Chapter 17 Aromatic Substitutions r electrophilic aromatic substitutions

Chapter 17 Aromatic Substitutions r electrophilic aromatic substitutions: E+ umechanism: uloss of aromaticity: higher DE, 673 Figure 17. 2 unitration: nitrobenzene (mixed acid); 673 bot. mformation Org. Chem-Chap 17 arenium ion, 672 Figure 17. 1 of a nitronium ion (+NO 2): 674 Figure 17. 3 1
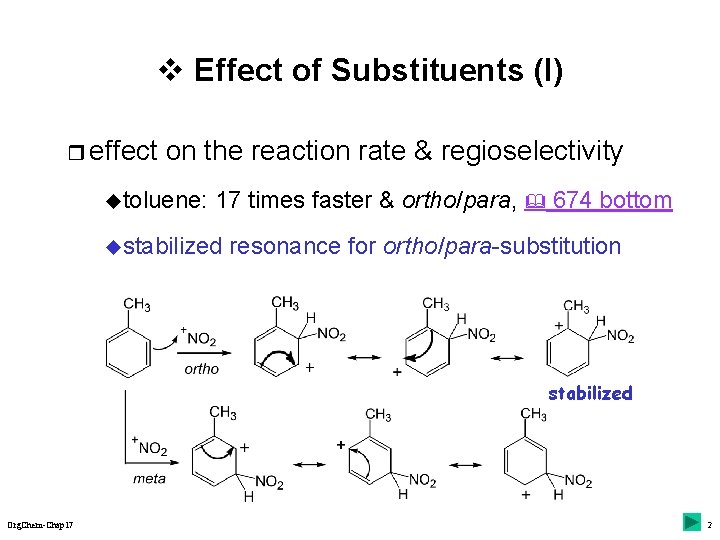
v Effect of Substituents (I) r effect on the reaction rate & regioselectivity utoluene: 17 times faster & ortho/para, 674 bottom ustabilized resonance for ortho/para-substitution stabilized Org. Chem-Chap 17 2
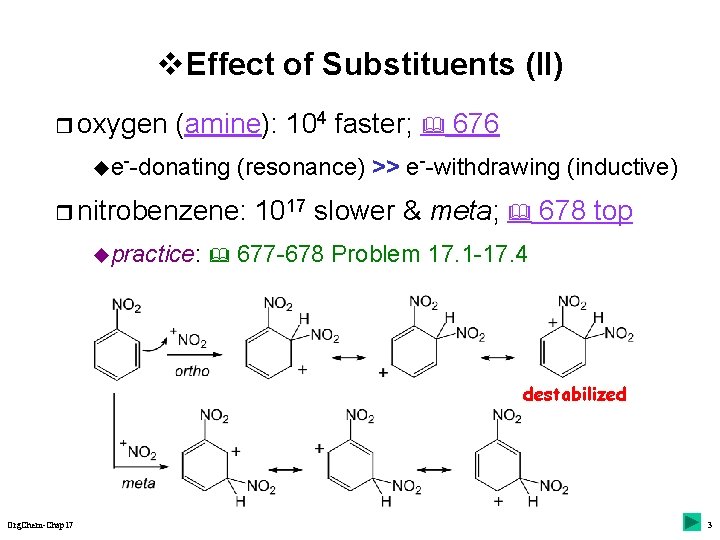
v Effect of Substituents (II) r oxygen (amine): 104 faster; 676 ue--donating (resonance) >> e--withdrawing (inductive) r nitrobenzene: upractice: 1017 slower & meta; 678 top 677 -678 Problem 17. 1 -17. 4 destabilized Org. Chem-Chap 17 3

v Effect of Substituents (III) r chlorine: 17 times slower & ortho/para, 679 ue--donating (resonance) < e--withdrawing (inductive) ugeneral r statistics utoluene r multiple substituent effect: 680 Table 17. 1 vs steric effect: 681 bottom (~1: 4) (~2: 1, 674); anisole (~1: 2, 676) substituents: dominating group, 682 upractice: Org. Chem-Chap 17 683 Problem 17. 7 -17. 8 4

v Nitration: Aniline Derivatives r nitrating reagents: HNO 3 & H+ 683 -684 u H+ : anhyd. HNO 3, H 2 SO 4 (mixed acid), CF 3 CO 2 H, CF 3 SO 3 H, NO 2+BF 4 -, N 2 O 4 / O 3 or O 2 (cat. ), Ac. OH ureduction r dinitration: r nitration to amine: aniline; 710 top vigorous conditions, 684 middle of aniline: ammonium ion, 684 bot. uprotection Org. Chem-Chap 17 to amides: activating, o-/p-, 685 bot. 5

v Halogenation / Sulfonation r less reactive arenes: formation of Cl+ or Br+ u. Lewis acid catalyst: Al. X 3 or Fe. X 3, 686 bottom r activated arenes: Cl 2 or Br 2 (no catalyst) umesitylene/aniline ucontrolled r sulfonation: ureversible: Org. Chem-Chap 17 (disubstitution): 687 bottom monosubstitution: 688 top SO 3, (H 2 SO 4); 689 top heating with H 3 O+; 689 bottom 6

v Friedel-Crafts Alkylation r carbocations (E+): alkyl halides/Lewis acids umechanism: mother SN 1 type, 690 -691 Figure 17. 4 carbocations: alcohols/alkenes & acids, 856 -7 limitations: multiple alkylation; no reaction with deactivating groups; rearrangement, 692 top u museful intramolecular alkylation: bicylics; 692 middle msynthetic detergents, BHT & BHA: 694 -6 Focus On upractice: Org. Chem-Chap 17 692 -3 Problem 17. 14, 17. 15 & 17. 16 7

v Friedel-Crafts Acylation r acylium ion (E+): mechanism, 696 bottom more useful: reaction with weakly deactivated arenes, no rearrangements, no multiple substitution u uanhydrides umore as well as acyl chlorides: 697 top para isomer: 697 bottom uintramolecular r electrophilic umore cyclization: polycyclics; 698 top substitution of polycyclic arenes reactive than benzene: 699 bottom uregioselectivity: Org. Chem-Chap 17 C-1 (kinetic) vs C-2 (thermodynamic) 8
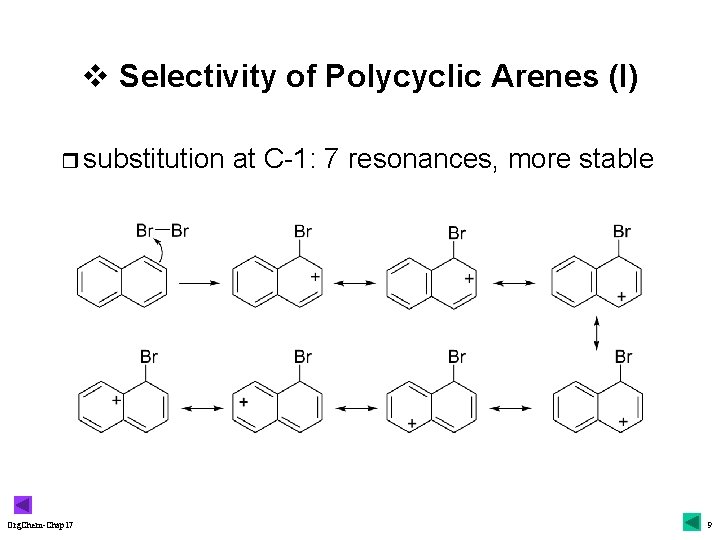
v Selectivity of Polycyclic Arenes (I) r substitution Org. Chem-Chap 17 at C-1: 7 resonances, more stable 9
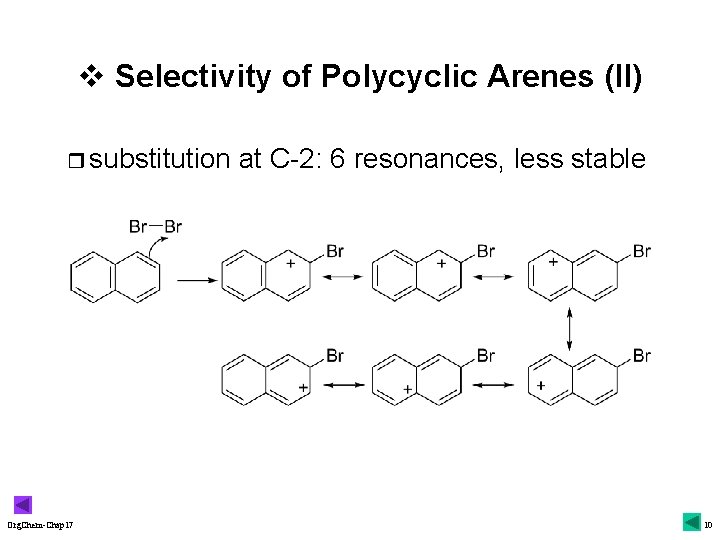
v Selectivity of Polycyclic Arenes (II) r substitution Org. Chem-Chap 17 at C-2: 6 resonances, less stable 10

v Nucleophilic Aromatic Substitution r diazonium ions: mechanism, 700 bottom usubstitution of N 2: I / F / OH / H; 701 -702 m. Sandmeyer reaction: Cu. X (X=Cl, Br, CN) r addition-elimination: u. EWG & rate (F > Cl > Br > I): 705 top r elimination-addition: u 707 Figure 17. 6 benzyne: 706 mid & 707 -9 Evidences upractice: Org. Chem-Chap 17 704 Figure 17. 5 702 -708 Problem 17. 23 -17. 26 11

v Synthesis of Aromatic Compounds r nitro (NO 2) amine (NH 2): 710 top CH 2: Clemmensen, 710 bot. / Wolff. Kishner, 711 top / catalytic H 2, 711 middle u. C=O museful r CHRR’ for arenes substituted with primary alkyl groups CO 2 H: 711 bottom & 712 top upractice: r summary 712 Problem 17. 27 -17. 28 of the reactions: 713 Figure 17. 7 upractice: m- / p-chloronitrobenzene, 714 top; obromophenol, 714 bot. ; m-bromochlorobenzene, 715 top; m-bromotoluene, 715 bottom Org. Chem-Chap 17 12
- Slides: 12