Chapter 17 Additional Aspects of Aqueous Equilibria 1

Chapter 17 Additional Aspects of Aqueous Equilibria 1

• Various aqueous solutions encountered in nature typically contain many solutes. • Many equilibria can take place simultaneously in these solutions. • The understanding of complex solutions is a further application of acid-base equilibria. 2

17. 1 The Common-Ion Effect • Def: A shift of an equilibrium induced by an ion common to the equilibrium • Consider a solution of acetic acid: HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2−(aq) • If acetate ion is added to the solution, Le Châtelier says the equilibrium will shift to the left. • The dissociation of the weak acid HC 2 H 3 O 2 decreases when the strong electrolyte Na. C 2 H 3 O 2, which has an ion in common with it, is added. 3

• The extent of ionization of a weak electrolyte is decreased by adding to the solution a strong electrolyte that has an ion in common with the weak electrolyte. • Equilibrium concentrations may be calculated like those for weak acids and weak bases in Ch 16. 4

Example 1: What is the p. H of a solution made by adding 0. 30 mol of acetic acid (HC 2 H 3 O 2) and 0. 30 mol of sodium acetate (Na. C 2 H 3 O 2) to enough water to make 1. 0 L of solution? 5

Example 2: Calculate the fluoride ion concentration and p. H of a solution that is 0. 20 M in HF and 0. 10 M in HCl. Ka for HF is 6. 8 10− 4. [H 3 O+] [F−] = 6. 8 10 -4 Ka = [HF] 6

HF(aq) + H 2 O(l) H 3 O+(aq) + F−(aq) Because HCl, a strong acid, is also present, the initial [H 3 O+] is not 0, but rather 0. 10 M. [HF], M [H 3 O+], M [F−], M Initially 0. 20 0. 10 0 Change −x +x +x 0. 20 − x 0. 20 0. 10 + x 0. 10 x At Equilibrium 7

6. 8 x= 10− 4 (0. 10) (x) = (0. 20) (6. 8 10− 4) (0. 10) x = 1. 4 10− 3 • Therefore, [F−] = x = 1. 4 10− 3 M [H 3 O+] = 0. 10 + x = 0. 10 + 1. 4 10− 3 = 0. 10 M • So, p. H = −log (0. 10) p. H = 1. 00 8

17. 2 Buffered Solutions • Solutions of a weak conjugate acid-base pair. • They are particularly resistant to p. H changes, even when strong acid or base is added. 9

Composition and Action of Buffered Solutions • A buffer resists changes in p. H because it contains both an acidic species to neutralize OH- ions and a basic species to neutralize H+ ions. • The acidic and basic species must not consume themselves through neutralization. • These requirements are met by using a weak conjugate acid-base pair: 10
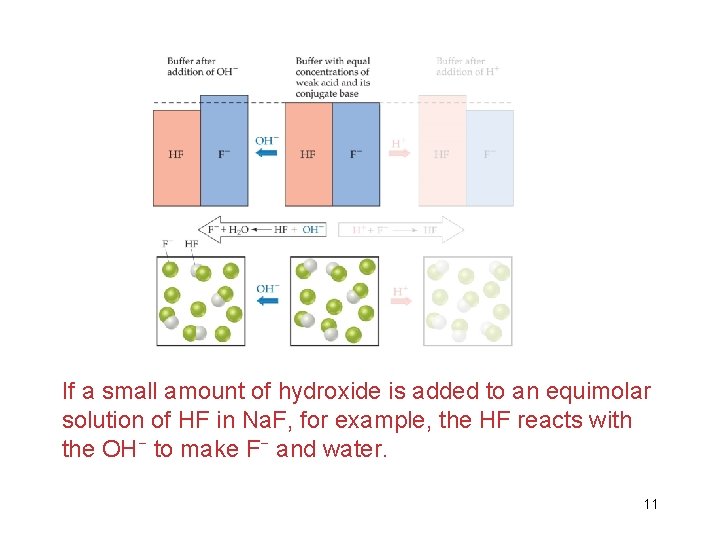
If a small amount of hydroxide is added to an equimolar solution of HF in Na. F, for example, the HF reacts with the OH− to make F− and water. 11
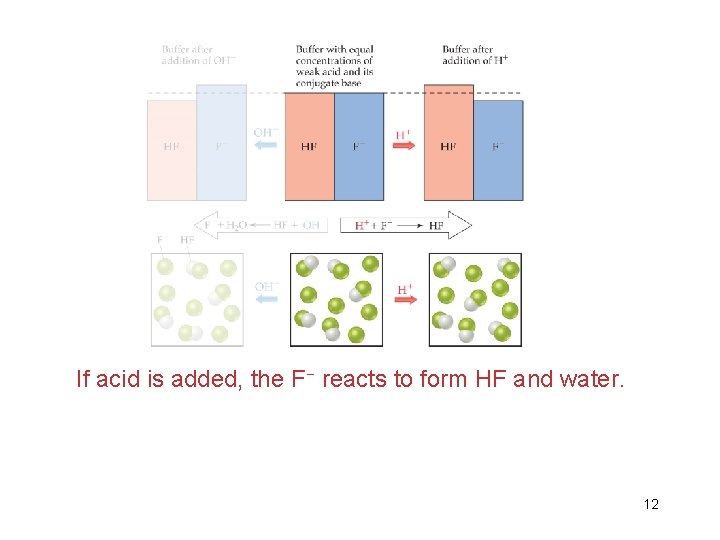
If acid is added, the F− reacts to form HF and water. 12

Calculating the p. H of a Buffer • Because conjugate acid-base pairs share a common ion, we can use the same procedures to calculate the p. H of a buffer that were used for th common-ion effect. • Consider the equilibrium constant expression for the dissociation of a generic acid, HA: HA + H 2 O H 3 O + + A − [H 3 O+] [A−] Ka = [HA] 13
![Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Taking the Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Taking the](http://slidetodoc.com/presentation_image_h2/6239c26e494f90591ea3b2cc675fdc9f/image-14.jpg)
Rearranging slightly, this becomes −] [A Ka = [H 3 O+] [HA] Taking the negative log of both side, we get −] [A base −log Ka = −log [H 3 O+] + −log [HA] p. Ka p. H acid 14
![• So [base] p. Ka = p. H − log [acid] • Rearranging, • So [base] p. Ka = p. H − log [acid] • Rearranging,](http://slidetodoc.com/presentation_image_h2/6239c26e494f90591ea3b2cc675fdc9f/image-15.jpg)
• So [base] p. Ka = p. H − log [acid] • Rearranging, this becomes [base] p. H = p. Ka + log [acid] • This is the Henderson–Hasselbalch equation. 15

Henderson–Hasselbalch Equation What is the p. H of a buffer that is 0. 12 M in lactic acid, HC 3 H 5 O 3, and 0. 10 M in sodium lactate? Ka for lactic acid is 1. 4 10− 4. [base] p. H = p. Ka + log [acid] (0. 10) p. H = −log (1. 4 10− 4) + log (0. 12) p. H = 3. 85 + (− 0. 08) p. H = 3. 77 16

Buffer Capacity and p. H Range • Buffer capacity is the amount of acid or base the buffer can neutralize before the p. H begins to change to an appreciable degree. • The greater the amounts of the conjugate acid -base pair, the more resistant the ratio of their concentrations, and hence the p. H, is to change. 17

• The p. H range is the range of p. H values over which a buffer system works effectively. • It is best to choose an acid with a p. Ka close to the desired p. H. 18
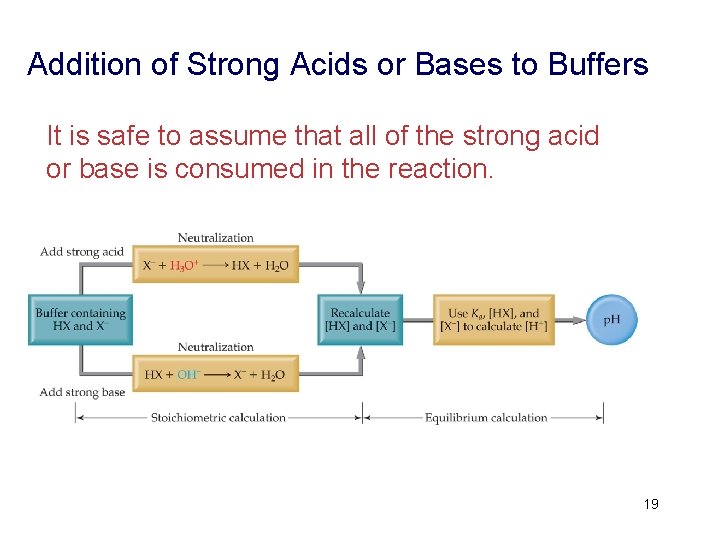
Addition of Strong Acids or Bases to Buffers It is safe to assume that all of the strong acid or base is consumed in the reaction. 19
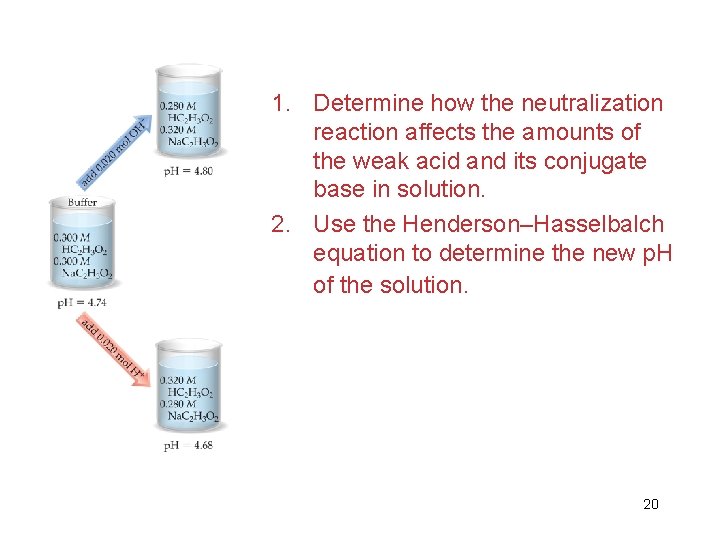
1. Determine how the neutralization reaction affects the amounts of the weak acid and its conjugate base in solution. 2. Use the Henderson–Hasselbalch equation to determine the new p. H of the solution. 20

Calculating p. H Change in a Buffer • A buffer is made by adding 0. 300 mol HC 2 H 3 O 2 and 0. 300 mol Na. C 2 H 3 O 2 to enough water to make 1. 00 L of solution. • The p. H of the buffer is 4. 74. • Calculate the p. H of this solution after 0. 020 mol of Na. OH is added. 21

Before the reaction, since mol HC 2 H 3 O 2 = mol C 2 H 3 O 2− p. H = p. Ka = −log (1. 8 10− 5) = 4. 74 22

Calculating p. H Changes in Buffers The 0. 020 mol Na. OH will react with 0. 020 mol of the acetic acid: HC 2 H 3 O 2(aq) + OH−(aq) C 2 H 3 O 2−(aq) + H 2 O(l) HC 2 H 3 O 2− OH− Before reaction 0. 300 mol 0. 020 mol After reaction 0. 280 mol 0. 320 mol 0. 000 mol 23

Now use the Henderson–Hasselbalch equation to calculate the new p. H: (0. 320) p. H = 4. 74 + log (0. 280) p. H = 4. 74 + 0. 06 p. H = 4. 80 Note: Read “Blood as a Buffered Solution” on page 731 24
- Slides: 24