Chapter 18 Air Pollution Air pollution over Mexico























- Slides: 43

Chapter 18 Air Pollution Air pollution over Mexico City. Photo from Lawrence Livermore National Laboratory

Atmospheric Chemistry Cycles and Residence Times • Atmosphere composition mostly Nitrogen (76. 6 %), Oxygen (23. 1 %), and other gases • Chemical materials cycle through the atmosphere as they do in other reservoirs – Residence Times (Table 17. 1) – Influenced by amounts present in a given reservoir • CO 2 – 4 years • O 2 - 7 million years • N 2 – 44 million years

Cost of Air Pollution • Air pollution is costly • $40 billion – Worldwide annual expense of forest -product harvest reduction due to air pollution • $16 billion annual U. S. expense (direct costs) • Medical costs are enormous: – – Illness Medical treatment Absenteeism Loss of production

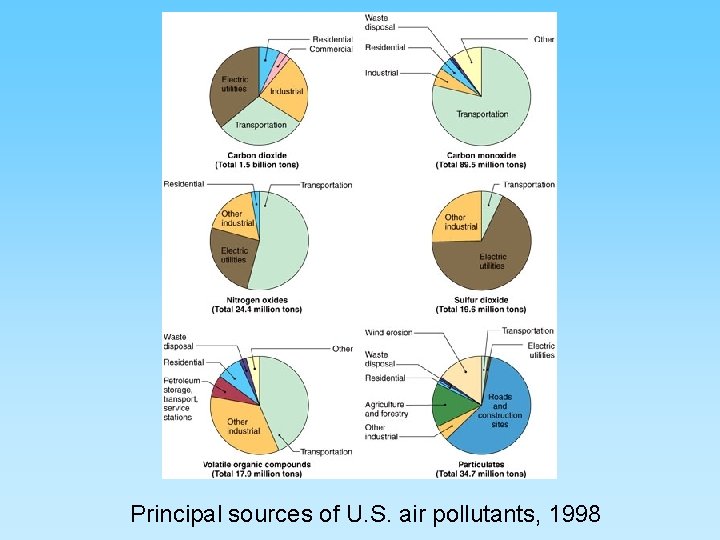
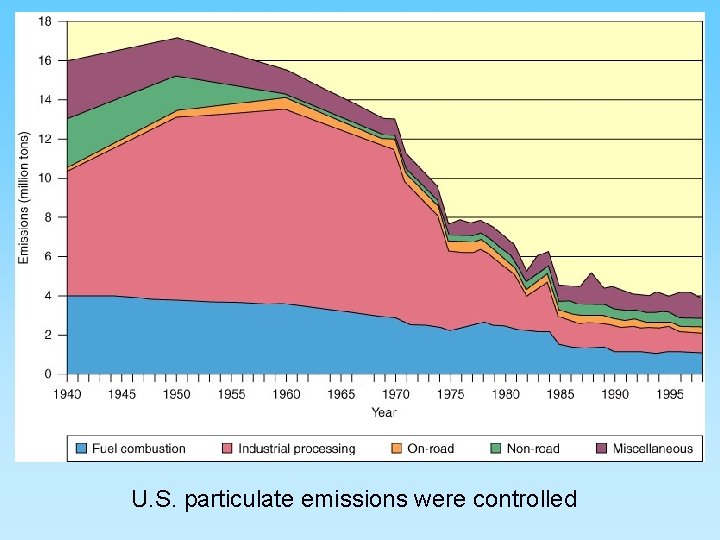
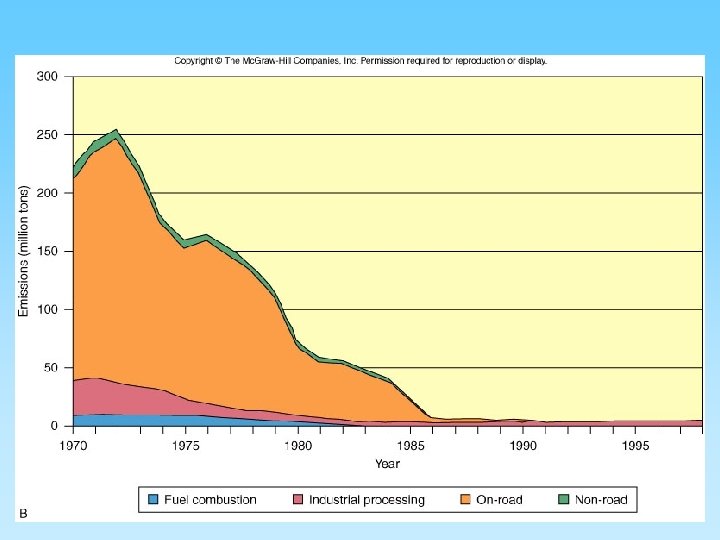
Types of Air Pollution • Gaseous pollutants – – CO and CO 2 SO 2 NO, NO 2 Ozone and CFCs • Particulates – – – Soot Smoke Ash (from burning coal) Dust (released from industrial processes) Other solids release by burning From 35 million tons/year (mainly combustion) to 180 million tons/year (mostly industrial)

Principal sources of U. S. air pollutants, 1998

U. S. particulate emissions were controlled

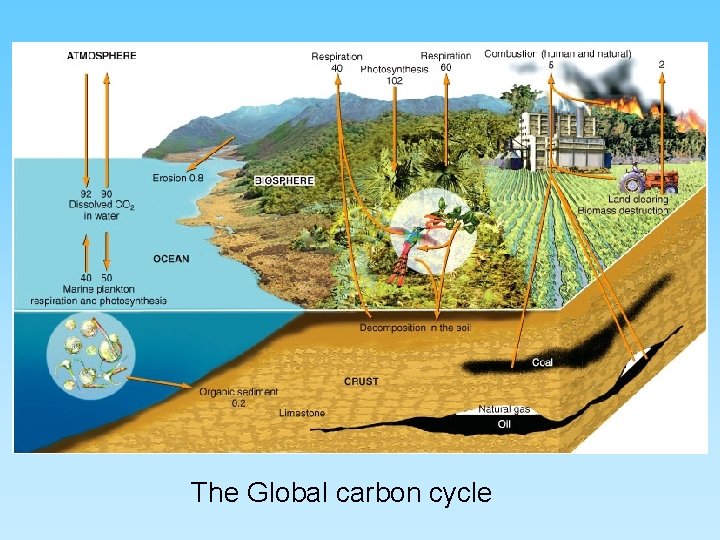
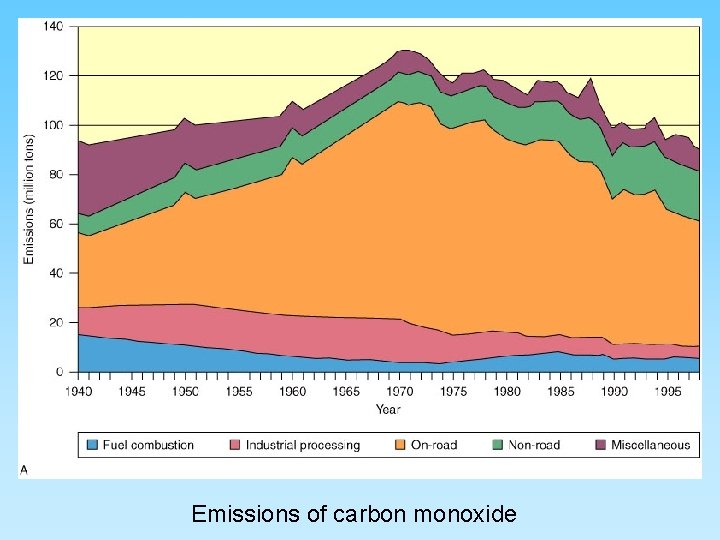
Carbon Gases • CO – carbon monoxide – Not very abundant in the atmosphere but deadly – Odorless, tasteless and invisible – toxic to animals – Very short residence time but easily added to the atmosphere by common anthropogenic sources (automobiles) • CO 2 - carbon dioxide – Essential for life of plants (photosynthesis) – Product of plant respiration, combustion, and volcanic eruptions – Short residence time and easily fluxes between oceans and atmosphere

The Global carbon cycle

Emissions of carbon monoxide

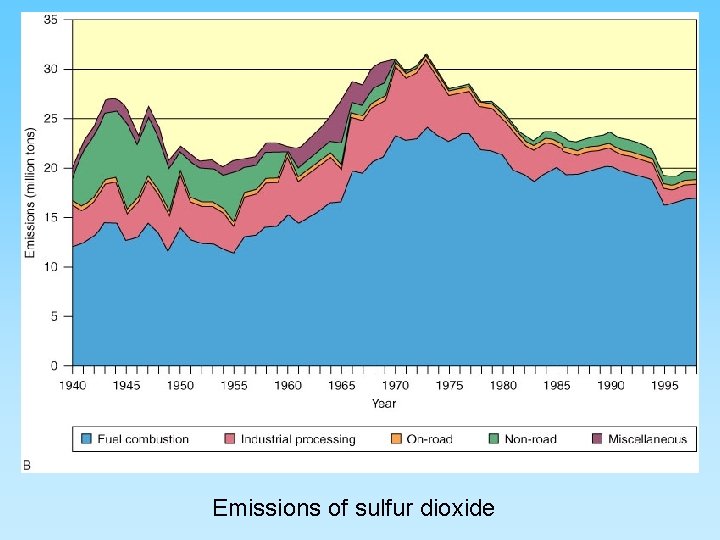
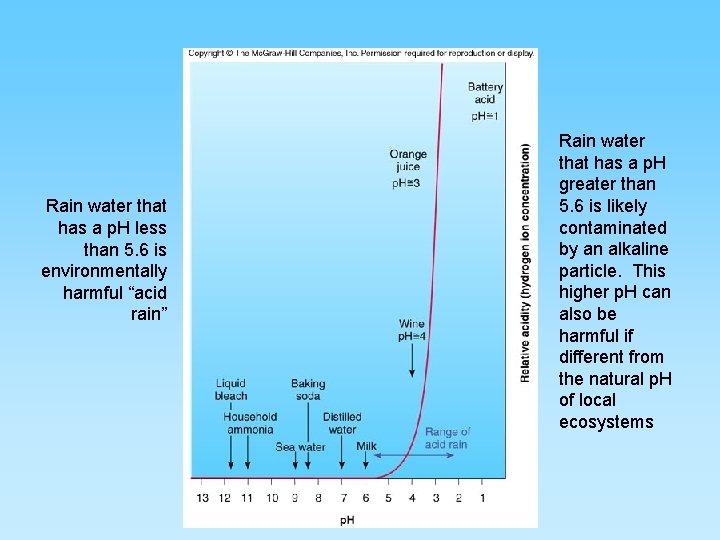
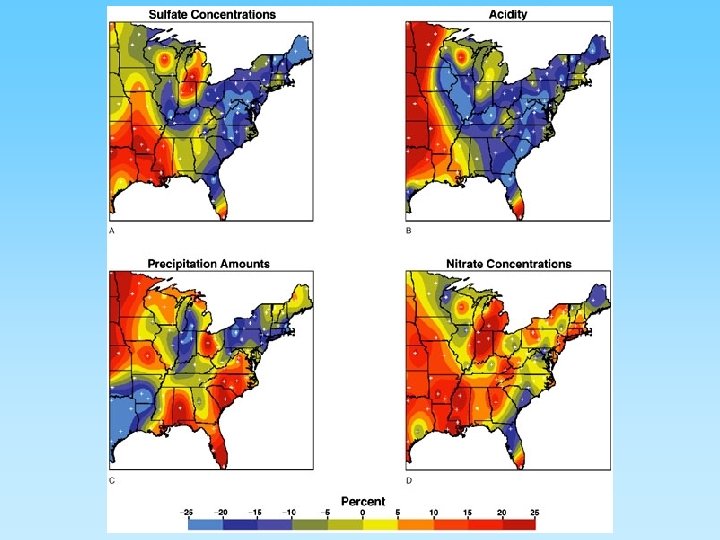
Sulfur Gases • More than 50 million tons of sulfur dioxide (SO 2) are emitted worldwide each year mainly from coal combustion • SO 2 - sulfur dioxide forms acid rain – very short residence time (days or hours) – By product from combusting coal – SO 2 + H 2 O + ½ O 2 = H 2 SO 4 (sulfuric acid) and will lower the p. H of rain to below 5. 6, the p. H of uncontaminated rain fall – Can contribute to acid rain causing lung and eye irritation – Becomes a contributor to water pollution

Emissions of sulfur dioxide

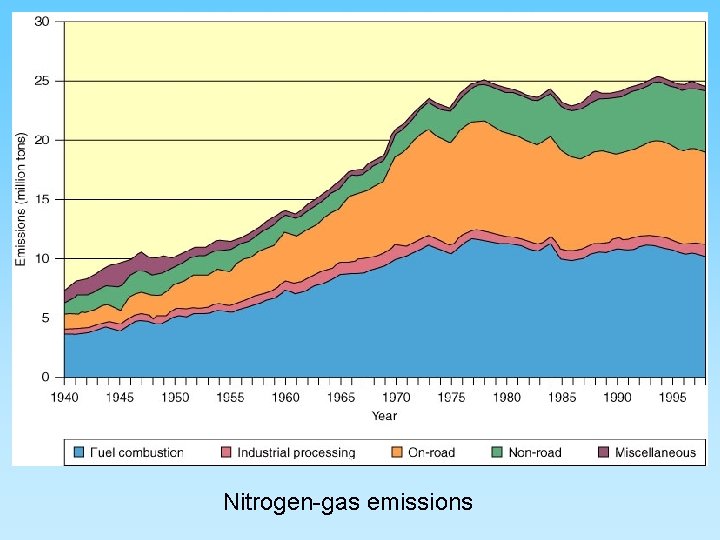
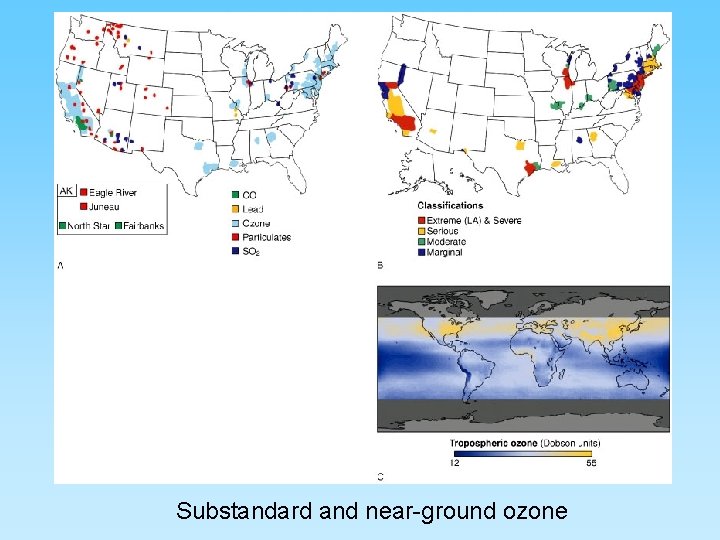
Nitrogen Gases and “Smog Ozone” • Complex geochemistry – oxygen and nitrogen are very abundant in the atmosphere – high temperatures cause nitrogen and oxygen to form nitrogen oxide compounds • 2 NO 2 + H 2 O + ½ O 2 = 2 HNO 2 (nitric acid) • NO 2 + strong sun light will produce photochemical smog • NO 2 can breakdown in sun light to NO and will react with common oxygen (O 2) to form ozone (O 3) • Ozone inhibits photosynthesis in plants and can cause severe medical problems to people in urban areas where abundant automobiles are in operation

Nitrogen-gas emissions

Substandard and near-ground ozone

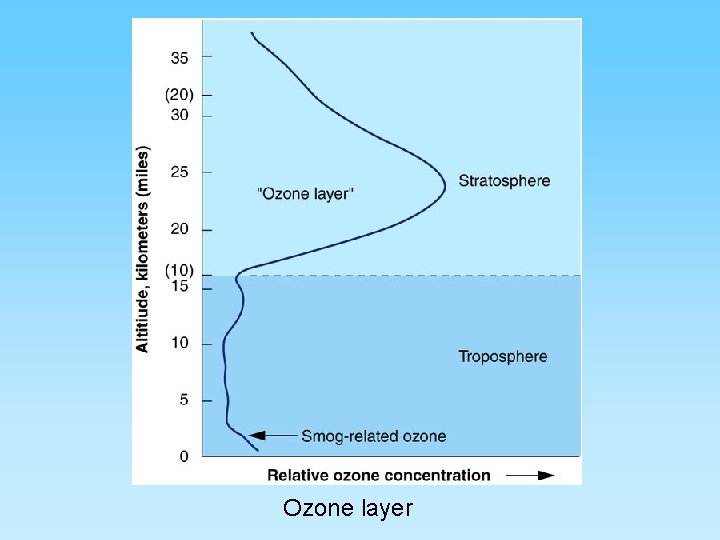
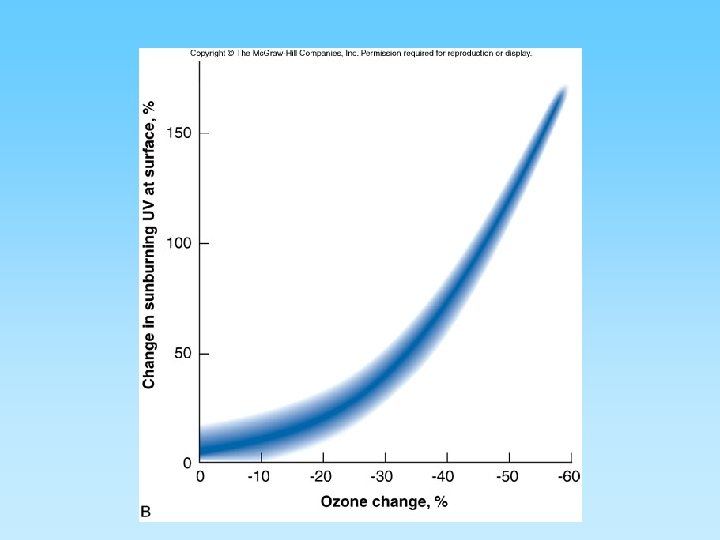
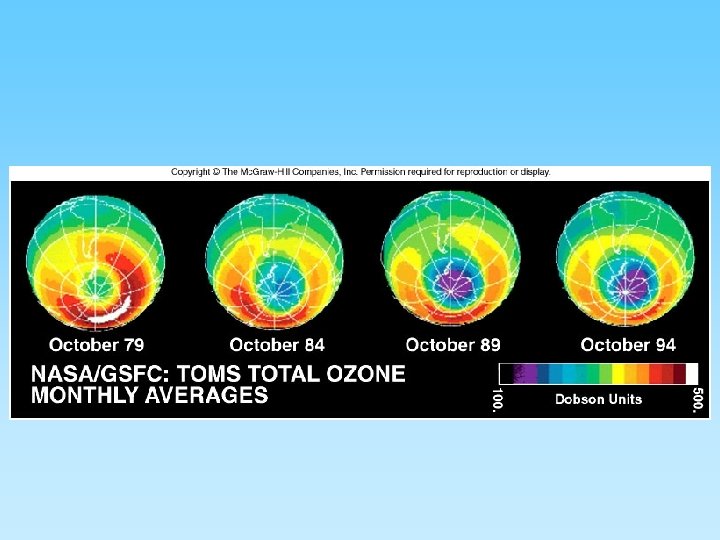
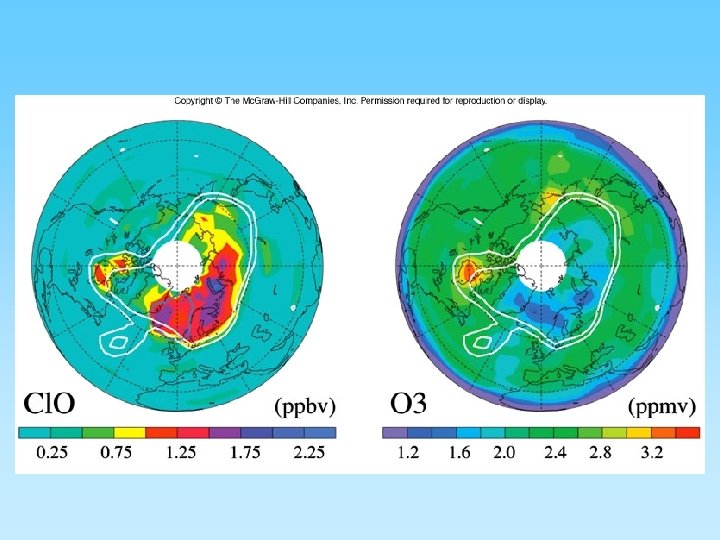
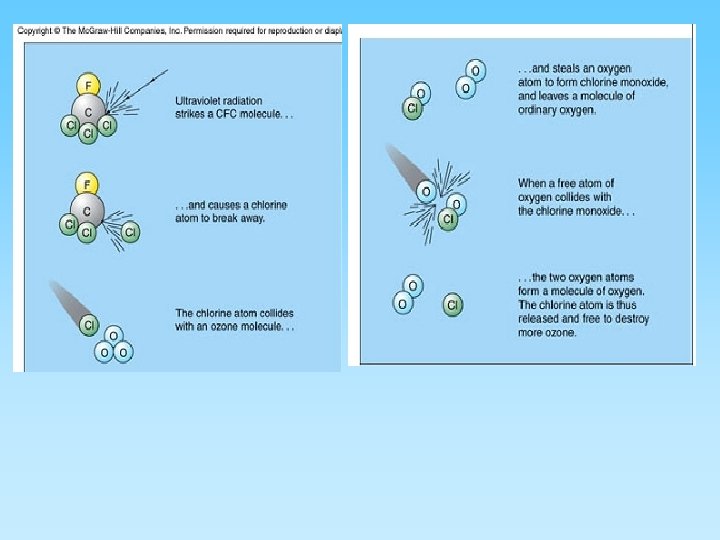
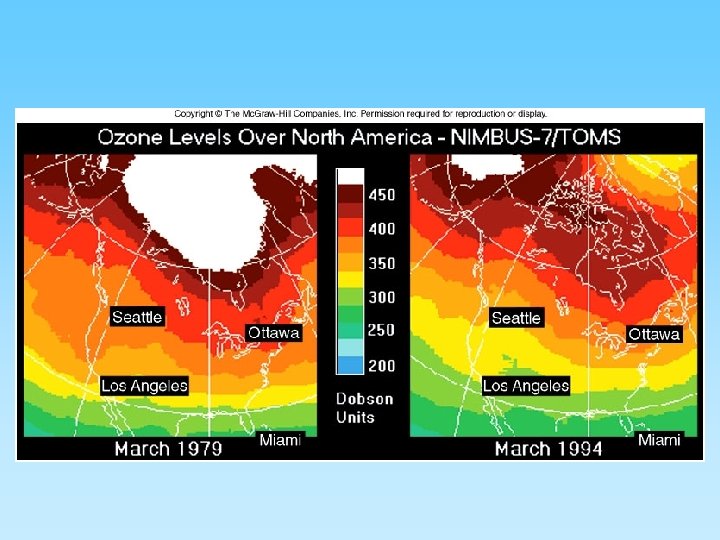
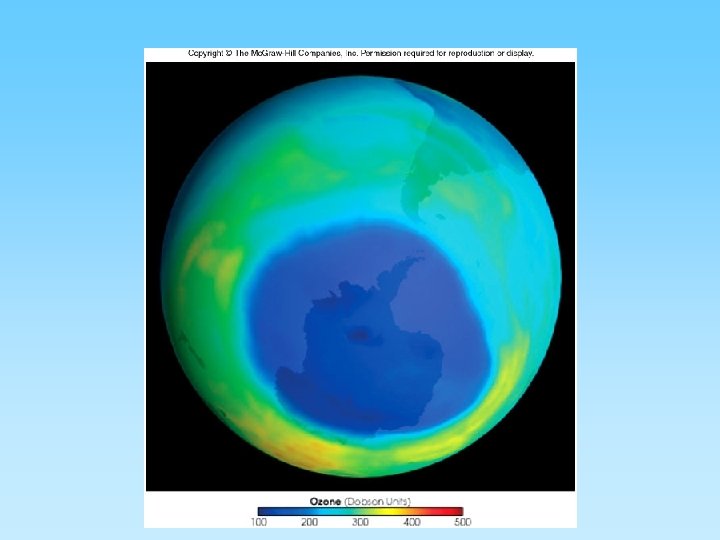
The Ozone Layer and Chlorofluorocarbons (CFCs) • Ozone (O 3) is a ‘chemically out of place pollutant’ – In upper atmosphere (stratosphere) the ozone layer absorbs harmful ultraviolet radiation – Ozone forms as O 2 + ½ O 2 = O 3 – At ground level it is very harmful to plants and animals – A thick ozone layer is beneficial to life on earth – Periodically, an ozone hole forms over either pole. This allows more ultra violet radiation to enter the lower atmosphere – CFCs attack ozone and can destroy, or thin, the ozone layer – CFCs build up their concentration in the upper atmosphere, have a residence time of about 100 years, and breakdown into various by-products including chlorine monoxide (Cl. O) – One Cl. O can destroy many ozone molecules

Ozone layer

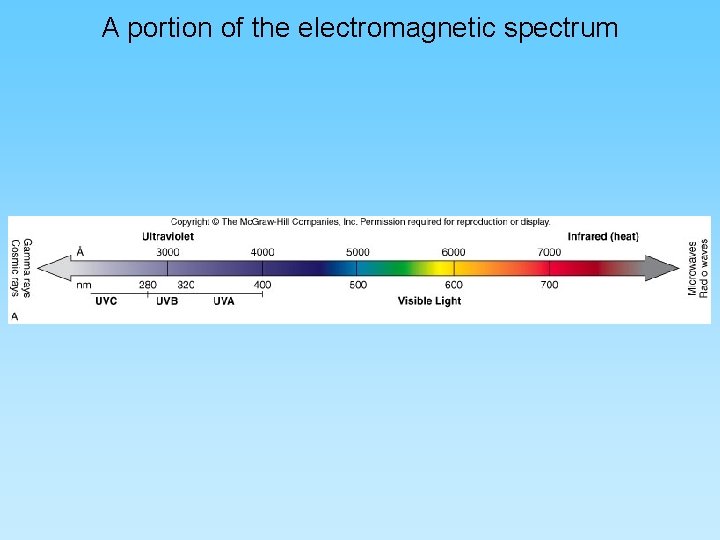
A portion of the electromagnetic spectrum








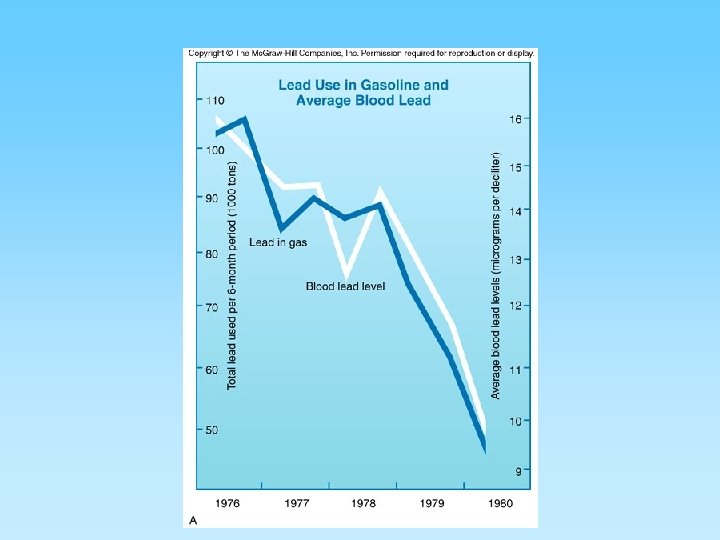
Particulate Pollutants • Lead - once a serious pollutant – Used in gasoline as an antiknock additive, tons of lead were released into the atmosphere by internal combustion engines – Unleaded fuels have reduced lead in air pollution • High doses of lead can cause brain damage, depression, apathy, and other psychological disorders



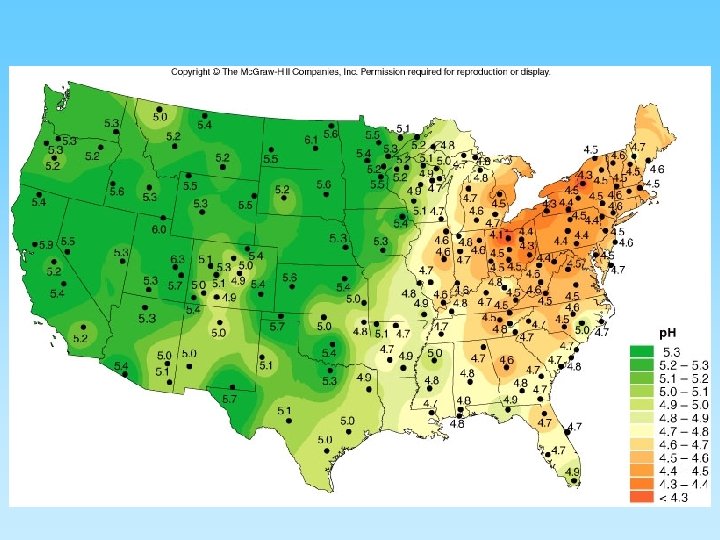
Acid Rain • Acidity is reported on the p. H scale. – An acid solution has more Hydrogen-ions (H+) – A basic solution would have fewer • Acid rain has more acid than normal rain – Certain gases in the atmosphere will complex with atmospheric water plus oxygen to form acids • Acid rain is harmful to plants, health of rivers and lakes, and animals • Acid rain causes increases in the build up of heavy metals (lead, zinc, selenium, copper, and aluminum) leached from rocks and soils • Toxic levels are then found in our waterways, fish, and fish eaters

Rain water that has a p. H less than 5. 6 is environmentally harmful “acid rain” Rain water that has a p. H greater than 5. 6 is likely contaminated by an alkaline particle. This higher p. H can also be harmful if different from the natural p. H of local ecosystems


Regional Variations in Rainfall Acidity and Impacts • Rain down wind of an industrial or populated areas have greater acidic conditions – Generally, industrial areas have notably higher sulfur dioxide emissions than non-industrial areas – Urban areas, with automobiles, generally have higher nitrogen oxide emissions than areas without autos • Local geology can reduce the acidity of waterways that receive acid rain. – Limestone can neutralize acidic water; granite can not


Air Pollution and Weather • Thermal Inversion – Pollutants released from ground sources generally are warmer than the surrounding air and tend to rise – As an air mass these plumes will rise and cool – In certain situations a cool air mass will have a warm air mass overlying it, it will trap the rising warm pollutant-bearing air mass; this condition is made worse by an air mass with stagnant conditions – As more pollutants are dumped into the cooler air mass they will become concentrated below the warm air mass – Eventually, the concentrations of pollutants may become very high and cause a health concern – Local topography – mountains, basins, valleys, and local wind patterns may amplify the situation also






Air Pollution and Weather • Air pollution can impact local weather – Reduce visibility – Reduce air quality – Modify air temperature – Produce acid rain – Produce abnormal rain


Air Pollution Control • Air Quality Standards – Clean Air Act (1970) – Designed to protect public health, clear the visible pollution in the air, and prevent damage to crops • Control Methods – Trapping or scrubbing emissions – conversion techniques that reduce the emission of harmful compounds to less harmful ones • Automobile Emissions – strong legislation creates the Environmental Protection Agency (EPA) – Goal to reduce automobile emissions – Limited success achieved by requiring the use of catalytic converters in new cars and increased fuel economy standards for all new cars • Cost and Effects – it has increased abatement and control cost while limited areas of success have occurred

Air Pollution Control • Also requires the control (abatement) of dust (solid particulate). Methods of dust control include – Using water trucks on construction sites to minimize dust – Using bag houses and water sprays in crushing and milling operations – Applying soil palliatives for longer term dust control

Fig. 17. 23