Chapter 17 THE FIRST LAW OF THERMODYNAMICS HEAT


















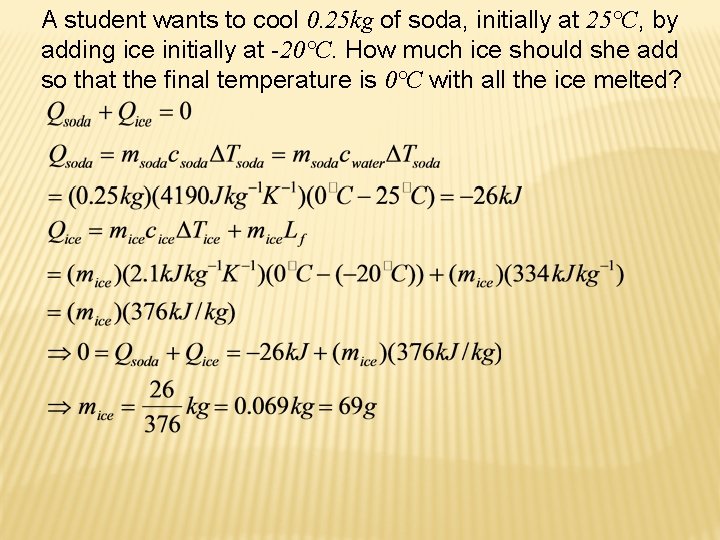



- Slides: 33

Chapter 17 THE FIRST LAW OF THERMODYNAMICS

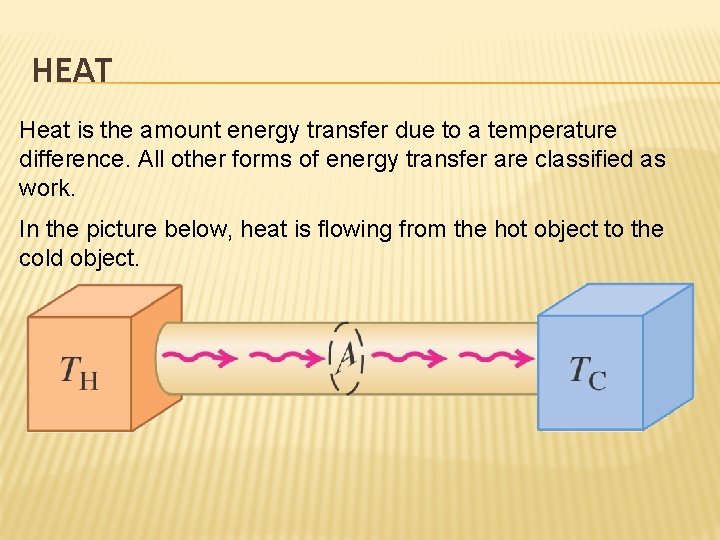
HEAT Heat is the amount energy transfer due to a temperature difference. All other forms of energy transfer are classified as work. In the picture below, heat is flowing from the hot object to the cold object.

UNIT OF HEAT People used to think that heat was not the same as energy. Therefore they invented a separate unit for heat, the calorie. 1 cal is defined as the heat required to raise the temperature of 1 g of water by 1°C. Terminology: cal: little cal Cal: big cal (this is the unit found on food labels)

ARE YOU ON DIET? Food Component Energy Density Cal/g Fat 9 Ethanol (Alcohol) 7 Proteins 4 Carbohydrates 4

UNITS OF HEAT

THE EQUIVALENCE BETWEEN HEAT AND ENERGY Joule proved that heat is equivalent to energy by the following experiment: He proved that one can raise the temperature of water by doing mechanical work (which involves no temperature difference or heating). He found it took 4. 186 J of energy to raise the temperature of 1 g of water by 1°C, hence 1 cal=4. 186 J.

SPECIFIC HEAT CAPACITY 1 cal is defined as the heat required to raise the temperature of 1 g of water by 1°C.

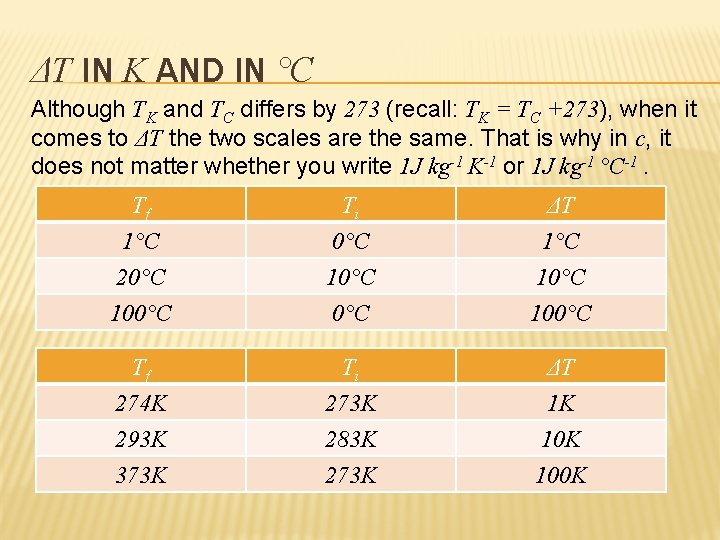
ΔT IN K AND IN °C Although TK and TC differs by 273 (recall: TK = TC +273), when it comes to ΔT the two scales are the same. That is why in c, it does not matter whether you write 1 J kg-1 K-1 or 1 J kg-1 °C-1. Tf 1°C 20°C 100°C Ti 0°C 10°C ΔT 1°C 100°C Tf 274 K 293 K 373 K Ti 273 K 283 K 273 K ΔT 1 K 100 K

MORE ON THE UNITS Make sure you know how to convert among the following units: Specific heat capacity is the amount of heat required to raise the temperature of 1 kg of a substance by 1 K.

EXAMPLE A 80 kg man has a fever of 39°C. How much heat is required to raise his temperature by that amount? [Normal temperature: 37°C, c =4190 Jkg-1 K-1]

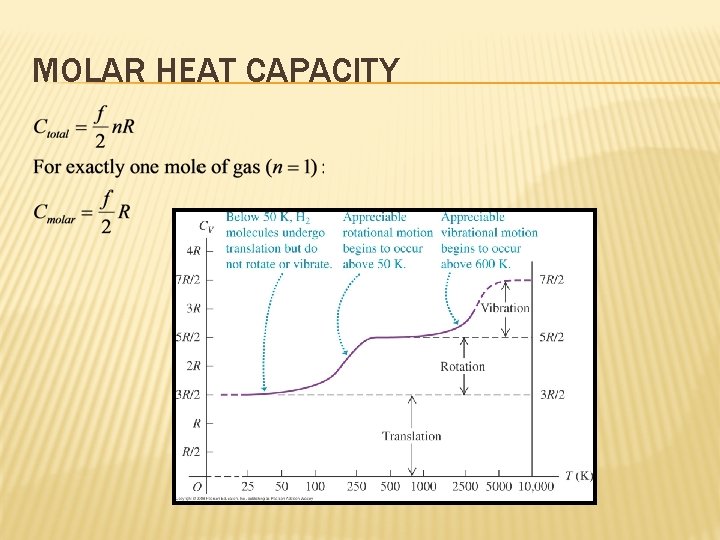
MOLAR HEAT CAPACITY First a reminder about moles: Define the molar mass M as the mass of one mole of substance, then n moles of the substances would have the mass of:

CHANGE OF PHASE AND LATENT HEAT There is no ΔT in the above equation, because there is no temperature change during phase change. Latent heat of fusion: Energy required to melt 1 kg of solid Latent heat of vaporization: Energy required to vaporize 1 kg of liquid Soli d Latent heat of fusion Latent heat of vaporization Liquid − + Ga s

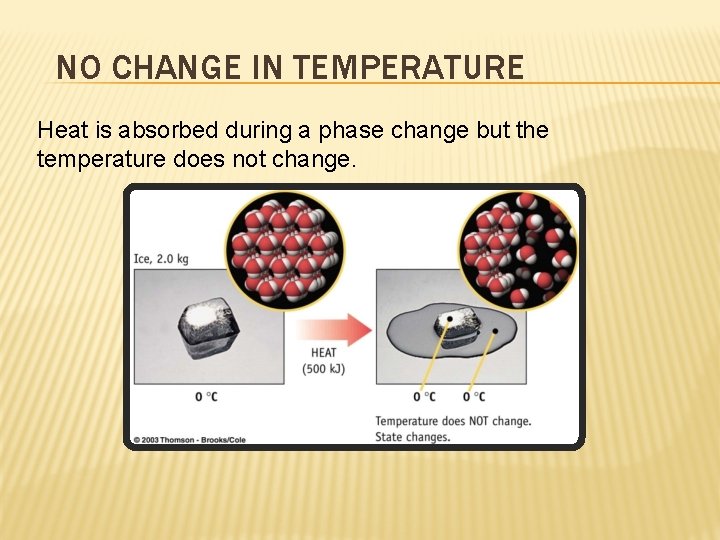
NO CHANGE IN TEMPERATURE Heat is absorbed during a phase change but the temperature does not change.

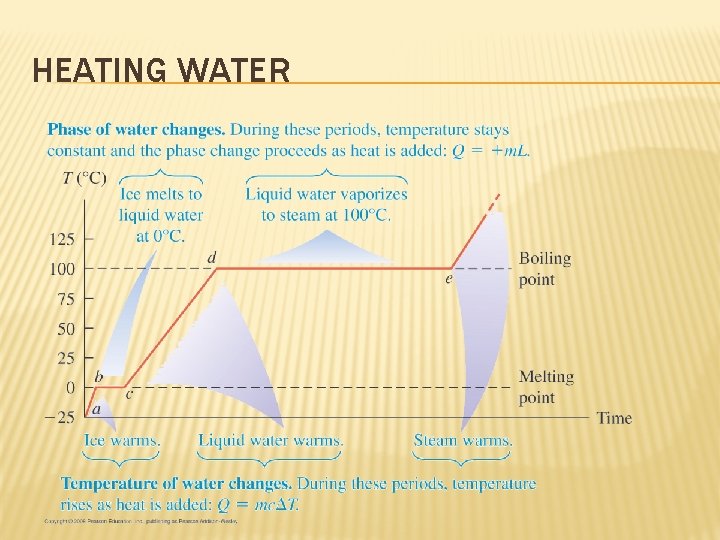
HEATING WATER

HEATING IN GENERAL

WATER: SPECIFIC HEAT CAPACITIES AND LATENT HEATS To warm ice (specific heat of ice) 2. 10 k. Jkg-1 K-1 To melt ice (latent heat of fusion) 334 k. Jkg-1 To warm water (specific heat of water) To vaporize water (latent heat of vaporization) To warm steam (specific heat of steam) 4. 19 k. Jkg-1 K-1 2260 k. Jkg-1 2. 02 k. Jkg-1 K-1

EXAMPLE Find the total amount of energy required to heat 3 g of water from -15°C to 155°C. To warm ice 3 g× 2. 10 k. Jkg-1 K-1× 15 K 94. 5 J To melt ice 3 g× 334 k. Jkg-1 1002 J 3 g× 4. 19 k. Jkg-1 K-1× 100 K 1257 J 3 g× 2260 k. Jkg-1 6780 J 3 g× 2. 02 k. Jkg-1 K-1× 55 K 333. 3 J To warm water To vaporize water To warm steam Total 9470 J

SODA IN AN ICE BOX Which will do a better job cooling your soda, a “cooler” filled with water at 0°C, or a cooler filled with ice at 0°C? A. Water B. Ice C. The same

HEAT TRANSFER When two objects are in thermal contact, the heat lost by one is the heat gain by the other by conservation of energy. For example, if QH =-150 J, then QC =+150 J. In general, we have: If one of the Q is positive, the other must be negative.

HEAT TRANSFER FOR MORE OBJECTS When there are more than two objects present, then the conservation law earlier generalizes to:

TYPICAL MIXING

IN GENERAL

EXAMPLE 2 kg of water at 300 K is mixed with 2 kg of water at 350 K. Find the final temperature.

EXAMPLE 1. 5 kg of X at temperature 400 K is dropped into 3 kg of water at 300 K. The final temperature is 360 K. Find the specific heat capacity of X.
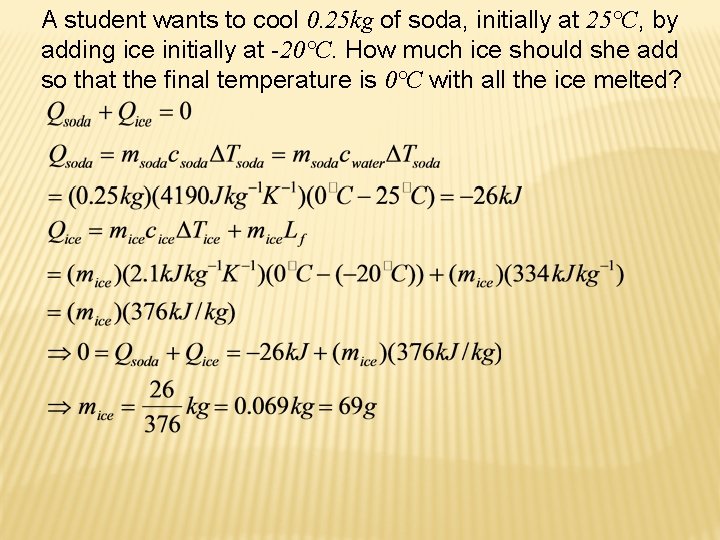
A student wants to cool 0. 25 kg of soda, initially at 25°C, by adding ice initially at -20°C. How much ice should she add so that the final temperature is 0°C with all the ice melted?

DEGREES OF FREEDOM AND EQUIPARTITION OF ENERGY Equipartition of energy: Each degree of freedom gives rise to a contribution of (1/2)k. T per particle. Degrees of freedom: Roughly speaking, the number of ways a particle can move.

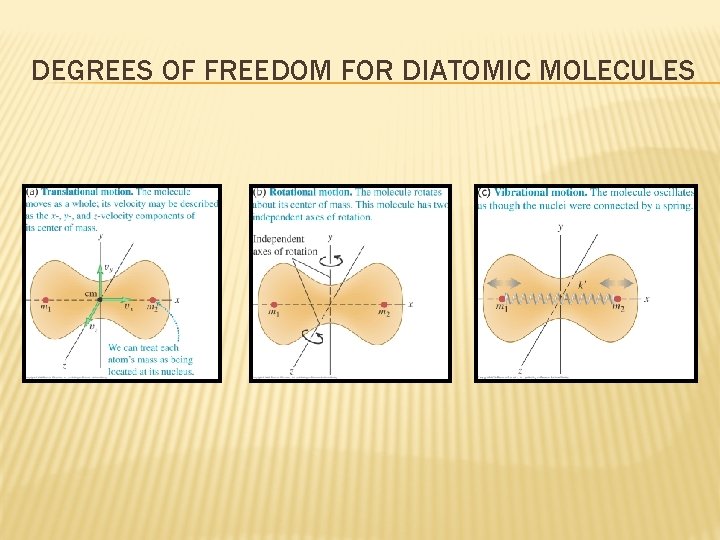
DEGREES OF FREEDOM FOR DIATOMIC MOLECULES

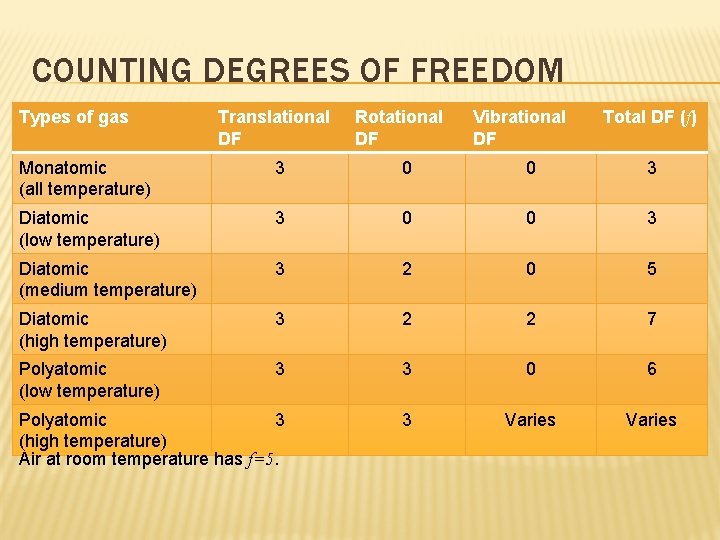
COUNTING DEGREES OF FREEDOM Types of gas Translational DF Rotational DF Vibrational DF Total DF (f) Monatomic (all temperature) 3 0 0 3 Diatomic (low temperature) 3 0 0 3 Diatomic (medium temperature) 3 2 0 5 Diatomic (high temperature) 3 2 2 7 Polyatomic (low temperature) 3 3 0 6 Polyatomic 3 (high temperature) Air at room temperature has f=5. 3 Varies

ENERGY OF A GAS What happens to U when T is changed? How much U is needed to change T by one unit? (This is the total heat capacity only when there is no work W)

MOLAR HEAT CAPACITY

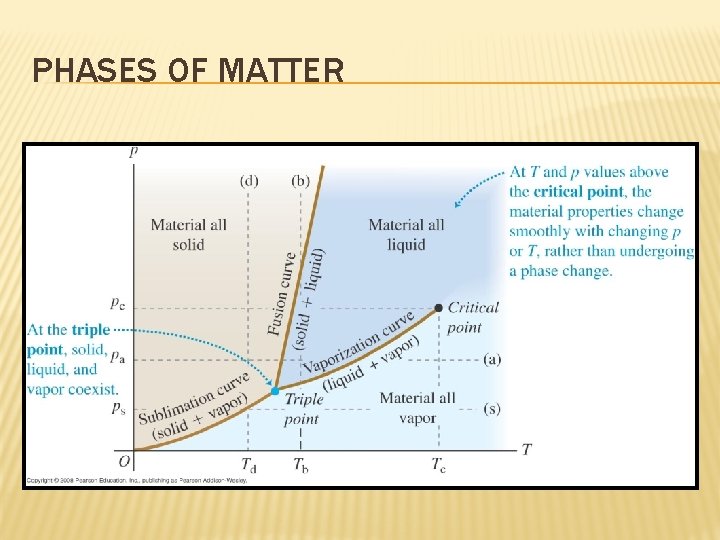
PHASES OF MATTER

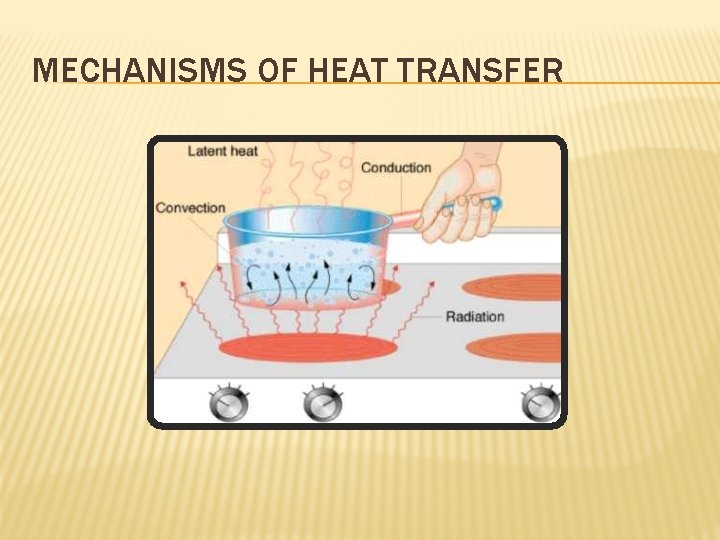
MECHANISMS OF HEAT TRANSFER

MORE EXAMPLES