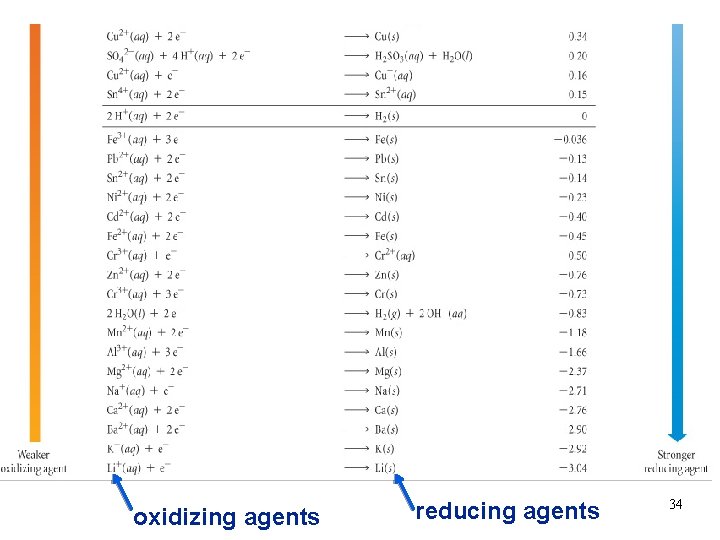
oxidizing agents reducing agents 34

Using Standard Potentials, o E Which is the best oxidizing agent: O 2 (1. 23 V); H 2 O 2 (1. 77 V) or Cl 2 (1. 36 V)? H 2 O 2 (1. 77 V) Which is the best reducing agent: Hg (+0. 79 V), Al (-1. 66 V), or Sn (-0. 14 V)? Al (-1. 66 V)

Using Standard Potentials, Eo Which substance is the best oxidizing agent? Cr 2 O 72 - + 6 e- + 14 H+ 2 Cr 3+ + 7 H 2 O (+1. 33 V) O 2 + 4 e- + 4 H+ 2 H 2 O (+1. 23 V) Fe 3+ + e- Fe 2+ (+0. 77 V) Cr 2 O 72 Which element/ion is the best reducing agent? Fe 3+ + e- Fe 2+ (+0. 77 V) I 2 + 2 e- 2 I(+0. 54 V) Sn 4+ + 2 e- Sn 2+ (+0. 15 V) Sn 2+

Standard Redox Potentials, Eo Any substance on the right will reduce any substance HIGHER than it on the LEFT. • Zn can reduce H+ and Cu 2+. • H 2 can reduce Cu 2+ but not Zn 2+ • Cu cannot reduce H+ or Zn 2+.

Standard Redox Potentials, Eo Ox. agent Cu 2+ + 2 e- --> Cu +0. 34 2 H+ + 2 e- --> H 2 0. 00 Zn 2+ + 2 e- --> Zn -0. 76 Red. agent Any substance on the right will reduce any substance higher than it on the left. Northwest-southeast rule: product-favored reactions occur between • reducing agent at southeast corner (ANODE) & • oxidizing agent at northwest corner (CATHODE)

Standard Redox Potentials, Eo Ox. agent Cu 2+ + 2 e- --> Cu +0. 34 V Ni 2++ 2 e- --> Ni -0. 25 V Zn 2+ + 2 e- --> Zn -0. 76 V Red. agent Zn will reduce Ni 2+, Cu 2+; Ni will reduce Cu 2+. Northwest-southeast rule: product-favored reactions occur between • reducing agent at southeast corner (ANODE) & • oxidizing agent at northwest corner (CATHODE)

Using Standard Potentials, Eo • • • In which direction do the following reactions go? Cu(s) + 2 Ag+(aq) Cu 2+(aq) + 2 Ag(s) +0. 46 V Cu 2+(aq) + Zn(s) Cu(s) + Zn 2+(aq) +1. 10 V üGo to the right as written • Fe 2+(aq) + Cd(s) Fe(s) + Cd 2+(aq) -0. 04 V üGoes LEFT, opposite to direction written • What is Eonet for this reverse reaction? +0. 04 V
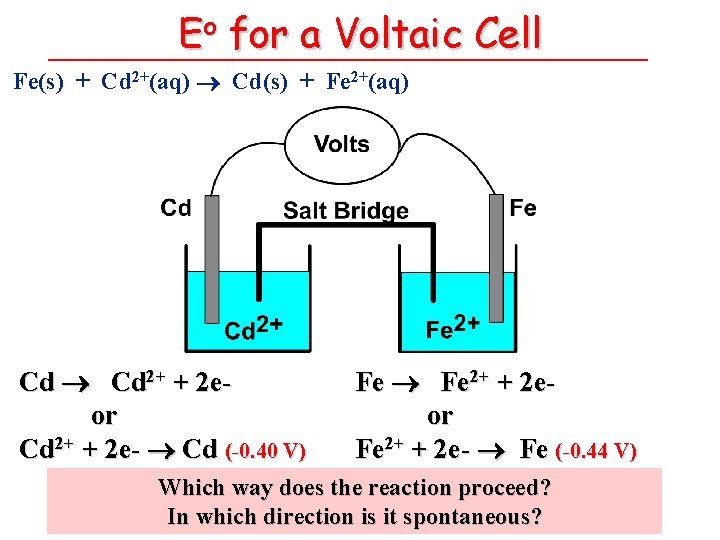
Eo for a Voltaic Cell Fe(s) + Cd 2+(aq) Cd(s) + Fe 2+(aq) Cd 2+ + 2 eor Cd 2+ + 2 e- Cd (-0. 40 V) Fe 2+ + 2 eor Fe 2+ + 2 e- Fe (-0. 44 V) Which way does the reaction proceed? In which direction is it spontaneous?
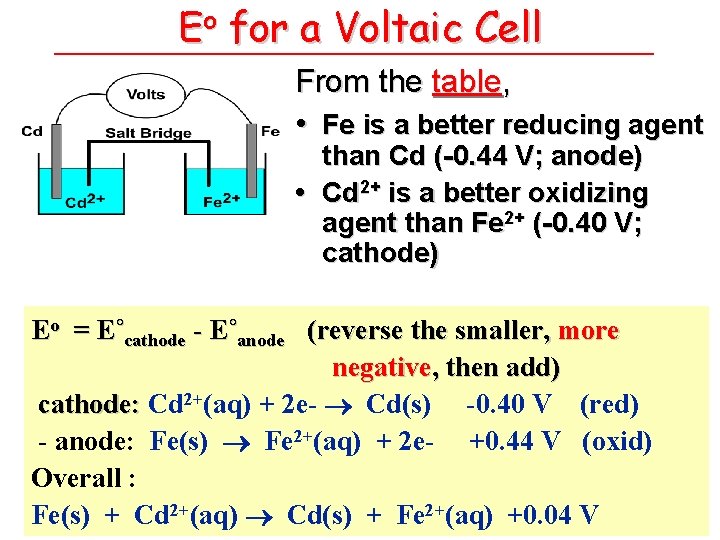
Eo for a Voltaic Cell From the table, • Fe is a better reducing agent than Cd (-0. 44 V; anode) • Cd 2+ is a better oxidizing agent than Fe 2+ (-0. 40 V; cathode) Eo = E˚cathode - E˚anode (reverse the smaller, more negative, then add) cathode: Cd 2+(aq) + 2 e- Cd(s) -0. 40 V (red) - anode: Fe(s) Fe 2+(aq) + 2 e- +0. 44 V (oxid) Overall : Fe(s) + Cd 2+(aq) Cd(s) + Fe 2+(aq) +0. 04 V

More 0 n Cell Voltage When two half-rxns (written as reduction) are joined in an electrochemical cell, the one with the larger half-cell potential occurs in the forward direction, and the one with the smaller potential occurs in the reverse direction. Cd 2+ + 2 e- Cd (-0. 40 V) Fe 2+ + 2 e- Fe (-0. 44 V) larger smaller (reverse this rxn) Cd 2+ + 2 e- Cd (-0. 40 V) Fe 2+ + 2 e- (+0. 44 V) larger overall: Cd 2+ + Fe Cd + Fe 2+ (+0. 04 V)

More 0 n Cell Voltage When two half-rxns (written as reduction) are joined in an electrochemical cell, the one with the larger half-cell potential occurs in the forward direction, and the one with the smaller potential occurs in the reverse direction. Ni 2+ + 2 e- Ni (-0. 23 V) larger Mn 2+ + 2 e- Mn (-1. 18 V) smaller (reverse this rxn) Ni 2+ + 2 e- Ni Mn 2+ + 2 e- (-0. 23 V) (+1. 18 V) overall: Ni 2+ + Mn Ni + Mn 2+ (+0. 95 V)

More 0 n Cell Voltage Assume I- ion can reduce water. 2 H 2 O + 2 e- H 2 + 2 OHCathode 2 I- I 2 + 2 e. Anode ------------------------2 I- + 2 H 2 O I 2 + 2 OH- + H 2 Assuming reaction occurs as written, E˚net = E˚cathode - E˚anode (from values in table) = (-0. 828 V) - (+0. 535 V) = -1. 363 V Minus Enet˚ means net rxn. occurs in the opposite direction (favors I- + H 2 O). I 2 + 2 OH- + H 2 2 I- + H 2 O (+1. 363 V)!!!

Calculate E cell for the reaction at 25 C Al(s) + NO 3−(aq) + 4 H+(aq) Al 3+(aq) + NO(g) + 2 H 2 O(l) (This is the reaction of Al with nitric acid) Separate the reaction into the oxidation and reduction half-reactions Find the E for each halfreaction and sum to get E cell ox: Al(s) Al 3+(aq) + 3 e− E ox = −E red = +1. 66 V red: NO 3−(aq) + 4 H+(aq) + 3 e− NO(g) + 2 H 2 O(l) E°red = +0. 96 V E ox = −(E red) = +1. 66 V E red = +0. 96 V E cell = (+1. 66 V) + (+0. 96 V) = +2. 62 V 46

For the reaction Al(s) + NO 3−(aq) + 4 H+(aq) Al 3+(aq) + NO(g) + 2 H 2 O(l) 3+ ox =+−E −red = +1. 66 V Al(s) Al. E 3 e (aq) red: NO 3−(aq) + 4 H+(aq) + 3 e− NO(g) + 2 H 2 O(l) E red = +0. 96 V ox: E cell = (+1. 66 V) + (+0. 96 V) = +2. 62 V The symbol of the cell is Al(s)| Al 3+(aq) || NO 3−(aq), 4 H+(aq) , NO(g) |Pt This is the symbol for the salt bridge This is the symbol for the electrode-solxn contact 47

E at Nonstandard Conditions • The NERNST EQUATION • • • E = potential under nonstandard conditions n = no. of electrons exchanged ln = “natural log” If [P] and [R] = 1 mol/L, then E = E˚ If [R] > [P], then E is LARGER than E˚ If [R] < [P], then E is smaller than E˚

Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s) Zn is anode (-0. 76 V) in 0. 40 M Zn 2+(aq) and Cu is cathode (0. 34 V) in 4. 8 10 -3 M Cu 2+(aq). Calculate the cell potential. Solution: (need standard cell potential, Eocell) Eºcell = Eºcathode– Eºanode = (0. 34 V) – (– 0. 76 V) = 0. 34 + 0. 76 = +1. 10 V Substituting

Zn(s) + Cu 2+(aq) Zn 2+(aq) + Cu(s) Zn is anode (-0. 76 V) and Cu is cathode (0. 34 V) n = 2 (Cu 2+ + 2 e- = Cu 0) Eocell = 1. 10 V Solution: E = 1. 10 V – (0. 01285)(4. 42) = 1. 04 V

2 Fe 3+(aq) + 3 Mg(s) 2 Fe(s) + 3 Mg 2+(aq) Fe 3+ = 1. 0 10 -3 M ; Mg 2+ = 2. 5 M Calculate the cell potential. Solution: (need standard cell potential, Eocell) Eºcell = Eºcathode– Eºanode = (-0. 036 V) – (– 2. 37 V) = -0. 036 + 2. 37 = +2. 33 V Substituting What is n? ? ?

2 Fe 3+(aq) + 3 Mg(s) 2 Fe(s) + 3 Mg 2+(aq) Fe 3+ = 1. 0 10 -3 M ; Mg 2+ = 2. 5 M Calculate the cell potential. Solution: (need standard cell potential, Eocell) Eºcell = +2. 33 V; n = 6 Substituting Ans = 2. 26 V

BATTERIES Primary, Secondary, and Fuel Cells
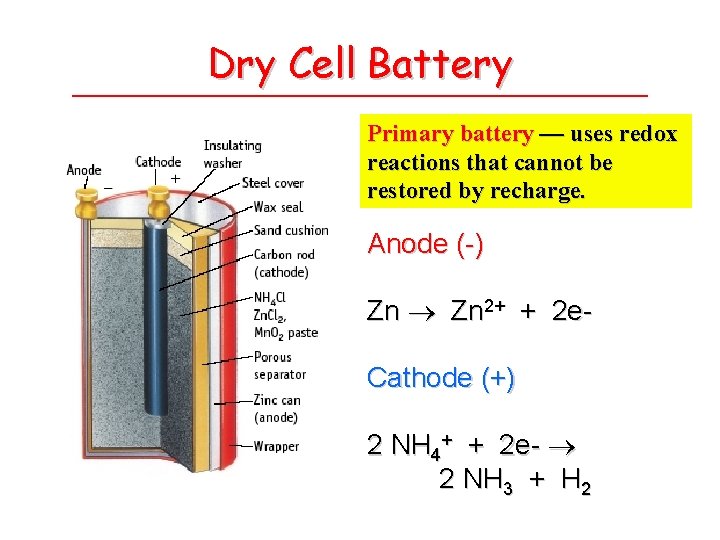
Dry Cell Battery Primary battery — uses redox reactions that cannot be restored by recharge. Anode (-) Zn 2+ + 2 e. Cathode (+) 2 NH 4+ + 2 e- 2 NH 3 + H 2
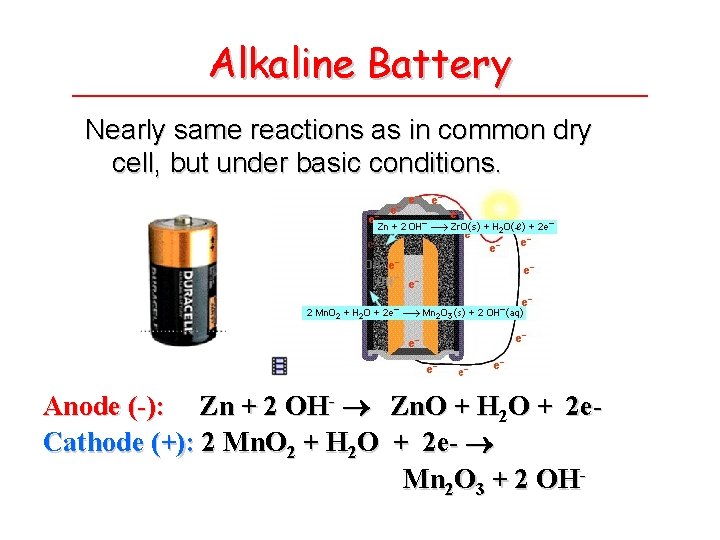
Alkaline Battery Nearly same reactions as in common dry cell, but under basic conditions. Anode (-): Zn + 2 OH- Zn. O + H 2 O + 2 e. Cathode (+): 2 Mn. O 2 + H 2 O + 2 e- Mn 2 O 3 + 2 OH-
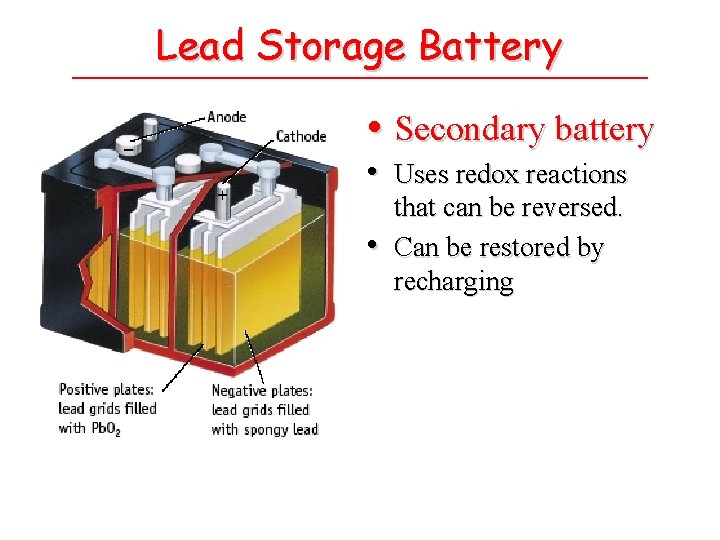
Lead Storage Battery • Secondary battery • Uses redox reactions • that can be reversed. Can be restored by recharging
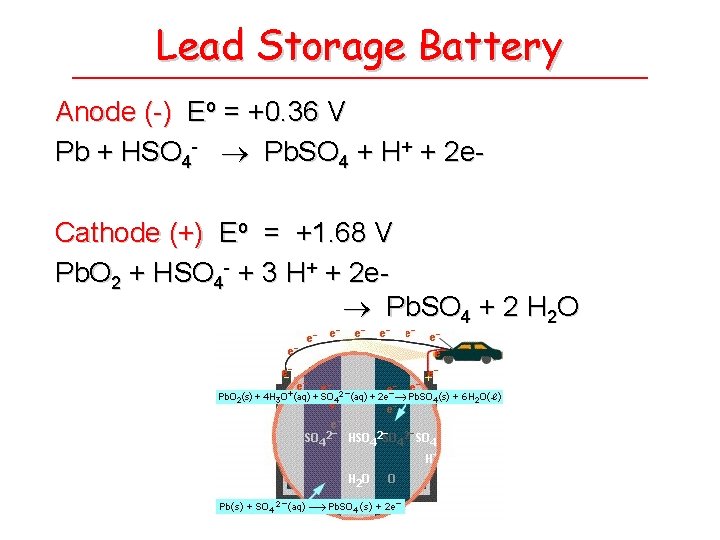
Lead Storage Battery Anode (-) Eo = +0. 36 V Pb + HSO 4 - Pb. SO 4 + H+ + 2 e. Cathode (+) Eo = +1. 68 V Pb. O 2 + HSO 4 - + 3 H+ + 2 e Pb. SO 4 + 2 H 2 O
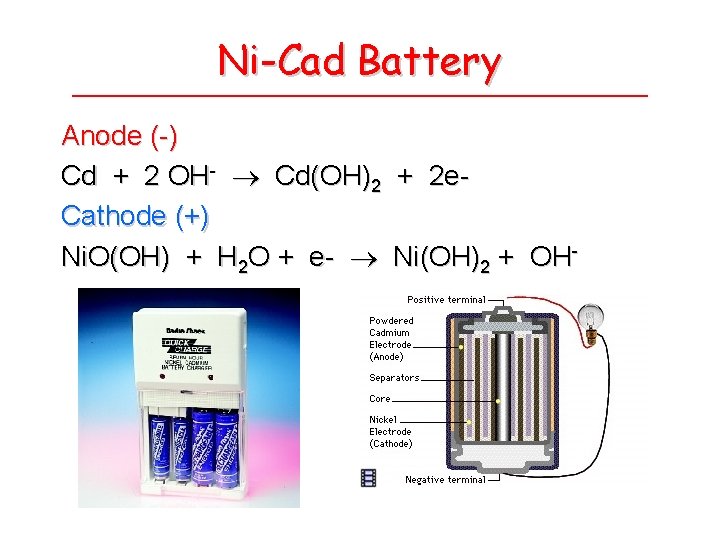
Ni-Cad Battery Anode (-) Cd + 2 OH- Cd(OH)2 + 2 e. Cathode (+) Ni. O(OH) + H 2 O + e- Ni(OH)2 + OH-

Fuel Cells: H 2 as a Fuel • Fuel cell - reactants are supplied continuously from an external source. • Cars can use electricity generated by H 2/O 2 fuel cells. • H 2 carried in tanks or generated from hydrocarbons.
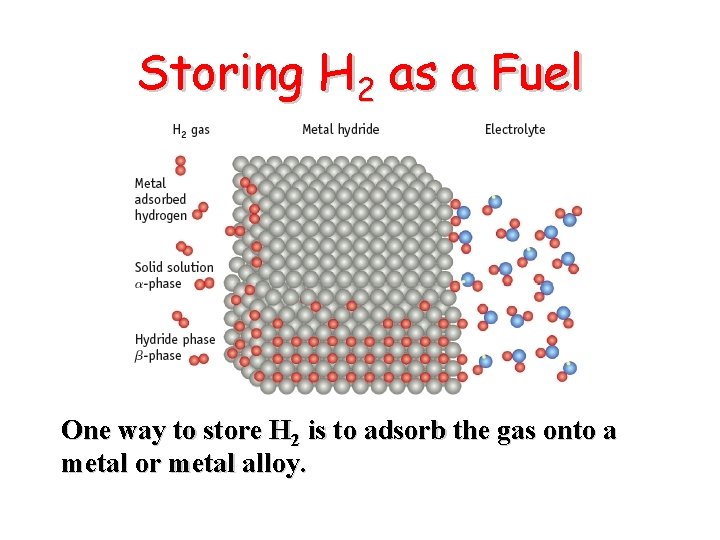
Storing H 2 as a Fuel One way to store H 2 is to adsorb the gas onto a metal or metal alloy.

Hydrogen—Air (O 2) Fuel Cell Anode: 2 H 2(g) 4 H+(aq) + 4 e. Cathode: O 2(g) + 2 H 2 O(liq) + 4 e- 4 OH- (aq) -----------------Net: O 2(g) + 2 H 2(g) 2 H 2 O(liq)
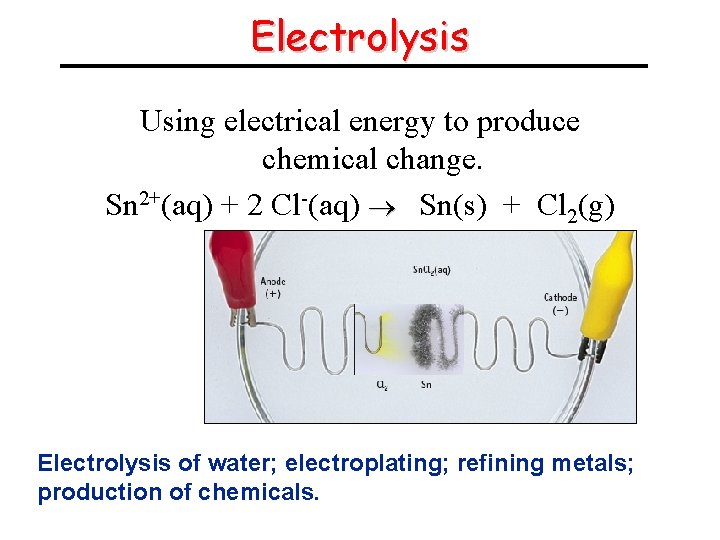
Electrolysis Using electrical energy to produce chemical change. Sn 2+(aq) + 2 Cl-(aq) Sn(s) + Cl 2(g) Electrolysis of water; electroplating; refining metals; production of chemicals.
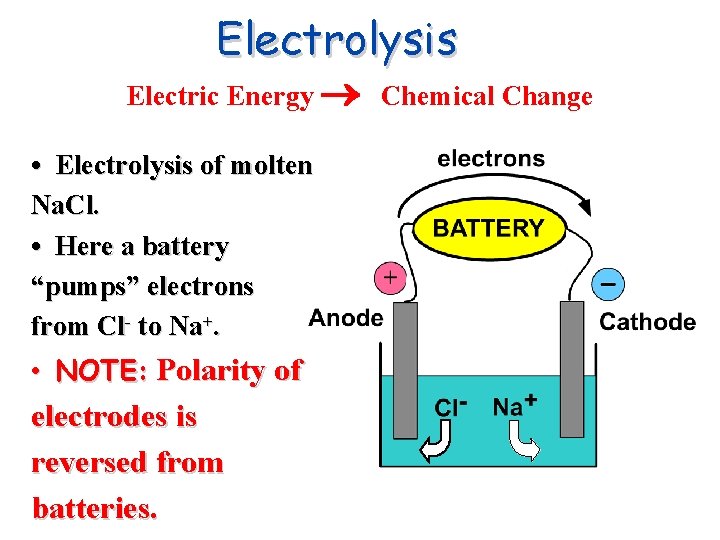
Electrolysis Electric Energy Chemical Change • Electrolysis of molten Na. Cl. • Here a battery “pumps” electrons from Cl- to Na+. • NOTE: Polarity of electrodes is reversed from batteries.
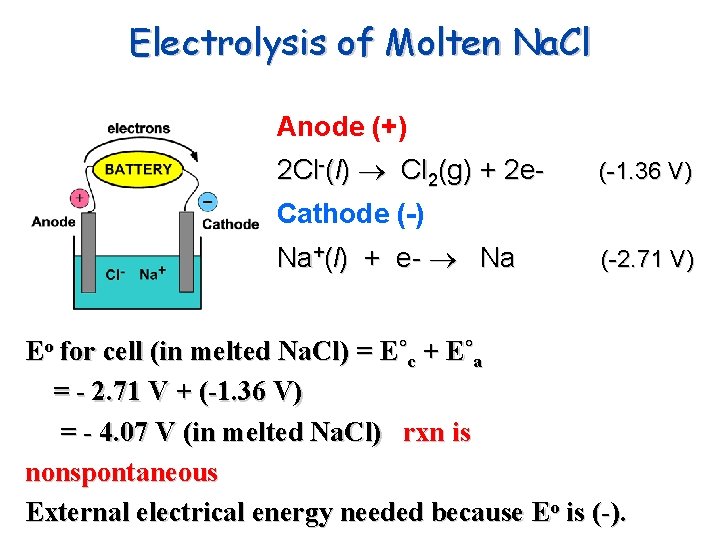
Electrolysis of Molten Na. Cl Anode (+) 2 Cl-(l) Cl 2(g) + 2 e- (-1. 36 V) Cathode (-) Na+(l) + e- Na (-2. 71 V) Eo for cell (in melted Na. Cl) = E˚c + E˚a = - 2. 71 V + (-1. 36 V) = - 4. 07 V (in melted Na. Cl) rxn is nonspontaneous External electrical energy needed because Eo is (-).

Electrolysis of Aqueous Na. OH + H 2 O Na+(aq) + OH-(aq) Electric Energy Chemical Change Anode (+) Cathode E° = -0. 40 V 4 OH- O 2(g) + 2 H 2 O + 4 e- Cathode (-) 4 H 2 O + 4 e- 2 H 2 + 4 OHEo for cell = -1. 23 V H 2 O is more easily reduced than Na+!! (Eo -2. 71 V)
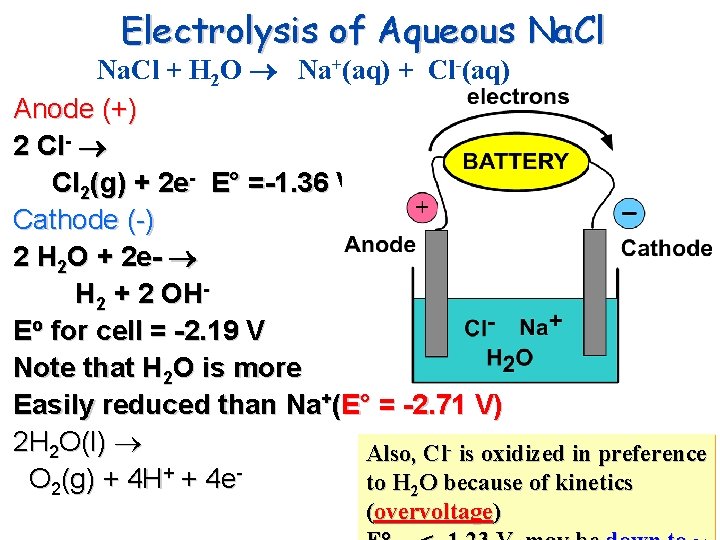
Electrolysis of Aqueous Na. Cl + H 2 O Na+(aq) + Cl-(aq) Anode (+) 2 Cl- Cl 2(g) + 2 e- E° =-1. 36 V Cathode (-) 2 H 2 O + 2 e- H 2 + 2 OHEo for cell = -2. 19 V Note that H 2 O is more Easily reduced than Na+(E° = -2. 71 V) 2 H 2 O(l) Also, Cl- is oxidized in preference O 2(g) + 4 H+ + 4 eto H 2 O because of kinetics (overvoltage)

Eo and Thermodynamics • Eo is related to ∆Go, the free energy change for the reaction. • ∆G˚ proportional to –n. E˚ ∆Go = -n. FEo where F = Faraday constant = 9. 6485 x 104 J/V • mol of e(or 9. 6485 104 coulombs/mol) and n is the number of moles of electrons transferred.
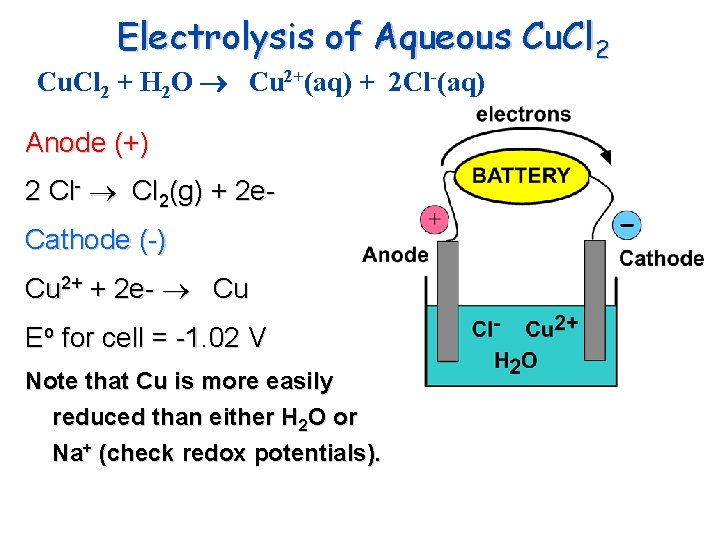
Electrolysis of Aqueous Cu. Cl 2 + H 2 O Cu 2+(aq) + 2 Cl-(aq) Anode (+) 2 Cl- Cl 2(g) + 2 e. Cathode (-) Cu 2+ + 2 e- Cu Eo for cell = -1. 02 V Note that Cu is more easily reduced than either H 2 O or Na+ (check redox potentials).

Calculate Go for the reaction, Zn 2+(aq) + Ni(s) Zn(s) + Ni 2+(aq) Solution: use Go = -n. FE° no. of electrons, n = 2 F = 9. 6485 104 C need Eocell Zn 2+(aq) + 2 e- Zn(s), -0. 763 V cathode Ni(s) Ni 2+(aq) + 2 e-, +0. 25 V anode

Eocell = Ecathode – (Eanode) Eocell = -0. 763 – (-0. 25 V) = -0. 51 V Go = (-2 96485 J/V -0. 51 V) 1 k. J/1000 J » 98 k. J ∆Go > 0, reaction is nonspontaneous Reagents are favored

Eo and ∆Go = - n F Eo For a product-favored reaction Reactants Products ∆Go < 0 and so Eo > 0 Eo is positive For a reactant-favored reaction Reactants Products ∆Go > 0 and so Eo < 0 Eo is negative
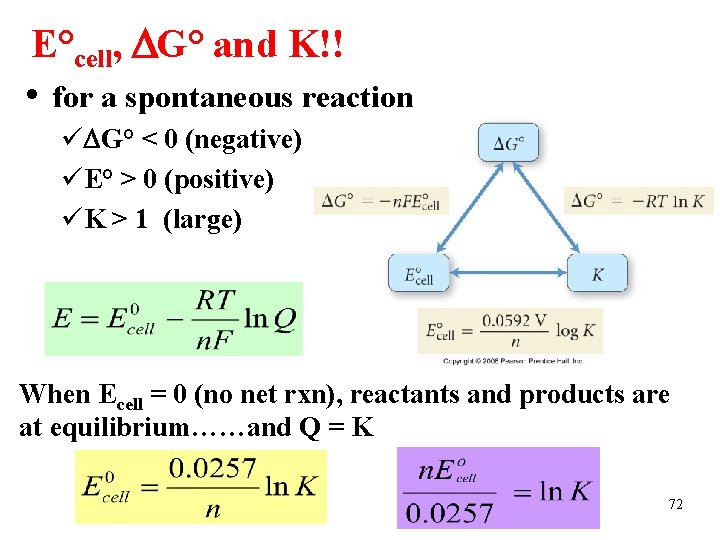
E°cell, G° and K!! • for a spontaneous reaction ü G° < 0 (negative) üE° > 0 (positive) üK > 1 (large) When Ecell = 0 (no net rxn), reactants and products are at equilibrium……and Q = K 72

Quantitative Aspects of Electrochemistry Consider electrolysis of aqueous silver ion. Ag+ (aq) + e- Ag(s) 1 mol e 1 mol Ag If we could measure the moles of e-, we could know the quantity of Ag formed. But do we measure moles of e-?

Quantitative Aspects of Electrochemistry But , how is charge related to moles of electrons? = = 96, 500 C/mol e 1 Faraday Michael Faraday 1791 -1867

Quantitative Aspects of Electrochemistry 1. 50 amps flow thru a Ag+(aq) solution for 15. 0 min. What mass of Ag metal is deposited? Solution (a) Calc. charge Charge (C) = current (A) x time (t) = (1. 5 amps)(15. 0 min)(60 s/min) = 1350 C

Quantitative Aspects of Electrochemistry 1. 50 amps flow thru a Ag+(aq) solution for 15. 0 min. What mass of Ag metal is deposited? Ag+ + e- Ag(s) Solution (a) Charge = 1350 C (b) Calculate moles of e- used (c) Calc. quantity of Ag

Quantitative Aspects of Electrochemistry The anode reaction in a lead storage battery is Pb(s) + HSO 4 -(aq) Pb. SO 4(s) + H+(aq) + 2 e. If a battery delivers 1. 50 amp, and there is 454 g of Pb, how long will the battery last? Solution a) 454 g Pb = 2. 19 mol Pb b) Calculate moles of eeach Pb atom is loosing 2 e− c) Calculate charge 4. 38 mol e- • 96, 500 C/mol e- = 423, 000 C

Quantitative Aspects of Electrochemistry The anode reaction in a lead storage battery is Pb(s) + HSO 4 -(aq) Pb. SO 4(s) + H+(aq) + 2 e. If a battery delivers 1. 50 amp, and you have 454 g of Pb, how long will the battery last? Solution a) 454 g Pb = 2. 19 mol Pb b) Mol of e- = 4. 38 mol c) Charge = 423, 000 C d) Calculate time About 78 hours
- Slides: 78