Chapter 17 Applications of Aqueous Equilibria By Ms

Chapter 17: Applications of Aqueous Equilibria By: Ms. Buroker

The Common Ion Effect Suppose we had a solution that contained the weak acid HF (Ka= 7. 2 x 10 -4) and its salt Na. F. What would be the major species in solution? HF, Na+, F-, H 2 O The common is this solution is F- because it comes from both the HF and the Na. F!!!

So … what effect does the presence of the Na. F have on the dissociation of HF? Let’s compare: 1. 0 M HF with (1. 0 M HF + 1. 0 M Na. F) According to Le. Chatelier’s principle we would expect the dissociation equilibrium for HF to be: Added F ions HF(aq) ↔ H+(aq) + F-(aq) from Na. F - Equilibrium shifts away from the added component.

Buffered Solutions A solution that resists a change in p. H when either hydroxide ions or protons are added. Buffered solutions contain either: – A weak acid and its salt – A weak base and its salt

Acid/Salt Buffering Pairs The salt will contain the anion of the acid, and the cation of a strong base (Na. OH, KOH) Weak Acid Formula Hydrofluoric acid HF HCN HC 2 H 3 O 2 Hydrocyanic acid Acetic Acid Example of salt of the weak acid KF Na. CN Mg(C 2 H 3 O 2)2

Base/Salt Buffering Pairs The salt will contain the cation of the base, and the anion of a strong acid (HCl, HNO 3) Weak Base Formula Ammonia NH 3 CH 3 NH 2 C 2 H 5 NH 2 Methylamine Example of salt of the weak base NH 4 Cl CH 3 NH 2 Cl C 2 H 5 NH 3 NO 3

Example #1 What is the p. H of a buffer solution composed of. 100 M propanic acid (HC 3 H 5 O 2; Ka=1. 3 x 10 -5) and. 100 M sodium propanate? To decide p. H, we need to figure out how the presence of the salt affects the acid’s equilibrium: HC 3 H 5 O 2 <=> H+ + C 3 H 5 O 2 -
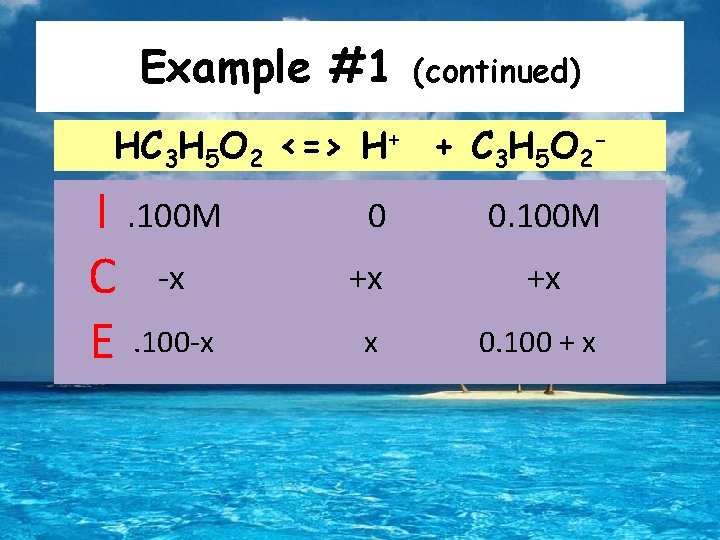
Example #1 (continued) HC 3 H 5 O 2 <=> H+ + C 3 H 5 O 2 - I. 100 M C -x E. 100 -x 0 0. 100 M +x +x x 0. 100 + x
![Example #1 (continued) +] [C H O -] [H 3 5 2 K= [HC Example #1 (continued) +] [C H O -] [H 3 5 2 K= [HC](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-9.jpg)
Example #1 (continued) +] [C H O -] [H 3 5 2 K= [HC 3 H 5 O 2] K= [x] [. 100+x] [. 100 -x] we will assume that the effect of “x” can be ignored (we will double check later) K = [x] = 1. 3 x 10 -5 p. H = -log[x] = 4. 88

How do buffers work? • The presence of the common ion from the conjugate helps moderate the equilibrium • More of the common ion shifts the equilibrium toward the non-dissociated side of the equilibrium • Adding more acid or base has a lesser effect on p. H because of this

Example #2 How does the addition of 0. 010 moles of solid Na. OH affect the p. H of 1 L of the buffer from the last example? HC 3 H 5 O 2 + OH- C 3 H 5 O 2 - + H 2 O • Stoichiometry Point of View • 0. 010 moles of the base will react with the same amount of the acid, and produce the same amount of the conjugate • Therefore we have: • 0. 100 -. 010 =. 090 moles of the acid remaining • 0. 100 +. 010 =. 110 moles of the conjugate present
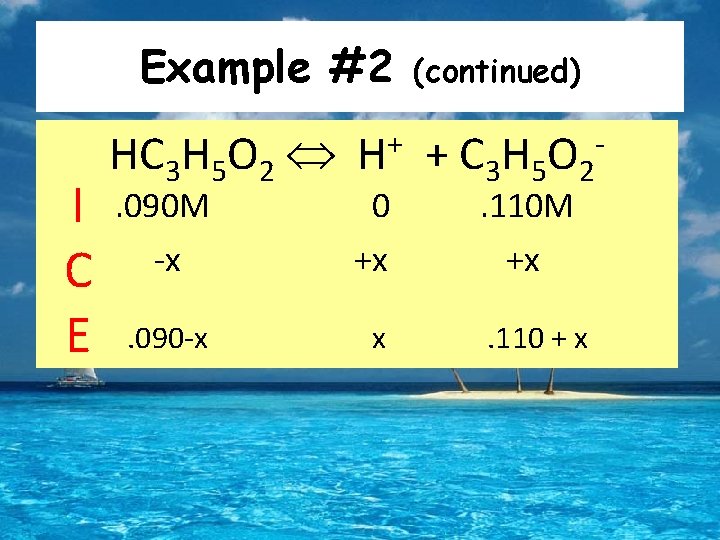
Example #2 I C E HC 3 H 5 O 2 + H . 090 M -x 0 +x . 090 -x x (continued) + C 3 H 5 O 2. 110 M +x . 110 + x -
![Example #2 +] [C H O -] [H 3 5 2 K= [HC 3 Example #2 +] [C H O -] [H 3 5 2 K= [HC 3](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-13.jpg)
Example #2 +] [C H O -] [H 3 5 2 K= [HC 3 H 5 O 2] (continued) K= [x] [. 110+x] [. 090 -x] we will assume that the effect of “x” can be ignored (we will double check later) 1. 3 x 10 -5 = [x] [. 110] [. 090] x = 1. 06 x 10 -5 p. H = -log[x] = 4. 97
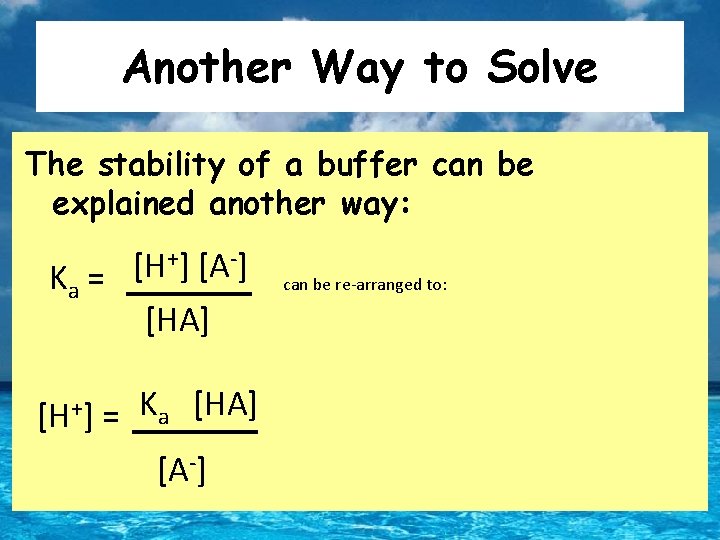
Another Way to Solve The stability of a buffer can be explained another way: +] [A-] [H Ka = [HA] [H+] = Ka [HA] [A-] can be re-arranged to:
![Another Way: Henderson- Hasselbalch Taking negative log of both sides gives: -log[H+] = -log(Ka) Another Way: Henderson- Hasselbalch Taking negative log of both sides gives: -log[H+] = -log(Ka)](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-15.jpg)
Another Way: Henderson- Hasselbalch Taking negative log of both sides gives: -log[H+] = -log(Ka) - log [HA] [A-] p. H = p. Ka + log [A-] [HA] p. H = p. Ka + log [base] Henderson-Hasselbalch Equation [acid]
![Example #2 (again) [base] p. H = p. Ka + log [acid] (. 110) Example #2 (again) [base] p. H = p. Ka + log [acid] (. 110)](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-16.jpg)
Example #2 (again) [base] p. H = p. Ka + log [acid] (. 110) p. H = p(1. 3 x 10 -5) + log (. 090) p. H = 4. 97

Example #3 A buffered solution contains. 50 M NH 3 (Kb=1. 8 x 10 -5)and. 30 M NH 4 Cl. What is its p. H? To solve H-H: Kw = (Ka)(Kb) 1 x 10 -14 = (Ka)(1. 8 x 10 -5) 5. 6 x 10 -10 = (Ka)
![Example #3 (continued) [base] p. H = p. Ka + log [acid] [. 50] Example #3 (continued) [base] p. H = p. Ka + log [acid] [. 50]](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-18.jpg)
Example #3 (continued) [base] p. H = p. Ka + log [acid] [. 50] p. H = p(5. 6 x 10 -10) + log [. 30] p. H = 9. 26 + (-. 22) = 9. 04

Acid- Base Titrations Titration: Quantitatively determining the concentration of a solution by reacting it with another solution of precisely known concentration. Terminology – Titrant: The solution of known concentration used to titrate a volume of the solution of unknown concentration. – Standardized (standard) solution: Solution of precisely known concentration – Equivalence point: The point at which stoichiometrically equivalent amounts of reactants have reacted – Endpoint: The point where an indicator (such as p. H) changes color.
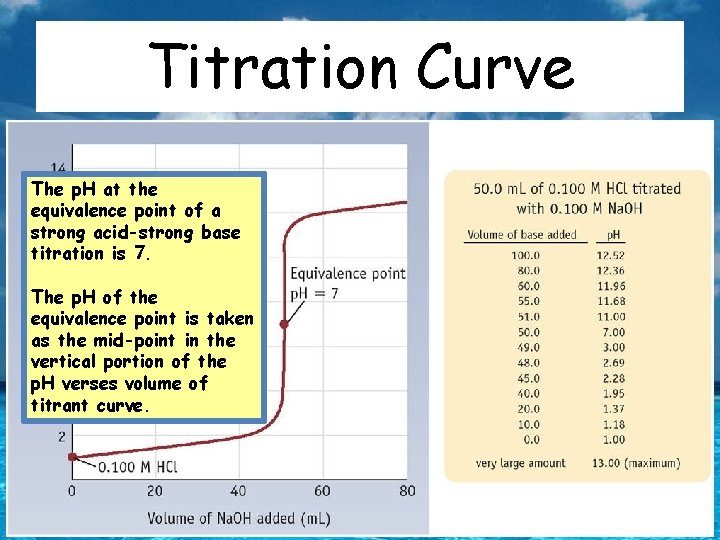
Titration Curve The p. H at the equivalence point of a strong acid-strong base titration is 7. The p. H of the equivalence point is taken as the mid-point in the vertical portion of the p. H verses volume of titrant curve.

Example What is the p. H after 25. 0 m. L of 0. 100 M Na. OH has been added to 50. 0 m. L of 0. 100 M HCl? What is the p. H after 50. 50 m. L of Na. OH has been added? 1. ) The first thing you need to do is find out how much of the acid will be left once the base has been added … you know all 25 m. L of the base will react.

Answer to First Part mol acid left: (0. 0500 L)(0. 100 M) – (. 0250 L)(0. 100 M) =. 0025 mol New [H 3 O+] =. 0025 mol/. 075 L =. 033 M p. H = -log(. 033) = 1. 48 Remember now that the base has been added to the acid … the volume has increased!! Draw a picture … it helps to keep things straight.

Now … back to the problem What is the p. H after 25. 0 m. L of 0. 100 M Na. OH has been added to 50. 0 m. L of 0. 100 M HCl? What is the p. H after 50. 50 m. L of Na. OH has been added? 2. ) Now you will notice that all of the acid has reacted and you have excess base remaining. So now you just have to find the hydroxide ion concentration and go from there.

Answer to Second Part Remember now that the base been added to At 50. 50 m. L of Na. OH all of the HClhas is neutralized the acid … the volume and an extra 0. 50 m. L of. 100 M Na. OH exists in a has increased!! Draw solution. picture … it helps to keep things straight. [OH-] = (0. 0005 L)(0. 100 M) /. 10050 L = 4. 975 x 10 -4 p. OH = -log(4. 975 x 10 -4) = 3. 303 p. H = 14. 0 – 3. 303 = 10. 7

Weak Acid- Strong Base Titrations *The p. H before the titration begins is given by the equilibrium expression using Ka of the weak acid. *The p. H at the equivalence point is dominated by the conjugate base (in the salt that forms) from the weak acid that was titrated. *The p. H at the halfway point (half-equivalence point) is given by: p. H = p. Ka + log([A-]/[HA]) *at the halfway point [A-] = [HA] and the log(1) = 0 Therefore p. H = p. Ka
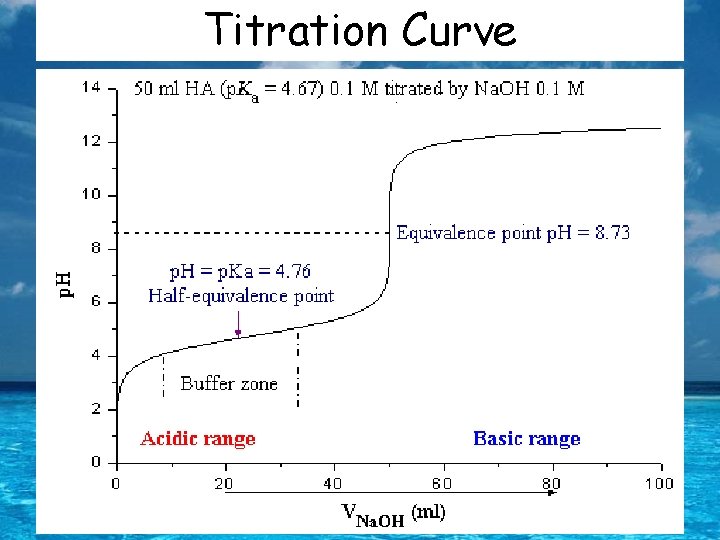
Titration Curve

Example What is the p. H of the solution when 35. 0 m. L of 0. 100 M Na. OH has been added to 100. 0 m. L of 0. 100 M acetic acid? Answer: HC 2 H 3 O 2 + Na. OH →Na. C 2 H 3 O 2 + H 2 O Moles Na. OH = (. 0350 L)(0. 100 M) =. 0035 mol Moles HC 2 H 3 O 2 = (. 1000 L)(0. 100 M) =. 0100 mol p. H = p. Ka + log ([C 2 H 3 O 2 -]/[HC 2 H 3 O 2]) p. H = 4. 74 + log (. 0259 M / 0. 0481 M)= 4. 47 Moles of acid minus the moles of base it reacted with divided by liters of solution.

Example Calculate the p. H after 75. 0 m. L of 0. 100 M HCl has been added to 100. 0 m. L of 0. 100 M NH 3 (Kb of NH 3 = 1. 8 x 10 -5) Answer: mol HCl = (. 0750 L)(0. 100 M) =. 00750 mol HCl initial moles NH 3 = (. 100 L)(0. 100 M) =. 0100 mol NH 3 Final: mol NH 4+ =. 00750 mol NH 3 =. 0100 -. 00750 =. 0025 mol p. OH = p. Kb + log([NH 4+]/[NH 3]) p. OH = 4. 74 + log(. 00750/. 0025) = 5. 22 p. H = 14. 00 – 5. 22 = 8. 78

Solubility of Salts For a given slightly soluble salt of the form: • Ax. By(s) ↔x. Ay+(aq) + y. Bx-(aq) • Ksp= [Ay+]x[Bx-]y • Where Ksp is the solubility product constant (equilibrium constant) for the salt (in aqueous solution) • Can be used to determine solubility (in mol/L or g/100 m. L H 2 O, etc. ) Molar solubility (mol/L) is the number of moles of solute dissolved in 1 L of a saturated solution. Solubility (g/L) is the number of grams of solute dissolved in 1 L of a saturated solution.
![Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Ag+ (aq) + Cl- (aq) Ksp Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Ag+ (aq) + Cl- (aq) Ksp](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-30.jpg)
Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Ag+ (aq) + Cl- (aq) Ksp is the solubility product constant Mg. F 2 (s) Mg 2+ (aq) + 2 F- (aq) Ag 2 CO 3 (s) 2 Ag+ (aq) + CO 32 - (aq) Ca 3(PO 4)2 (s) Ksp = [Mg 2+][F-]2 Ksp = [Ag+]2[CO 32 -] 3 Ca 2+ (aq) + 2 PO 43 - (aq) Ksp = [Ca 2+]3[PO 33 -]2 Dissolution of an ionic solid in aqueous solution: Q < Ksp Unsaturated solution Q = Ksp Saturated solution Q > Ksp Supersaturated solution No precipitate Precipitate will form

What is the solubility of silver chloride in g/L ? Ag+ (aq) + Cl- (aq) Ksp = 1. 6 x 10 -10 Initial (M) 0. 00 Ksp = [Ag+][Cl-] Change (M) +x +x Ksp = x 2 x x Ag. Cl (s) Equilibrium (M) [Ag+] = 1. 3 x 10 -5 M Solubility of Ag. Cl = x = 1. 3 x 10 -5 [Cl-] = 1. 3 x 10 -5 M 1. 3 x 10 -5 mol Ag. Cl 1 L soln x x = Ksp 143. 35 g Ag. Cl 1 mol Ag. Cl = 1. 9 x 10 -3 g/L

Example Knowing that the Ksp value for Mg. F 2 is 5. 2 x 10 -11, calculate the solubility of the salt in a) moles per liter and Reaction: b) grams per liter Mg. F 2 ↔ Mg+2(aq) + 2 F-(aq) Answer: Ksp = [Mg+2][F-]2 5. 2 x 10 -11 = [X][2 X]2 X = 2. 35 x 10 -4 = 2. 4 x 10 -4 mol/L(62. 3 g/mol) = 0. 015 g. Mg. F 2/L

If 2. 00 m. L of 0. 200 M Na. OH are added to 1. 00 L of 0. 100 M Ca. Cl 2, will a precipitate form? The ions present in solution are Na+, OH-, Ca 2+, Cl-. Only possible precipitate is Ca(OH)2 (solubility rules). Is Q > Ksp for Ca(OH)2? [Ca 2+]0 = 0. 100 M Q = [Ca 2+]0[OH-]0 = 4. 0 x 10 -4 M 2 = 0. 10 x (4. 0 x 10 -4)2 = 1. 6 x 10 -8 Ksp = [Ca 2+][OH-]2 = 8. 0 x 10 -6 Q < Ksp No precipitate will form

Solubility and the Common Ion Effect The effect of having an ion already in solution that is common to one of the ions in a substance being added to the solution (with a given Ksp) is to reduce the solubility of that substance. Think Le. Chatelier!!! Example: Calculate the solubility of Ba. SO 4 a) in pure water and b) in the presence of 0. 010 M Ba(NO 3)2. Ksp for Ba. SO 4 is 1. 1 x 10 -10.
![Answer a. ) Ksp = [Ba 2+][SO 42 -] so … 1. 1 x Answer a. ) Ksp = [Ba 2+][SO 42 -] so … 1. 1 x](http://slidetodoc.com/presentation_image_h/032313a1dff3199116e6b595e3c3108c/image-35.jpg)
Answer a. ) Ksp = [Ba 2+][SO 42 -] so … 1. 1 x 10 -10 = (x)(x) solving for x gives … x= 1. 0 x 10 -5 mol/L b. ) 1. 1 x 10 -10 = (x +. 010)(x) solving for x gives … x = 1. 1 x 10 -8 mol/L This comes from the common ion that was added to the solution of Ba. SO 4.

Complex Ion Equilibria and Solubility A complex ion is an ion containing a central metal cation bonded to one or more molecules or ions. Co 2+ (aq) + 4 Cl- (aq) Co(H 2 O)6 2+ Co. Cl 24 (aq)

16. 10

Complex Ion Formation • These are usually formed from a transition metal surrounded by ligands (polar molecules or negative ions). • As a "rule of thumb" you place twice the number of ligands around an ion as the charge on the ion. . . example: the dark blue Cu(NH 3)42+ (ammonia is used as a test for Cu 2+ ions), and Ag(NH 3)2+. • Memorize the common ligands.

Common Ligands Names used in the ion H 2 O NH 3 OHCl. Br. CNSCN- aqua ammine hydroxy chloro bromo cyano thiocyanato (bonded through sulfer) isothiocyanato (bonded through nitrogen)

Names • Names: ligand first, then cation Examples: – tetraamminecopper(II) ion: Cu(NH 3)42+ – diamminesilver(I) ion: Ag(NH 3)2+. – tetrahydroxyzinc(II) ion: Zn(OH)4 2 - • The charge is the sum of the parts (2+) + 4(-1)= -2.

When Complexes Form • Aluminum also forms complex ions as do some post transitions metals. Ex: Al(H 2 O)63+ • Transitional metals, such as Iron, Zinc and Chromium, can form complex ions. • The odd complex ion, Fe. SCN 2+, shows up once in a while • Acid-base reactions may change NH 3 into NH 4+ (or vice versa) which will alter its ability to act as a ligand. • Visually, a precipitate may go back into solution as a complex ion is formed. For example, Cu 2+ + a little NH 4 OH will form the light blue precipitate, Cu(OH)2. With excess ammonia, the complex, Cu(NH 3)42+, forms. • Keywords such as "excess" and "concentrated" of any solution may indicate complex ions. Ag. NO 3 + HCl forms the white precipitate, Ag. Cl. With excess, concentrated HCl, the complex ion, Ag. Cl 2 -, forms and the solution clears.

Coordination Number • Total number of bonds from the ligands to the metal atom. • Coordination numbers generally range between 2 and 12, with 4 (tetracoordinate) and 6 (hexacoordinate) being the most common.
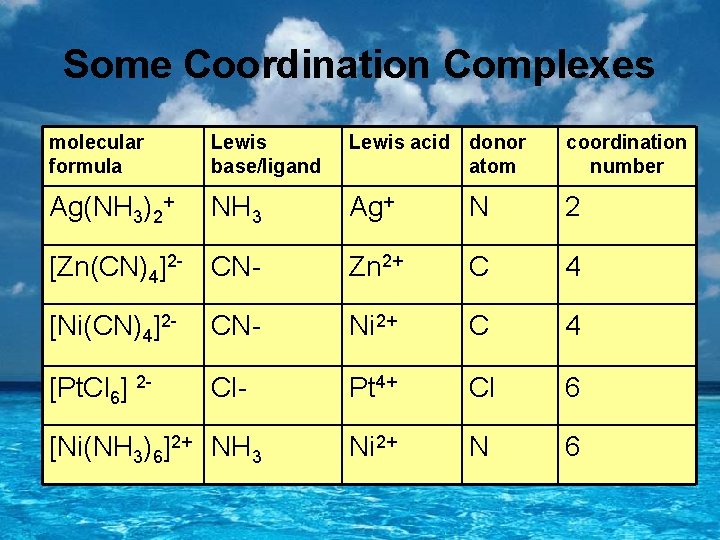
Some Coordination Complexes molecular formula Lewis base/ligand Lewis acid donor atom coordination number Ag(NH 3)2+ NH 3 Ag+ N 2 [Zn(CN)4]2 - CN- Zn 2+ C 4 [Ni(CN)4]2 - CN- Ni 2+ C 4 [Pt. Cl 6] 2 - Cl- Pt 4+ Cl 6 Ni 2+ N 6 [Ni(NH 3)6]2+ NH 3
- Slides: 43