Chapter 17 Nucleophilic Substitution Reaction of Alkyl Halides

Chapter 17 Nucleophilic Substitution Reaction of Alkyl Halides

Purpose • In this experiment eleven representative alkyl halides are treated with sodium iodide in acetone and with ethanolic solution of silver nitrate. In the first case (I/acetone) the SN 2 reaction is favored, while the second system (Ag+/Et. OH) effects the SN 1 reaction. In your analysis compare reactivity of the given alkyl halides in SN 2 and SN 1 reactions.
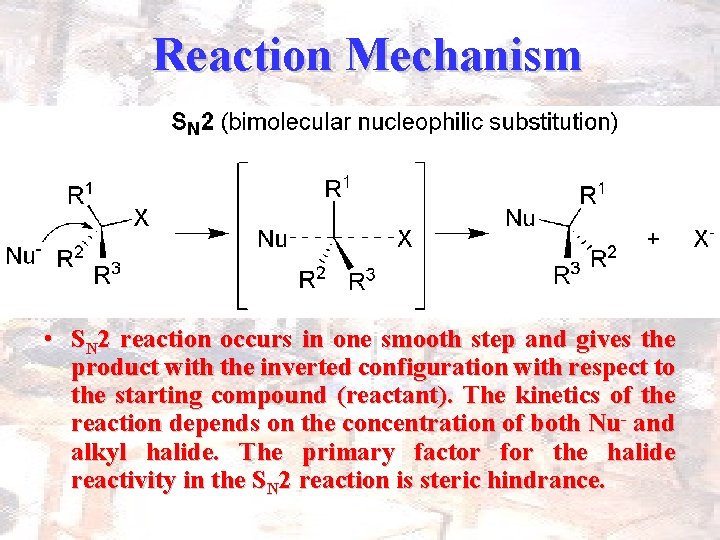
Reaction Mechanism • SN 2 reaction occurs in one smooth step and gives the product with the inverted configuration with respect to the starting compound (reactant). The kinetics of the reaction depends on the concentration of both Nu- and alkyl halide. The primary factor for the halide reactivity in the SN 2 reaction is steric hindrance.

Reaction Mechanism • SN 1 reaction occurs in two discrete steps and gives the product as an equimolar mixture of both enantiomers because the intermediate is planar. The kinetics of the reaction depends only on the concentration of halide. The primary factor for the halide reactivity in the SN 1 reaction is the relative stability of the intermediate carbocation.

Comments • Follow pp. 338 -340 (microscale). • Follow the lab handout for all experimental procedures. Wearing rubber gloves is necessary during this lab. You will work with harmful and toxic compounds. • Note the starting time when you mix the reagents together, and then record the time precipitate formed (if formed at all) in a test tube.

Safety • Flammable solvents and reagents are used in this experiment, keep away from any open flame. • Organic halides are flammable, and irritating to eyes, respiratory system and skin. Harmful by inhalation, may cause lung damage if swallowed. Do not swallow and inhale, avoid contact with skin and eyes.
- Slides: 6