Chapter 18 18 1 AcidBase Equilibria Arrhenius AcidBase
Chapter 18 18 -1 Acid-Base Equilibria

Arrhenius Acid-Base Definition This acid-base definition, which classifies these substances in terms of their behavior in water ; An acid is a substance with H+ in its molecule that dissociates to yield H 3 O+. A base is a substance with OH- in its molecule that dissociates to yield OH- in solution. Because they are ionic compounds, Arrhenius bases contain discrete OH- ions in their structures. But , Arrhenius acids never contain discrete H+ ions. Instead, they contain covalently bonded H atoms that ionize when molecules of the acid dissolve in water. Arrhenius realized that even though some substances do not have discrete OHions, they still behave as bases. For example, NH 3 and K 2 CO 3 also yield OH in water. Then a broader acid-base definitions are required to include species that do not have OH in their formula When an strong acid reacts with a strong base, they undergo neutralization: H+(aq) + OH-(aq) → H 2 O(l) DH°rxn = -55. 9 k. J 18 -2

Strong and Weak Acids A strong acid dissociates completely into ions in water: HA(g or l) + H 2 O(l) → H 3 O+(aq) + A-(aq) A dilute solution of a strong acid contains no HA molecules. A weak acid dissociates slightly to form ions in water: HA(aq) + H 2 O(l) H 3 O+(aq) + A-(aq) In a dilute solution of a weak acid, most HA molecules are undissociated. Kc = 18 -3 [H 3 O+][A-] [HA][H 2 O] has a very small value.
![The Acid Dissociation Constant, Ka HA(aq) + H 2 O(l) [H 3 O+][A–] Kc The Acid Dissociation Constant, Ka HA(aq) + H 2 O(l) [H 3 O+][A–] Kc](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-4.jpg)
The Acid Dissociation Constant, Ka HA(aq) + H 2 O(l) [H 3 O+][A–] Kc = [HA][H 2 O] H 3 O+(aq) + A–(aq) [H 3 O+][A–] Kc[H 2 O] = Ka = [HA] The value of Ka is an indication of acid strength. Stronger acid higher [H 3 O+] Weaker acid lower % dissociation of HA 18 -4 larger Ka smaller Ka

Table 18. 2 18 -5 Ka Values for some Monoprotic Acids at 25°C

Classifying the Relative Strengths of Acids • Strong acids include – the hydrohalic acids (HCl, HBr, and HI) and – oxoacids in which the number of O atoms exceeds the number of ionizable protons by two or more (eg. , HNO 3, H 2 SO 4, HCl. O 4. ) • Weak acids include – the hydrohalic acid HF, – acids in which H is not bonded to O or to a halogen (eg. , HCN), – oxoacids in which the number of O atoms equals or exceeds the number of ionizable protons by one (eg. , HCl. O, HNO 2), and – carboxylic acids, which have the general formula RCOOH (eg. , CH 3 COOH and C 6 H 5 COOH. ) 18 -6

Classifying the Relative Strengths of Bases • Strong bases include – water-soluble compounds containing O 2 - or OH- ions. – The cations are usually those of the most active metals: • M 2 O or MOH, where M = Group 1 A(1) metal (Li, Na, K, Rb, Cs) • MO or M(OH)2 where M = group 2 A(2) metal (Ca, Sr, Ba). • Weak bases include – ammonia (NH 3), – amines, which have the general formula – The common structural feature is an N atom with a lone electron pair. 18 -7
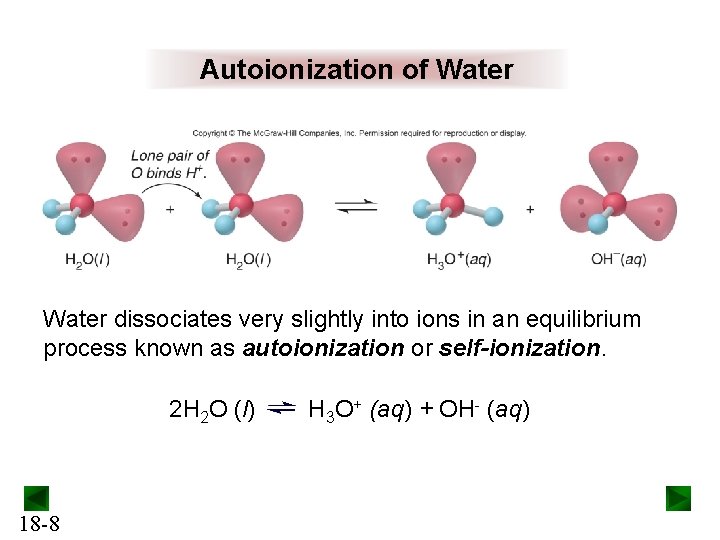
Autoionization of Water dissociates very slightly into ions in an equilibrium process known as autoionization or self-ionization. 2 H 2 O (l) 18 -8 H 3 O+ (aq) + OH- (aq)

The Ion-Product Constant for Water (Kw) 2 H 2 O (l) H 3 O+ (aq) + OH- (aq) [H 3 O+][OH-] Kc = [H 2 O]2 Kc[H 2 O]2 = Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 (at 25°C) In pure water, ______ [H 3 O+] = [OH-] = √ 1 X 10 -14 = 1. 0 x 10 -7 (at 25°C) Both ions are present in all aqueous systems. 18 -9
![A change in [H 3 O+] causes an inverse change in [OH-], and vice A change in [H 3 O+] causes an inverse change in [OH-], and vice](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-10.jpg)
A change in [H 3 O+] causes an inverse change in [OH-], and vice versa. Higher [H 3 O+] lower [OH-] Higher [OH-] lower [H 3 O+] We can define the terms “acidic” and “basic” in terms of the relative concentrations of H 3 O+ and OH– ions: In an acidic solution, In a neutral solution, In a basic solution, 18 -10 [H 3 O+] > [OH–] [H 3 O+] = [OH–] [H 3 O+] < [OH–]
![Figure 18. 3 18 -11 The relationship between [H 3 O+] and [OH-] and Figure 18. 3 18 -11 The relationship between [H 3 O+] and [OH-] and](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-11.jpg)
Figure 18. 3 18 -11 The relationship between [H 3 O+] and [OH-] and the relative acidity of solutions.
![Sample Problem 18. 2 Calculating [H 3 O+] or [OH–] in an Aqueous Solution Sample Problem 18. 2 Calculating [H 3 O+] or [OH–] in an Aqueous Solution](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-12.jpg)
Sample Problem 18. 2 Calculating [H 3 O+] or [OH–] in an Aqueous Solution PROBLEM: Adding a measured amount of HCl gas to pure water at 25°C and obtains a solution with [H 3 O+] = 3. 0 x 10– 4 M. Calculate [OH–]. Is the solution neutral, acidic, or basic? PLAN: Using the known value of Kw at 25°C (1. 0 x 10– 14) and the given [H 3 O+] to solve for [OH–]. We can then compare [H 3 O+] with [OH–] to determine whether the solution is acidic, basic, or neutral. SOLUTION: Kw = 1. 0 x 10– 14 = [H 3 O+] [OH–] so Kw – [OH ] = [H 3 O+] 1. 0 x 10– 14 = 3. 0 x 10– 4 = 3. 3 x 10– 11 M [H 3 O+] is > [OH–] and the solution is acidic. 18 -12
![The p. H Scale p. H = -log[H 3 O+] The p. H of The p. H Scale p. H = -log[H 3 O+] The p. H of](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-13.jpg)
The p. H Scale p. H = -log[H 3 O+] The p. H of a solution indicates its relative acidity: In an acidic solution, In a neutral solution, In a basic solution, p. H < 7. 00 p. H = 7. 00 p. H > 7. 00 Why use a log scale? It is easier to graph and do calculations using logs. The higher the p. H, the lower the [H 3 O+] and the less acidic the solution. 18 -13
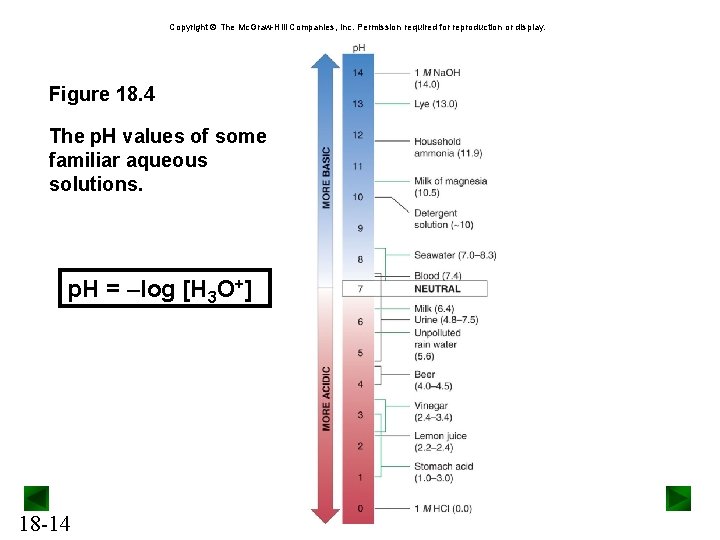
Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Figure 18. 4 The p. H values of some familiar aqueous solutions. p. H = –log [H 3 O+] 18 -14

Table 18. 3 The Relationship between Ka and p. Ka Acid Name (Formula) Ka at 25°C p. Ka Hydrogen sulfate ion (HSO 4 -) 1. 0 x 10– 2 1. 99 Nitrous acid (HNO 2) 7. 1 x 10– 4 3. 15 Acetic acid (CH 3 COOH) 1. 8 x 10– 5 4. 75 Hypobromous acid (HBr. O) 2. 3 x 10– 9 8. 64 Phenol (C 6 H 5 OH) 1. 0 x 10– 10 10. 00 p. Ka = –log. Ka A low p. Ka corresponds to a high Ka. 18 -15
![p. H, p. OH, and p. Kw Kw = [H 3 O+][OH–] = 1. p. H, p. OH, and p. Kw Kw = [H 3 O+][OH–] = 1.](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-16.jpg)
p. H, p. OH, and p. Kw Kw = [H 3 O+][OH–] = 1. 0 x 10– 14 at 25°C p. H = –log[H 3 O+] p. OH = –log[OH–] p. Kw = p. H + p. OH = 14. 00 at 25°C p. H + p. OH = p. Kw for any aqueous solution at any temperature. Since Kw is a constant, the values of p. H, p. OH, [H 3 O+], and [OH–] are interrelated: • If [H 3 O+] increases, [OH–] decreases (and vice versa). • If p. H increases, p. OH decreases (and vice versa). 18 -16
![Figure 18. 5 The relations among [H 3 O+], p. H, [OH-], and p. Figure 18. 5 The relations among [H 3 O+], p. H, [OH-], and p.](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-17.jpg)
Figure 18. 5 The relations among [H 3 O+], p. H, [OH-], and p. OH. Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 18 -17
![Sample Problem 18. 3 Calculating [H 3 O+], p. H, [OH-], and p. OH Sample Problem 18. 3 Calculating [H 3 O+], p. H, [OH-], and p. OH](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-18.jpg)
Sample Problem 18. 3 Calculating [H 3 O+], p. H, [OH-], and p. OH for Strong Acids and Bases PROBLEM: Calculate [H 3 O+], p. H, [OH–], and p. OH for each solution at 25°C: (a) 0. 30 M HNO 3, used for etching copper metal; and (b) 0. 0042 M Ca(OH)2, used in leather tanning to remove hair from hides. PLAN: HNO 3 is a strong acid; so it dissociates completely, and [H 3 O+] = [HNO 3]init. Likewise, Ca(OH)2 is a strong base that dissociates completely, and [OH–] = 2[Ca(OH)2]init. We use the given concentrations and the value of Kw at 25°C to find [OH–] and [H 3 O+]. We can then calculate p. H and p. OH. 18 -18
![SOLUTION: (a) Calculating the values for 0. 30 M HNO 3: [H 3 O+] SOLUTION: (a) Calculating the values for 0. 30 M HNO 3: [H 3 O+]](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-19.jpg)
SOLUTION: (a) Calculating the values for 0. 30 M HNO 3: [H 3 O+] = 0. 30 M p. H = –log[H 3 O+] = -log(0. 30) = 0. 52 – 14 K 1. 0 x 10 w -14 M = 3. 3 x 10 = [OH–] = [H 3 O+] 0. 30 p. OH = –log[OH–] = –log(3. 3 x 10– 14) = 13. 48 (b) Calculating the values for 0. 0042 M Ca(OH)2: Ca(OH)2 is a strong electrolyte: Ca(OH)2(aq) → Ca 2+(aq) + 2 OH–(aq) [OH–] = 2 (0. 0042 M) = 0. 0084 M p. OH = –log[OH–] = -log 0. 0084 = 2. 08 1. 0 x 10 -14 Kw + = = 1. 2 x 10– 12 M [H 3 O ] = 0. 0084 [OH–] p. H = –log[H 3 O+] = –log(1. 2 x 10– 12) = 11. 92 18 -19

Brønsted-Lowry Acid-Base Definition An acid is a proton donor, any species that donates an H+ ion. • An acid must contain H in its formula. A base is a proton acceptor, any species that accepts an H+ ion. • A base must contain a lone pair of electrons to bond to H+. An acid-base reaction is a proton-transfer process. 18 -20
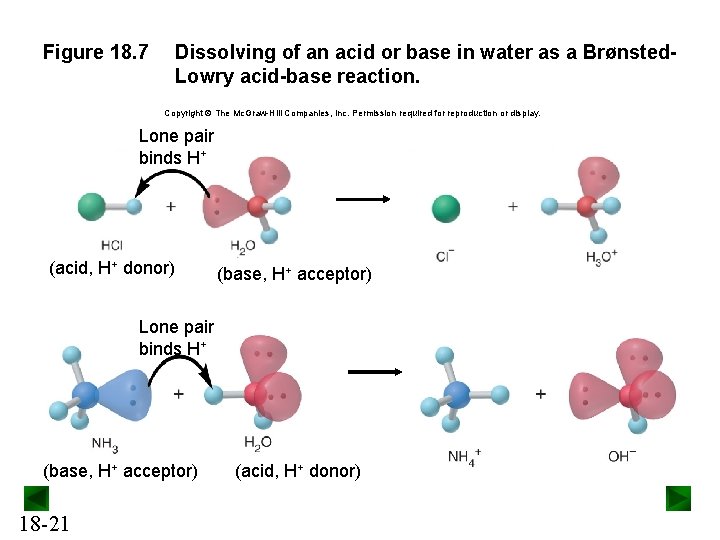
Figure 18. 7 Dissolving of an acid or base in water as a Brønsted. Lowry acid-base reaction. Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Lone pair binds H+ (acid, H+ donor) (base, H+ acceptor) Lone pair binds H+ (base, H+ acceptor) 18 -21 (acid, H+ donor)
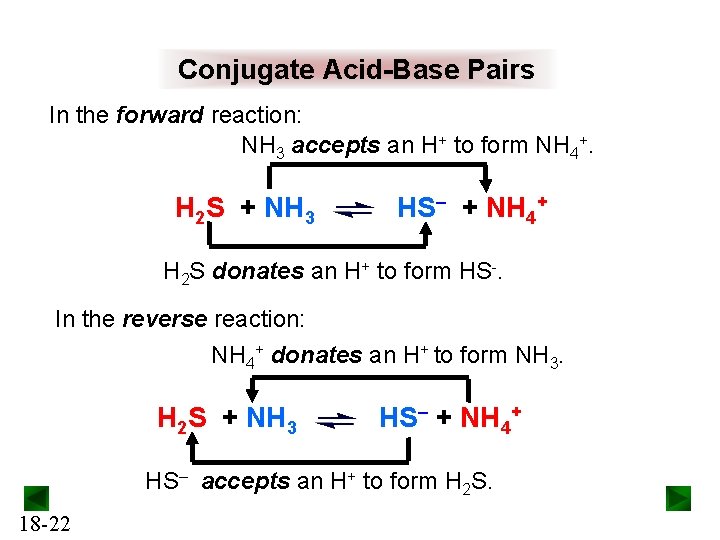
Conjugate Acid-Base Pairs In the forward reaction: NH 3 accepts an H+ to form NH 4+. H 2 S + NH 3 HS– + NH 4+ H 2 S donates an H+ to form HS-. In the reverse reaction: NH 4+ donates an H+ to form NH 3. H 2 S + NH 3 HS– + NH 4+ HS– accepts an H+ to form H 2 S. 18 -22

Conjugate Acid-Base Pairs H 2 S + NH 3 HS– + NH 4+ H 2 S and HS– are a conjugate acid-base pair: HS– is the conjugate base of the acid H 2 S. NH 3 and NH 4+ are a conjugate acid-base pair: NH 4+ is the conjugate acid of the base NH 3. A Brønsted-Lowry acid-base reaction occurs when an acid and a base react to form their conjugate base and conjugate acid, respectively. acid 1 + base 2 18 -23 base 1 + acid 2
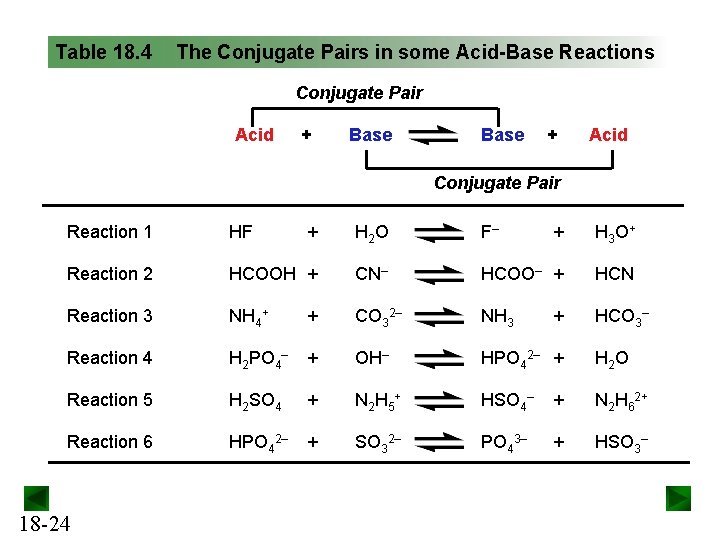
Table 18. 4 The Conjugate Pairs in some Acid-Base Reactions Conjugate Pair Acid + Base + Acid Conjugate Pair Reaction 1 HF + H 2 O F– + H 3 O + Reaction 2 HCOOH + CN– HCOO– + HCN Reaction 3 NH 4+ + CO 32– NH 3 HCO 3– Reaction 4 H 2 PO 4– + OH– HPO 42– + H 2 O Reaction 5 H 2 SO 4 + N 2 H 5 + HSO 4– + N 2 H 62+ Reaction 6 HPO 42– + SO 32– PO 43– + HSO 3– 18 -24 +

Sample Problem 18. 4 Identifying Conjugate Acid-Base Pairs PROBLEM: The following reactions are important environmental processes. Identify the conjugate acid-base pairs. (a) H 2 PO 4–(aq) + CO 32– (aq) (b) H 2 O(l) + SO 32–(aq) HPO 42 -–(aq) + HCO 3–(aq) OH–(aq) + HSO 3–(aq) PLAN: To find the conjugate pairs, we find the species that donated an H+ (acid) and the species that accepted it (base). The acid donates an H+ to become its conjugate base, and the base accepts an H+ to become its conjugate acid. SOLUTION: (a) H 2 PO 4–(aq) + CO 32–(aq) base 2 acid 1 HPO 42–(aq) + HCO 3–(aq) acid 2 base 1 The conjugate acid-base pairs are H 2 PO 4–/HPO 42– and CO 32–/HCO 3–. 18 -25
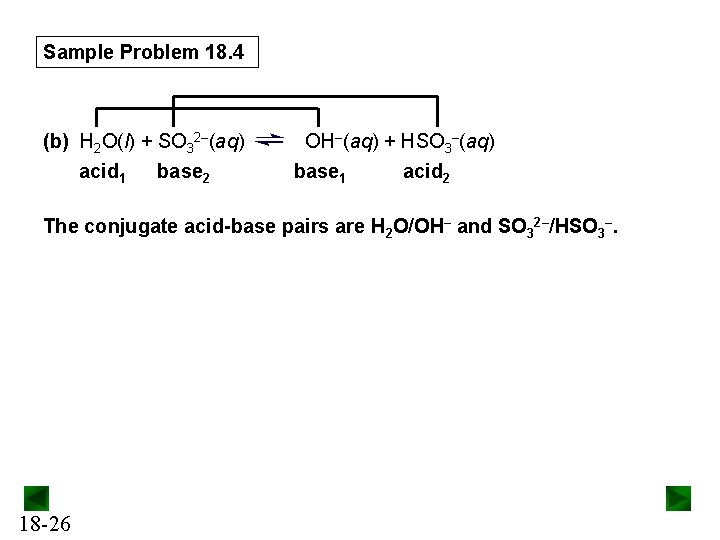
Sample Problem 18. 4 (b) H 2 O(l) + SO 32–(aq) acid 1 base 2 OH–(aq) + HSO 3–(aq) base 1 acid 2 The conjugate acid-base pairs are H 2 O/OH– and SO 32–/HSO 3–. 18 -26
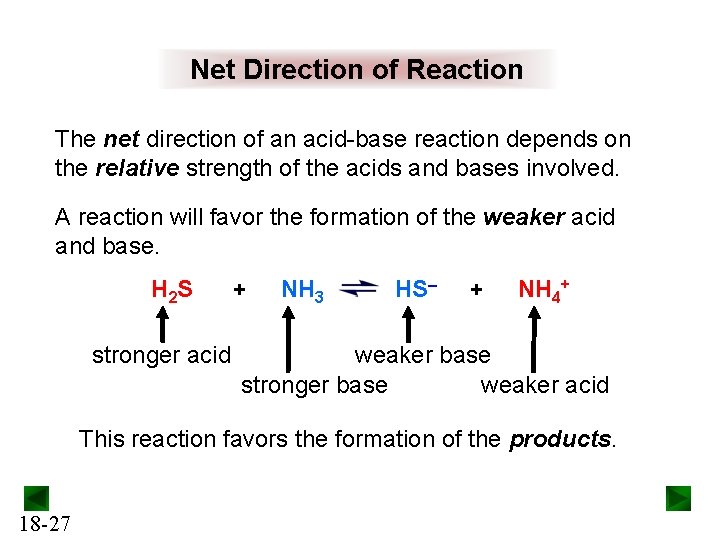
Net Direction of Reaction The net direction of an acid-base reaction depends on the relative strength of the acids and bases involved. A reaction will favor the formation of the weaker acid and base. H 2 S stronger acid + NH 3 HS– + NH 4+ weaker base stronger base weaker acid This reaction favors the formation of the products. 18 -27

Figure 18. 8 Strengths of conjugate acid-base pairs. Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. The stronger the acid is, the weaker its conjugate base. When an acid reacts with a base that is farther down the list, the reaction proceeds to the right (Kc > 1). 18 -28

Sample Problem 18. 5 PROBLEM: Predicting the Net Direction of an Acid-Base Reaction Predict the net direction and whether Kc is greater or less than 1 for each of the following reactions (assume equal initial concentrations of all species): (a) H 2 PO 4–(aq) + NH 3(aq) (b) H 2 O(l) + HS–(aq) PLAN: HPO 42–(aq) + NH 4+(aq) OH–(aq) + H 2 S(aq) We identify the conjugate acid-base pairs and consult figure 18. 8 to see which acid and base are stronger. The reaction favors the formation of the weaker acid and base. SOLUTION: (a) H 2 PO 4–(aq) + NH 3(aq) stronger acid stronger base HPO 42–(aq) + NH 4+(aq) weaker base weaker acid The net direction for this reaction is to the right, so Kc > 1. (b) H 2 O(l) + weaker acid HS–(aq) OH–(aq) + weaker base stronger base H 2 S(aq) stronger acid The net direction for this reaction is to the left, so Kc < 1 18 -29

Solving Problems Involving Weak-Acid Equilibria The notation system Problem-solving approach • Molar concentrations are indicated by [ ]. 1. Write a balanced equation. 2. Write an expression for Ka. 3. Define x as the change in concentration that occurs during the reaction. 4. Construct a reaction table in terms of x. • [H 3 O+] from the autoionization of H 2 O is negligible. 5. Make assumptions that simplify the calculation. • A weak acid has a small Ka and its dissociation is negligible. [HA] ≈ [HA]init. 6. Substitute values into the Ka expression and solve for x. 7. Check that the assumptions are justified. 18 -30 • A bracketed formula with no subscript indicates an equilibrium concentration The assumptions

Sample Problem 18. 7 Finding Ka of a Weak Acid from the Solution p. H PROBLEM: Phenylacetic acid (C 6 H 5 CH 2 COOH, simplified here as HPAc) builds up in the blood of persons with phenylketonuria, an inherited disorder that, if untreated, causes mental retardation and death. A study of the acid shows that the p. H of 0. 12 M HPAc is 2. 62. What is the Ka of phenylacetic acid? PLAN: We start with the balanced dissociation equation and write the expression for Ka. We assume that [H 3 O+] from H 2 O is negligible and use the given p. H to find [H 3 O+], which equals [PAc–] and [HPAc]dissoc. We assume that [HPAc] ≈ [HPAc]init because HPAc is a weak acid. SOLUTION: HPAc(aq) + H 2 O(l) Ka = [H 3 O+][PAc–] [HPAc] 18 -31 H 3 O+(aq) + PAc–(aq)

Sample Problem 18. 7 Concentration (M) HPAc(aq) + Initial Change Equilibrium 0. 12 –x 0. 12 – x H 2 O(l) - H 3 O+(aq) + PAc–(aq) 0 +x x [H 3 O+] = 10–p. H = 2. 4 x 10– 3 M which is >> 10– 7 (the [H 3 O+] from water) x ≈ 2. 4 x 10– 3 M ≈ [H 3 O+] ≈ [PAc–] [HPAc] = 0. 12 - x ≈ 0. 12 M (2. 4 x 10– 3) So Ka = = 4. 8 x 10– 5 0. 12 Checking the assumptions by finding the percent error in concentration: 1 x 10– 7 M + [H 3 O ] from H O = x 100 = 4 x 10– 3 % (< 5%; assumption is justified). 2. 4 x 10– 3 M – 3 [HPAc]dissoc = 2. 4 x 10 M x 100 = 2. 0 % (< 5%; assumption is justified). 0. 12 M 2 18 -32
![Sample Problem 18. 8 Determining Concentration from Ka and Initial [HA] PROBLEM: Propanoic acid Sample Problem 18. 8 Determining Concentration from Ka and Initial [HA] PROBLEM: Propanoic acid](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-33.jpg)
Sample Problem 18. 8 Determining Concentration from Ka and Initial [HA] PROBLEM: Propanoic acid (CH 3 CH 2 COOH, which we simplify as HPr) is a carboxylic acid whose salts are used to retard mold growth in foods. What is the [H 3 O+] of 0. 10 M HPr (Ka = 1. 3 x 10− 5)? PLAN: We write a balanced equation and the expression for Ka. We know [HPr]init but not [HPr] (i. e. , the concentration at equilibrium). We define x as [HPr]dissoc and set up a reaction table. We assume that, since HPr has a small Ka value, it dissociates very little and therefore [HPr] ≈ [HPr]init. SOLUTION: HPr(aq) + H 2 O(l) H 3 O+(aq) + Pr−(aq) Ka = [H 3 O+][Pr−] [HPr] 18 -33

Sample Problem 18. 8 Concentration (M) Initial Change Equilibrium HPr(aq) + H 2 O(l) 0. 10 -x - 0. 10 - x H 3 O+(aq) + Pr–(aq) - 0 +x - x x Since Ka is small, we will assume that x << 0. 10 and [HPr] ≈ 0. 10 M. Ka = 1. 3 x 10– 5 = [H 3 O+][Pr–] [HPr] ≈ x 2 0. 10 x ≈ √(0. 10)(1. 3 x 10– 5) = 1. 1 x 10– 3 M = [H 3 O+] 1. 1 x 10– 3 M Check: [HPr]diss = x 100 = 1. 1% (< 5%; assumption is justified. ) 0. 10 M 18 -34
![Concentration and Extent of Dissociation Percent HA dissociated = [HA]dissoc [HA]init x 100 As Concentration and Extent of Dissociation Percent HA dissociated = [HA]dissoc [HA]init x 100 As](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-35.jpg)
Concentration and Extent of Dissociation Percent HA dissociated = [HA]dissoc [HA]init x 100 As the initial acid concentration decreases, the percent dissociation of the acid increases. HA(aq) + H 2 O(l) H 3 O+(aq) + A–(aq) A decrease in [HA]init means a decrease in [HA]dissoc = [H 3 O+] = [A–], causing a shift toward the products. The fraction of ions present increases, even though the actual [HA]dissoc decreases. 18 -35

Sample Problem 18. 9 Finding the Percent Dissociation of a Weak Acid PROBLEM: In 2011, researchers showed that hypochlorous acid (HCl. O) generated by white blood cells kills bacteria. Calculate the percent dissociation of (a) 0. 40 M HCl. O; (b) 0. 035 M HCl. O (Ka = 2. 9 x 10– 8). PLAN: Percent dissociation of HCl. O = [HCl. O]dissoc x 100 [HCl. O]init The value of [HCl. O]init is given. To calculate [HCl. O]dissoc, we write the balanced equation for the dissociation of HCl. O, set up a reaction table with x = [HCl. O]dissoc = [Cl. O–] = [H 3 O+], and use the expression for Ka. We assume that HCl. O dissociates to a very small extent since Ka is small (2. 9 x 10– 8). SOLUTION: (a) Writing the balanced equation and the expression for Ka: HCl. O(aq) + H 2 O(l) Ka = 18 -36 H 3 O+(aq) + Cl. O– (aq) [H 3 O+][Cl. O–] [HCl. O] = 2. 9 x 10– 8

Sample Problem 18. 9 Concentration (M) HCl. O(aq) + H 2 O(l) Initial Change Equilibrium 0. 40 –x 0. 40 – x - H 3 O+(aq) + Cl. O–(aq) - 0 +x - x x Since Ka is small, we will assume that x << 0. 40 and [HCl. O] ≈ 0. 40 M. Ka = [H 3 O+][Cl. O–] [HCl. O] = 2. 9 x 10– 8 = x 2 0. 40 x ≈ √ (0. 40)(2. 9 x 10– 8) = 1. 1 x 10– 4 M = [HCl. O]dissoc % Dissociation = 18 -37 1. 1 x 10– 4 M 0. 40 M x 100 = 0. 028% (< 5%; assumption is justified. )
![Sample Problem 18. 9 (b) Performing the same calculations using [HCl. O]init = 0. Sample Problem 18. 9 (b) Performing the same calculations using [HCl. O]init = 0.](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-38.jpg)
Sample Problem 18. 9 (b) Performing the same calculations using [HCl. O]init = 0. 035 M: Ka = [H 3 O+][Cl. O–] [HCl. O] = 2. 9 x 10– 8 = x 2 0. 035 x ≈ √(0. 035)(2. 9 x 10– 8)= 3. 2 x 10– 5 M = [HCl. O]dissoc % Dissociation = 18 -38 3. 2 x 10– 5 M 0. 035 M x 100 = 0. 091% (< 5%; assumption is justified. )

Polyprotic Acids A polyprotic acid is an acid with more than one ionizable proton. In solution, each dissociation step has a different value for Ka: H 3 PO 4(aq) + H 2 O(l) H 2 PO 4 HPO 4 –(aq) 2–(aq) + H 2 O(l) H 2 PO 4 –(aq) HPO 4 2–(aq) PO 4 3–(aq) + H 3 O+(aq) Ka 1 = Ka 2 = Ka 3 = [H 3 O+][H 2 PO 4–] [H 3 PO 4] [H 3 O+][HPO 42–] [H 2 PO 4–] [H 3 O+][PO 43–] [HPO 4 2–] = 7. 2 x 10– 3 = 6. 3 x 10– 8 = 4. 2 x 10– 13 Ka 1 >> Ka 2 >> Ka 3 We usually neglect [H 3 O+] produced after the first dissociation. 18 -39

Table 18. 5 18 -40 Successive Ka values for Some Polyprotic Acids at 25°C

Sample Problem 18. 10 Calculating Equilibrium Concentrations for a Polyprotic Acid PROBLEM: Ascorbic acid (H 2 C 6 H 6 O 6; H 2 Asc for this problem), known as vitamin C, is a diprotic acid (Ka 1 = 1. 0 x 10– 5 and Ka 2 = 5 x 10– 12) found in citrus fruit. Calculate [H 2 Asc], [HAsc–], [Asc 2–], and the p. H of 0. 050 M H 2 Asc. PLAN: We first write the dissociation equations and the associated Ka expressions. Since Ka 1 >> Ka 2, we can assume that the first dissociation produces almost all the H 3 O+. Also, since Ka 1 is small, the amount of H 2 ASc that dissociates can be neglected. We set up a reaction table for the first dissociation, with x equal to [H 2 Asc]dissoc, and solve for [H 3 O+] and [HAsc-]. SOLUTION: H 2 Asc(aq) + H 2 O(l) HAsc–(aq) + H 2 O(l) 18 -41 Hasc–(aq) + H 3 O+(aq) Asc 2–(aq) + H 3 O+(aq) Ka 1 = Ka 2 = [HAsc–][H 3 O+] [H 2 Asc] [Asc 2–][H 3 O+] [HAsc–] = 1. 0 x 10 -5 = 5 x 10 -12

Sample Problem 18. 10 Concentration (M) H 2 Asc(aq) + H 2 O(l) Initial Change Equilibrium 0. 050 –x - 0. 050 – x HAsc–(aq) + H 3 O+(aq) - 0 +x - x x x 2 [HAsc–][H 3 O+] – 5 ≈ Ka 1 = = 1. 0 x 10 = 0. 050 – x [H 2 Asc] x 2 0. 050 x = [H 3 O+] = [Asc–] = √(0. 050)(1. 0 x 10– 5) = 7. 1 x 10– 4 M p. H = –log[H 3 O+] = –log(7. 1 x 10– 4) = 3. 15 18 -42
![Sample Problem 18. 10 Checking assumptions: 1. [H 3 O+] from HAsc- << [H Sample Problem 18. 10 Checking assumptions: 1. [H 3 O+] from HAsc- << [H](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-43.jpg)
Sample Problem 18. 10 Checking assumptions: 1. [H 3 O+] from HAsc- << [H 3 O+] from H 2 Asc ≈ √ [HAsc–](Ka 2) HAsc- This is even less than [H 3 O+] : For any second dissociation, = √ (7. 1 x 10– 4)(5 x 10– 12) from H 2 O , so the assumption is justifed. 2. [H 2 Asc]dissoc << [H 2 Asc]init: 7. 1 x 10– 4 M x 100 = 1. 4% (< 5%; assumption is justified). 0. 050 M 18 -43 = 6 x 10– 8 M

Weak Bases A Brønsted-Lowry base is a species that accepts an H+. For a weak base that dissolves in water: B(aq) + H 2 O(l) BH+(aq) + OH–(aq) The base-dissociation or base-ionization constant is given by: [BH+][OH–] Kb = [B] Note that no base actually dissociates in solution, but ions are produced when the base reacts with H 2 O. 18 -44
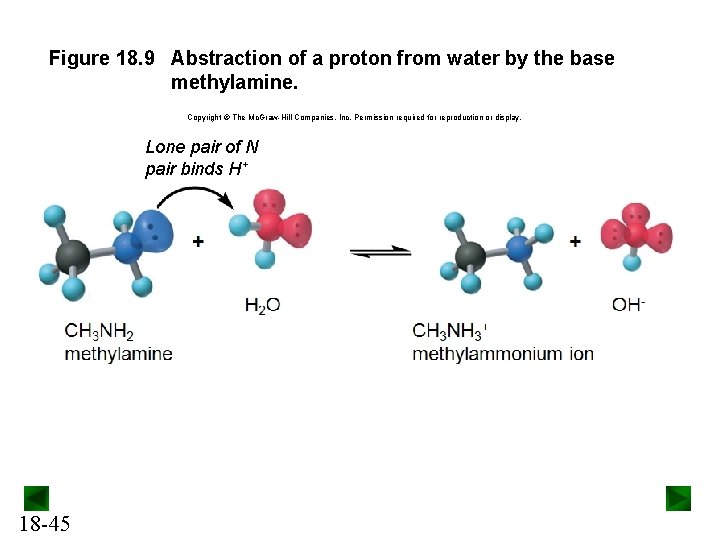
Figure 18. 9 Abstraction of a proton from water by the base methylamine. Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. Lone pair of N pair binds H+ 18 -45

Table 18. 6 18 -46 Kb Values for Some Molecular (Amine) Bases at 25°C
![Sample Problem 18. 11 Determining p. H from Kb and Initial [B] PROBLEM: Dimethylamine, Sample Problem 18. 11 Determining p. H from Kb and Initial [B] PROBLEM: Dimethylamine,](http://slidetodoc.com/presentation_image_h2/7b9c85573b1359269515e25da42b42a2/image-47.jpg)
Sample Problem 18. 11 Determining p. H from Kb and Initial [B] PROBLEM: Dimethylamine, (CH 3)2 NH, a key intermediate in detergent manufacture, has a Kb of 5. 9 x 10– 4. What is the p. H of 1. 5 M (CH 3)2 NH? PLAN: We start with the balanced equation for the reaction of the amine with H 2 O, remembering that it is a weak base. We then write the expression for Kb, set up a reaction table and solve for [OH–]. From [OH–] we can calculate [H 3 O+] and p. H. We make similar assumptions to those made for weak acids. Since Kb >> Kw, the [OH–] from H 2 O is negligible. Since Kb is small, we can assume that the amount of amine reacting is also small, so [(CH 3)2 NH] ≈ [(CH 3)2 NH]init. SOLUTION: (CH 3)2 NH(aq) + H 2 O(l) [(CH 3)2 NH 2+][OH–] Kb = [(CH 3)2 NH] 18 -47 (CH 3)2 NH 2+(aq) + OH–(aq)

Sample Problem 18. 11 Concentration (M) (CH 3)2 NH(aq) + H 2 O(l) Initial Change Equilibrium (CH 3)2 NH 2+(aq) + OH–(aq) 1. 5 -x - 0 +x 1. 5 - x x Since Kb is small, x << 1. 5 and 1. 5 – x ≈ 1. 5 Kb = [(CH 3)2 NH 2+][OH–] [(CH 3)2 NH] = 5. 9 x 10– 4 ≈ x 2 1. 5 x = [OH–] = 3. 0 x 10– 2 M Check assumption: 3. 0 x 10– 2 M x 100 = 2. 0% (< 5%; assumption is justified). 1. 5 M 18 -48
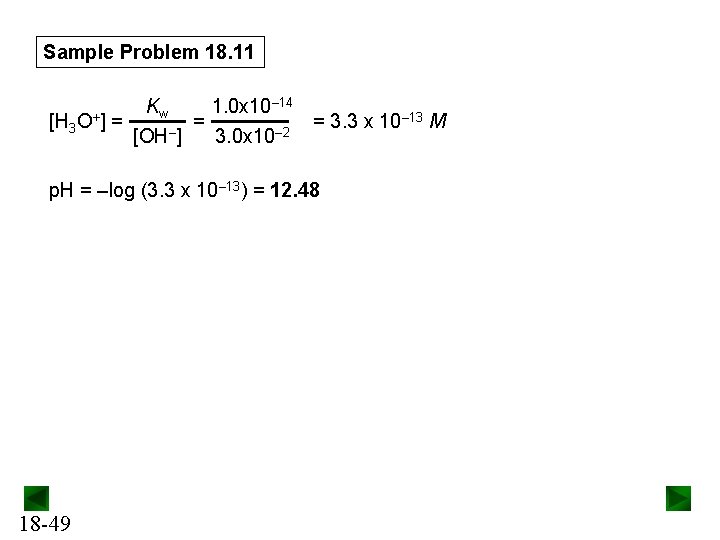
Sample Problem 18. 11 – 14 K 1. 0 x 10 w – 13 M [H 3 O+] = = = 3. 3 x 10 [OH–] 3. 0 x 10– 2 p. H = –log (3. 3 x 10– 13) = 12. 48 18 -49

Anions of Weak Acids as Weak Bases The anions of weak acids often function as weak bases. –] [HA][OH A-(aq) + H 2 O(l) HA(aq) + OH-(aq) Kb = [A–] A solution of HA is acidic, while a solution of A- is basic. HF(aq) + H 2 O(l) H 3 O+(aq) + F–(aq) HF is a weak acid (much weaker than H 3 O+), so this equilibrium lies to the left. [HF] >> [F–], but [H 3 O+]from HF >> [OH–] from H O; the solution is therefore acidic. 2 18 -50

If Na. F is dissolved in H 2 O, it dissolves completely, and Fcan act as a weak base: F–(aq) + H 2 O(l) HF(aq) + OH–(aq) Equilibrium favors the formation of the weaker acid and base. Since HF (although a weak acid) is a stronger acid than H 2 O, this equilibrium also lies to the left. [F–] >> [HF], but [OH–] from F >> [H 3 O+ ] from H O; the solution is therefore basic. - 18 -51 2
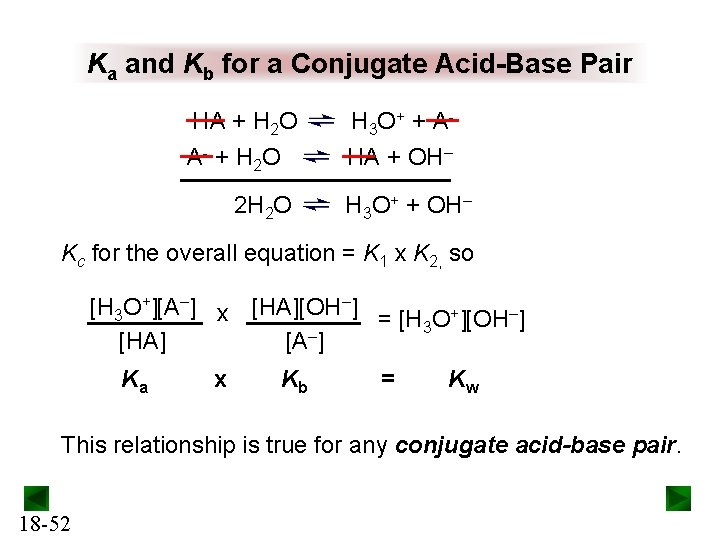
Ka and Kb for a Conjugate Acid-Base Pair HA + H 2 O H 3 O + + A - + H 2 O HA + OH– 2 H 2 O H 3 O+ + OH– Kc for the overall equation = K 1 x K 2, so [H 3 O+][A–] x [HA][OH–] = [H O+][OH–] 3 – [HA] [A ] Ka x Kb = Kw This relationship is true for any conjugate acid-base pair. 18 -52

Sample Problem 18. 12 Determining the p. H of a Solution of A- PROBLEM: Sodium acetate (CH 3 COONa, or Na. Ac for this problem) has applications in photographic development and textile dyeing. What is the p. H of 0. 25 M Na. Ac at 25ºC? Ka of acetic acid (HAc) is 1. 8 x 10– 5. PLAN: Sodium salts are soluble in water and acetate is the anion of HAc so it acts as a weak base. We write the base dissociation equation and the expression for Kb, and solve for [OH-]. We recall that any soluble ionic salt dissociates completely in solution, so [Ac–]init = 0. 25 M. SOLUTION: Ac–(aq) + H 2 O(l) HAc(aq) + OH–(aq) [HAc][OH–] Kb = [Ac–] 18 -53

Sample Problem 18. 12 Concentration (M) Initial Change Equilibrium Kb of Kb = Ac- = 5. 6 x 10 -10 Kw Ka Ac–(aq) + H 2 O(l) HAc(aq) + OH–(aq) 0. 25 -x - 0 +x 0. 25 - x x 1. 0 x 10– 14 – 10 M = = 5. 6 x 10 1. 8 x 10– 5 x 2 [HAc][OH–] ≈ = – [Ac ] 0. 25 so x = [OH–] = 1. 2 x 10– 5 M Checking the assumption: 1. 2 x 10– 5 x 100 = 4. 8 x 10 -3% (< 5%; assumption is justified) 0. 25 18 -54
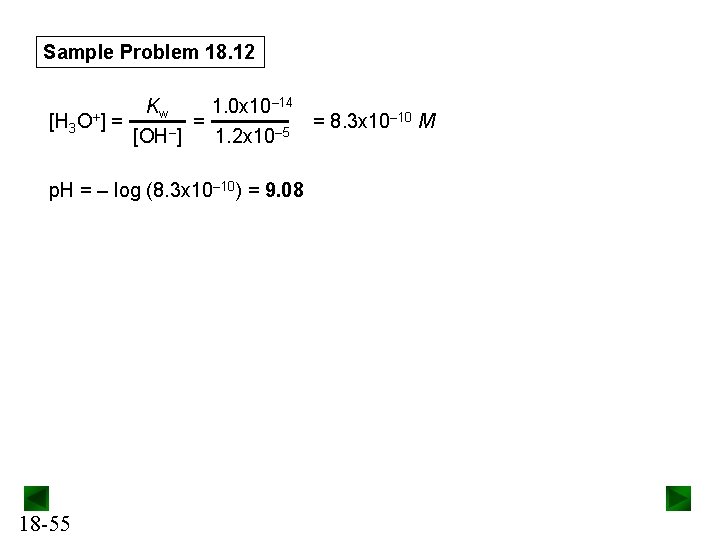
Sample Problem 18. 12 – 14 K 1. 0 x 10 w – 10 M [H 3 O+] = = = 8. 3 x 10 [OH–] 1. 2 x 10– 5 p. H = – log (8. 3 x 10– 10) = 9. 08 18 -55

Acid Strength of Nonmetal Hydrides For nonmetal hydrides (E-H), acid strength depends on: • the electronegativity of the central nonmetal (E), and • the strength of the E-H bond. Across a period, acid strength increases. Electronegativity increases across a period, so the acidity of E-H increases. Down a group, acid strength increases. The length of the E-H bond increases down a group and its bond strength therefore decreases. 18 -56
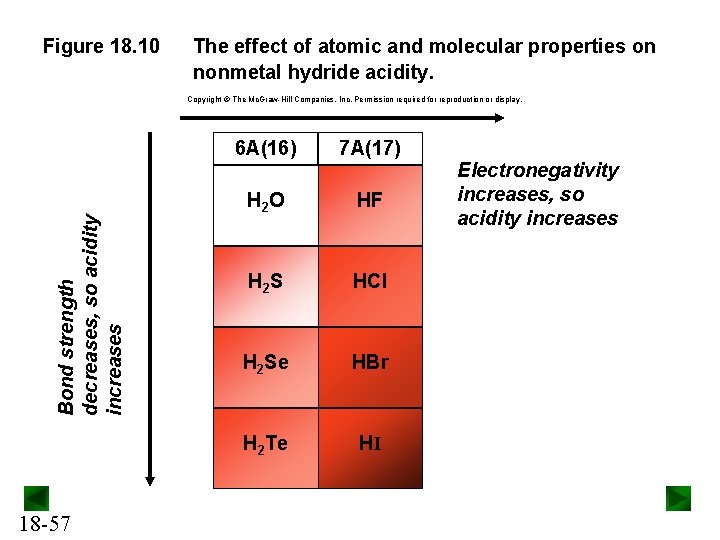
Figure 18. 10 The effect of atomic and molecular properties on nonmetal hydride acidity. Bond strength decreases, so acidity increases Copyright The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display. 18 -57 6 A(16) 7 A(17) H 2 O HF H 2 S HCl H 2 Se HBr H 2 Te HI Electronegativity increases, so acidity increases

Acid Strength of Oxoacids All oxoacids have the acidic H bonded to an O atom. Acid strength of oxoacids depends on: • the electronegativity of the central nonmetal (E), and • the number of O atoms around E. For oxoacids with the same number of O atoms, acid strength increases as the electronegativity of E increases. For oxoacids with different numbers of O atoms, acid strength increases with the number of O atoms. 18 -58
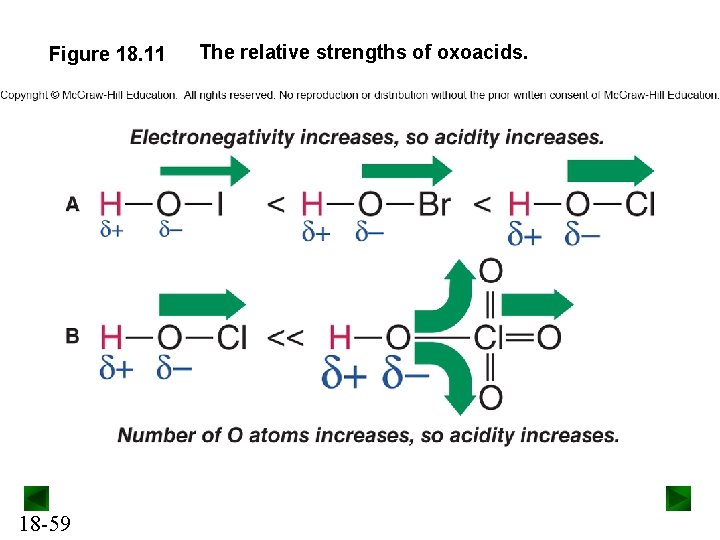
Figure 18. 11 18 -59 The relative strengths of oxoacids.

Hydrated Metal Ions Some hydrated metal ions are able to transfer an H+ to H 2 O. These metal ions will form acidic solutions. Consider a metal ion in solution, Mn+: Mn+(aq) + H 2 O(l) → M(H 2 O)xn+(aq) If Mn+ is small and highly charged, it will withdraw enough e - density from the O-H bonds of the bound H O molecules to 2 release H+: M(H 2 O)xn+(aq) + H 2 O(l) 18 -60 M(H 2 O)x-1 OH(n-1)+(aq) + H 3 O+(aq)
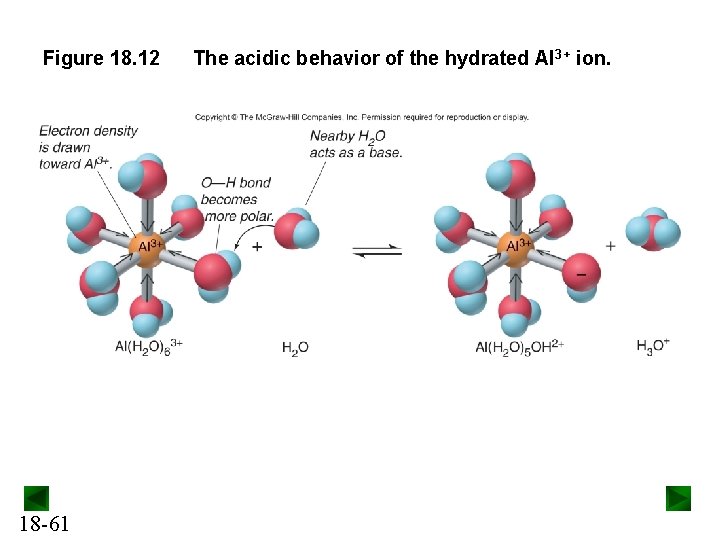
Figure 18. 12 18 -61 The acidic behavior of the hydrated Al 3+ ion.

Salts that Yield Neutral Solutions A salt that consists of the cation of a strong base and the anion of a strong acid yields a neutral solution. Na. NO 3 Na+ is the cation of Na. OH, a strong base. NO 3– is the anion of HNO 3, a strong acid. This solution will be neutral, because neither Na+ nor NO 3– will react with H 2 O to any great extent. 18 -62

Salts that Yield Acidic Solutions A salt that consists of the cation of a weak base and the anion of a strong acid yields an acidic solution. NH 4 Cl NH 4 + is the cation of NH 3, a weak base. Cl– is the anion of HCl, a strong acid. This solution will be acidic, because NH 4+ will react with H 2 O to produce H 3 O+: NH 4+(aq) + H 2 O(l) 18 -63 NH 3(aq) + H 3 O+(aq)

Salts that Yield Acidic Solutions A salt that consists of a small, highly charged metal cation and the anion of a strong acid yields an acidic solution. Fe(NO 3)3 Fe 3+ is a small, highly charged metal cation. NO 3 - is the anion of HNO 3, a strong acid. This solution will be acidic, because the hydrated Fe 3+ ion will react with H 2 O to produce H 3 O+: Fe(H 2 O)63+(aq) + H 2 O(l) 18 -64 Fe(H 2 O)5 OH 2+(aq) + H 3 O+(aq)

Salts that Yield Basic Solutions A salt that consists of the anion of a weak acid and the cation of a strong base yields a basic solution. CH 3 COONa CH 3 COO– is the anion of CH 3 COOH, a weak acid. Na+ is the cation of Na. OH, a strong base. This solution will be basic, because CH 3 COO– will react with H 2 O to produce OH–: CH 3 COO–(aq) + H 2 O(l) 18 -65 CH 3 COOH(aq) + OH–(aq)

Sample Problem 18. 13 Predicting Relative Acidity of Salt Solutions from Reactions of the Ions with Water PROBLEM: Predict whether aqueous solutions of the following are acidic, basic, or neutral, and write an equation for the reaction of any ion with water: (a) Potassium perchlorate, KCl. O 4 (b) Sodium benzoate, C 6 H 5 COONa (c) Chromium(III) nitrate, Cr(NO 3)3 PLAN: We identify the cation and anion from the formula for each salt. Depending on an ion’s ability to react with water, the solution will be neutral (strong-acid anion with strong-base cation or small, highly charged metal cation), acidic (weak-base cation with strong-acid anion), or basic (weak-acid anion and strongbase cation). SOLUTION: (a) K+ is the cation of a strong base (KOH) while Cl. O 4– is the anion of a strong acid (HCl. O 4). This solution will be neutral. 18 -66

Sample Problem 18. 13 (b) Na+ is the cation of a strong base (Na. OH) while the benzoate anion (C 6 H 5 COO–) is the anion of a weak acid (benzoic acid). The benzoate ion will react with H 2 O to produce OH– ions: C 6 H 5 COO–(aq) + H 2 O(l) C 6 H 5 COOH(aq) + OH– (aq) This solution will be basic. (c) NO 3– is the anion of a strong acid (HNO 3) and will not react with H 2 O to any great extent. Cr 3+ is a small metal cation with a fairly high charge density. It will become hydrated and the hydrated ion will react with H 2 O to form H 3 O+ ions: Cr(H 2 O)63+(aq) + H 2 O(l) This solution will be acidic. 18 -67 Cr(H 2 O)5 OH 2+(aq) + H 3 O+(aq)

Salts of Weakly Acidic Cations and Weakly Basic Anions If a salt that consists of the cation of a weak base and the anion of a weak acid, the p. H of the solution will depend on the relative acid strength or base strength of the ions. NH 4 CN NH 4+ is the cation of a weak base, NH 3. 18 -68 CN– is the anion of a weak acid, HCN. NH 4+(aq) + H 2 O(l) NH 3(aq) + H 3 O+(aq) CN–(aq) + H 2 O(l) HCN(aq) + OH– (aq)
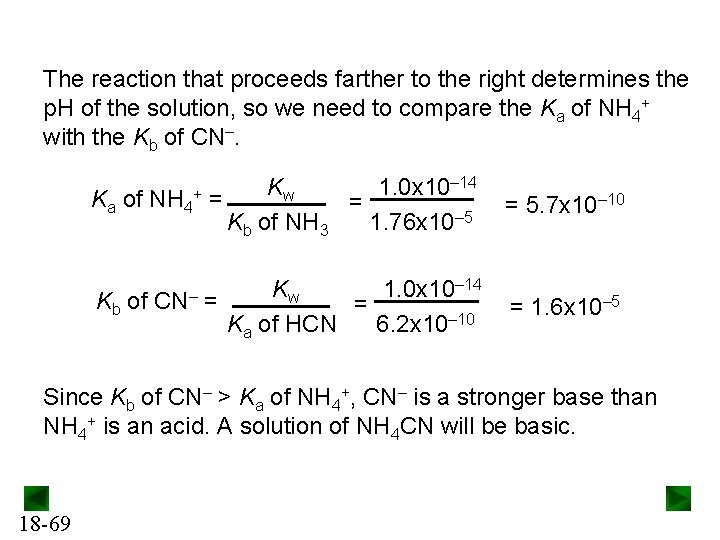
The reaction that proceeds farther to the right determines the p. H of the solution, so we need to compare the Ka of NH 4+ with the Kb of CN–. Ka of NH 4 Kb of + CN– Kw 1. 0 x 10– 14 = = Kb of NH 3 1. 76 x 10– 5 = 5. 7 x 10– 10 Kw 1. 0 x 10– 14 = = Ka of HCN 6. 2 x 10– 10 = 1. 6 x 10– 5 Since Kb of CN– > Ka of NH 4+, CN– is a stronger base than NH 4+ is an acid. A solution of NH 4 CN will be basic. 18 -69

Table 18. 7 18 -70 The Acid-Base Behavior of Salts in Water

Sample Problem 18. 14 Predicting the Relative Acidity of Salt Solutions from Ka and Kb of the Ions PROBLEM: Determine whether an aqueous solution of zinc formate, Zn(HCOO)2, at 25°C is acidic, basic, or neutral. PLAN: Zn 2+ is a small, highly charged metal cation, while HCOO– is the anion of a weak acid. Both will react with H 2 O, so to determine the acidity of the solution we must compare the Ka of the hydrated Zn 2+ ion with the Kb of the HCOO– ion. SOLUTION: Zn(H 2 O)62+(aq) + H 2 O(l) HCOO–(aq) + H 2 O(l) Zn(H 2 O)5 OH+(aq) + H 3 O+(aq) HCOOH(aq) + OH–(aq) Ka of Zn(H 2 O)62+ = 1 x 10– 9 (from Appendix C. ) Kw 1. 0 x 10– 14 – Kb of HCOO = = = 5. 6 x 10– 11 – 4 Ka of HCOOH 1. 8 x 10 Ka for Zn(H 2 O)62+ >> Kb HCOO–, therefore the solution is acidic. 18 -71

The Leveling Effect All strong acids and bases are equally strong in water. All strong acids dissociate completely to form H 3 O+, while all strong bases dissociate completely to form OH–. In water, the strongest acid possible is H 3 O+ and the strongest base possible is OH–. H 2 O exerts a leveling effect on any strong acid or base. 18 -72

The Lewis Acid-Base Definition A Lewis base is any species that donates an electron pair to form a bond. A Lewis acid is any species that accepts an electron pair to form a bond. The Lewis definition views an acid-base reaction as the donation and acceptance of an electron pair to form a covalent bond. 18 -73

Lewis Acids and Bases A Lewis base must have a lone pair of electrons to donate. Any substance that is a Brønsted-Lowry base is also a Lewis base. A Lewis acid must have a vacant orbital (or be able to rearrange its bonds to form one) to accept a lone pair and form a new bond. Many substances that are not Brønsted-Lowry acids are Lewis acids. The Lewis definition expands the classes of acids. 18 -74
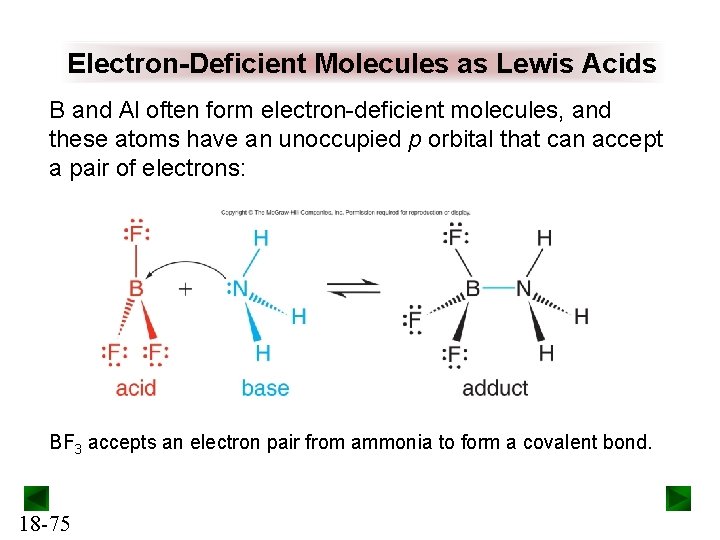
Electron-Deficient Molecules as Lewis Acids B and Al often form electron-deficient molecules, and these atoms have an unoccupied p orbital that can accept a pair of electrons: BF 3 accepts an electron pair from ammonia to form a covalent bond. 18 -75
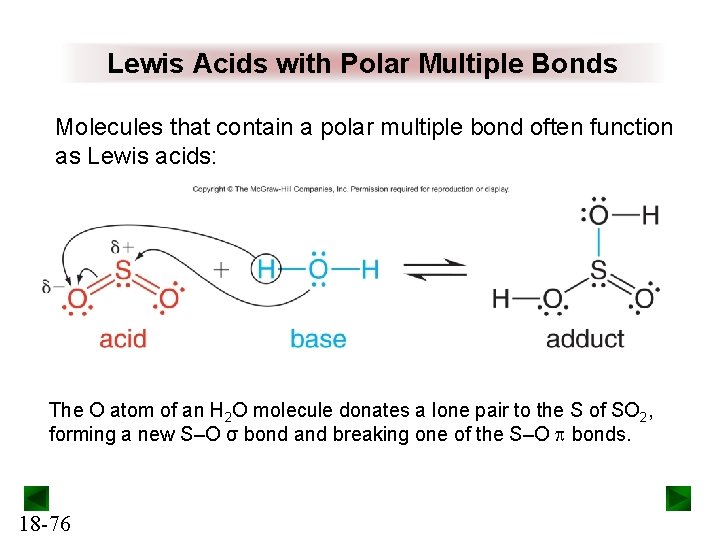
Lewis Acids with Polar Multiple Bonds Molecules that contain a polar multiple bond often function as Lewis acids: The O atom of an H 2 O molecule donates a lone pair to the S of SO 2, forming a new S‒O σ bond and breaking one of the S‒O p bonds. 18 -76
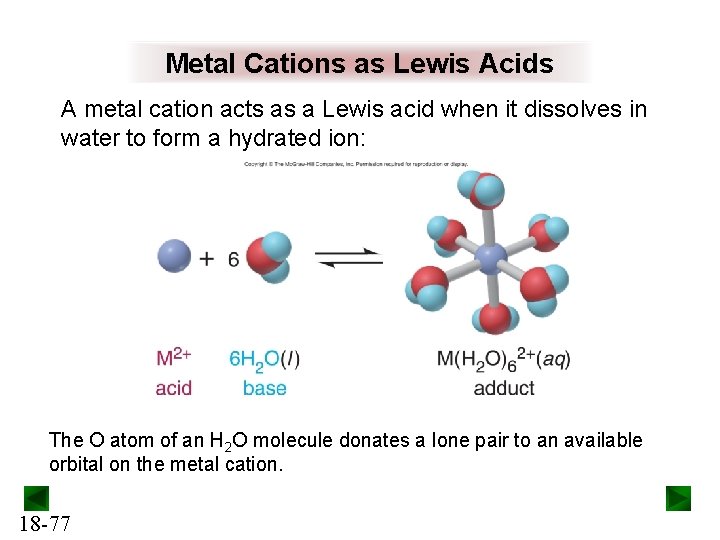
Metal Cations as Lewis Acids A metal cation acts as a Lewis acid when it dissolves in water to form a hydrated ion: The O atom of an H 2 O molecule donates a lone pair to an available orbital on the metal cation. 18 -77
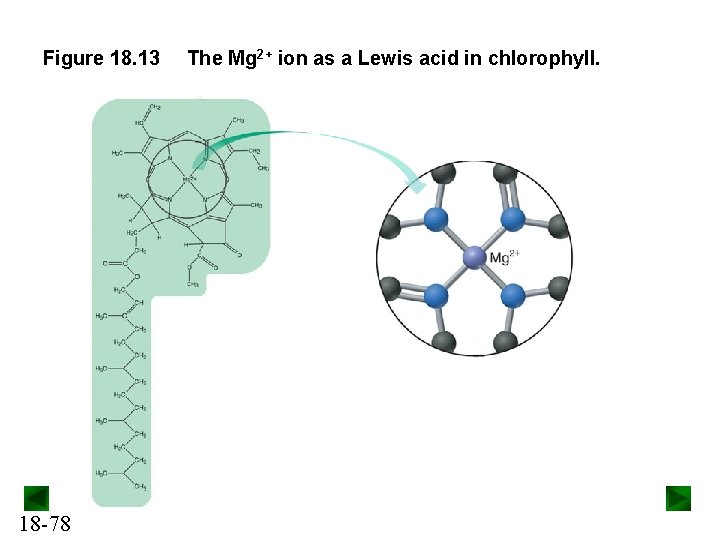
Figure 18. 13 18 -78 The Mg 2+ ion as a Lewis acid in chlorophyll.

Sample Problem 18. 15 Identifying Lewis Acids and Bases PROBLEM: Identify the Lewis acids and Lewis bases in the following reactions: (a) H+ + OH– H 2 O (b) Cl– + BCl 3 BCl 4– (c) K+ + 6 H 2 O K(H 2 O)6+ PLAN: We examine the formulas to see which species accepts the electron pair (Lewis acid) and which donates it (Lewis base) in forming the adduct. SOLUTION: (a) The H+ ion accepts the electron pair from OH–. H+ is the Lewis acid and OH– is the Lewis base. (b) BCl 3 accepts an electron pair from Cl– is the Lewis base and BCl 3 is the Lewis acid. (c) An O atom from each H 2 O molecule donates an electron pair to K+. H 2 O is therefore the Lewis base, and K+ is the Lewis acid. 18 -79
- Slides: 79