Chapter 16 sustainability and Green Chemistry Sustainability Treat

Chapter 16: sustainability and Green Chemistry

Sustainability Treat the earth well: it was not given to you by your parents, it was loaned to you by your children. We do not inherit the Earth from our Ancestors, we borrow it from our Children. ~ Ancient American Indian Proverb

Sustainability Is the goal Meeting the needs of the present generation without compromising the needs of future generations
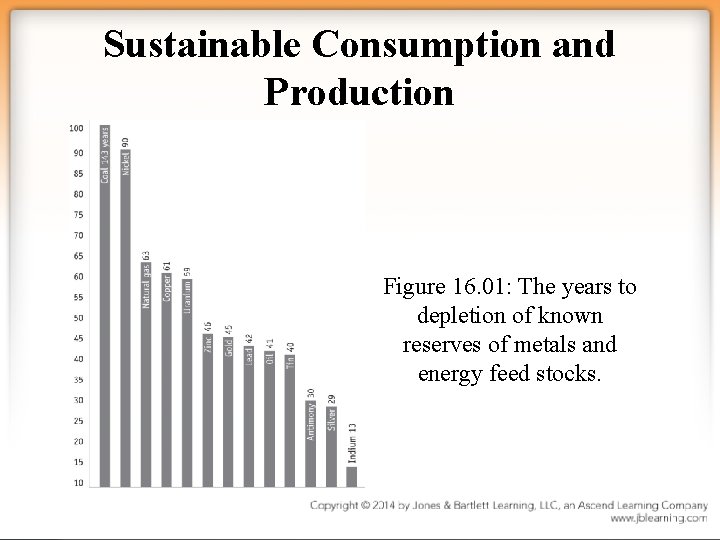
Sustainable Consumption and Production Figure 16. 01: The years to depletion of known reserves of metals and energy feed stocks.

Five Axioms of Sustainability • Axiom 1: Any society that continues to use critical resources unsustainably will collapse. • Axiom 2: Population growth and/or growth in the rates of consumption of resources cannot be sustained. • Axiom 3: To be sustainable, the use of renewable resources must proceed at a rate that is less than or equal to the rate of natural replenishment. • Axiom 4: To be sustainable, the use of nonrenewable resources must proceed at a rate that is declining, and the rate of decline must be greater than or equal to the rate of depletion. • Axiom 5: Sustainability requires that substances introduced into the environment from human activities be minimized and rendered harmless to biosphere functions.


GREEN CHEMISTRY DEFINITION Green Chemistry is the utilisation of a set of principles that reduces or eliminates the use or generation of hazardous substances in the design, manufacture and application of chemical products. GREEN CHEMISTRY IS ABOUT • • • Waste Minimization at Source Use of Catalysts in place of Reagents Using Non-Toxic Reagents Use of Renewable Resources Improved Atom Efficiency Use of Solvent Free or Recyclable Environmentally Benign Solvent systems

Green Chemistry Is About. . . Waste Materials Hazard Risk Energy Cost

Why do we need Green Chemistry ? • Chemistry is undeniably a very prominent part of our daily lives. • Chemical developments also bring new environmental problems and harmful unexpected side effects, which result in the need for ‘greener’ chemical products. • A famous example is the pesticide DDT.

• Green chemistry looks at pollution prevention on the molecular scale and is an extremely important area of Chemistry due to the importance of Chemistry in our world today and the implications it can show on our environment. • The Green Chemistry program supports the invention of more environmentally friendly chemical processes which reduce or even eliminate the generation of hazardous substances. • This program works very closely with the twelve principles of Green Chemistry.

The 12 Principles of Green Chemistry (1 -6)

The 12 Principles of Green Chemistry (7 -12) 7 Use of Renewable Feedstocks A raw material or feedstock should be renewable rather than depleting whenever technically and economically practicable . 8 Reduce Derivatives Unnecessary derivatization (use of blocking groups, protection/de-protection, and temporary modification of physical/chemical processes) should be minimised or avoided if possible, because such steps require additional reagents and can generate waste. 9 Catalysis Catalytic reagents (as selective as possible) are superior to stoichiometric reagents. 10 Design for Degradation Chemical products should be designed so that at the end of their function they break down into innocuous degradation products and do not persist in the environment. 11 Real-time Analysis for Pollution Prevention Analytical methodologies need to be further developed to allow for real-time, in-process monitoring and control prior to the formation of hazardous substances. 12 Inherently Safer Chemistry for Accident Prevention Substances and the form of a substance used in a chemical process should be chosen to minimise the potential for chemical accidents, including releases, explosions, and fires.

Principle 1 (Prevention): It is better to prevent waste than to treat or cleanup waste after it is formed Chemical Process
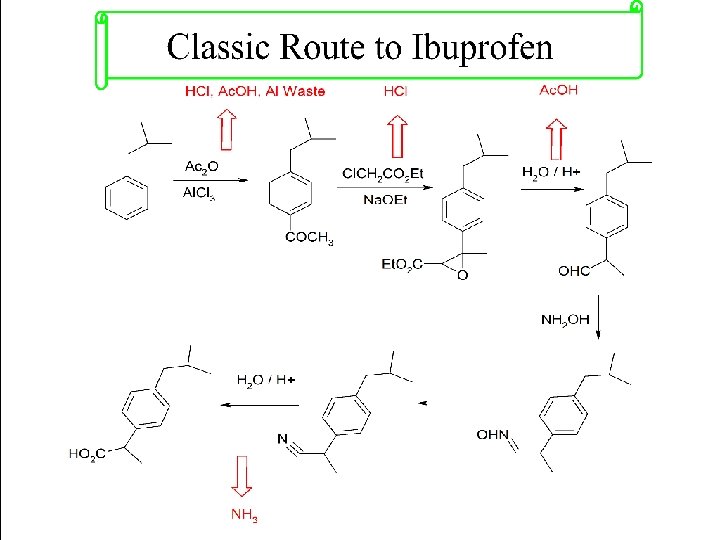
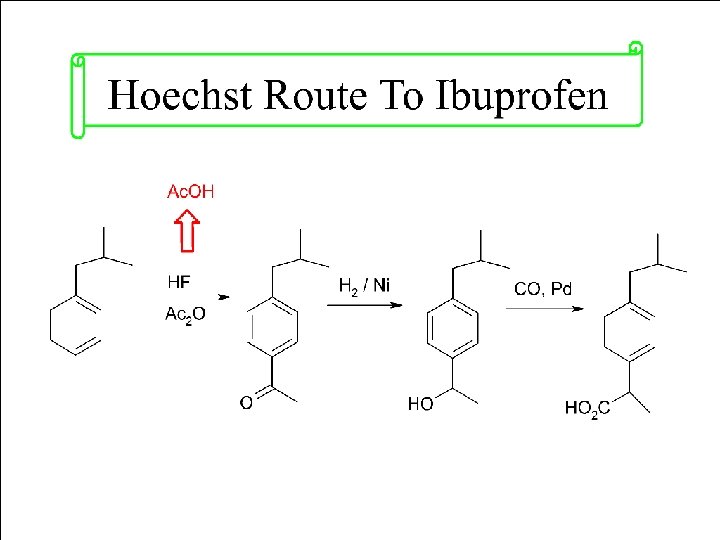

Principle 2. Atom Economy Synthetic methods should be designed to maximize the incorporation of all materials used in the process into the final product.
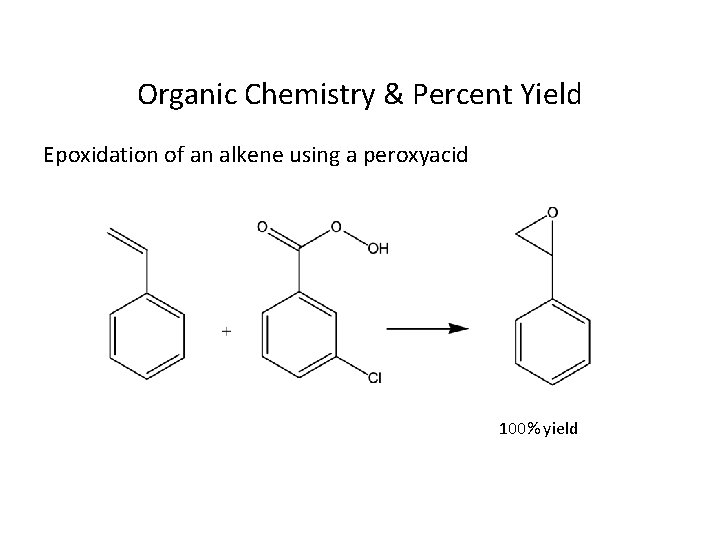
Organic Chemistry & Percent Yield Epoxidation of an alkene using a peroxyacid 100% yield

Percent yield: •
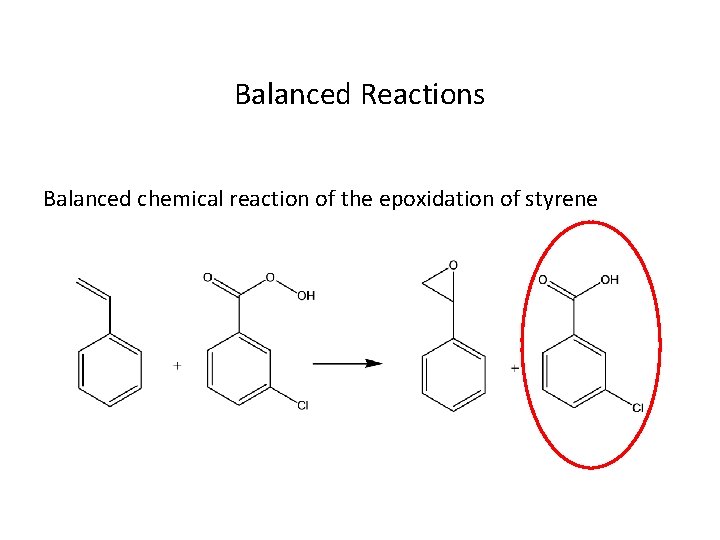
Balanced Reactions Balanced chemical reaction of the epoxidation of styrene

Green Chemistry Metrics • Percent (Chemical) Yield • Atom Economy – How much of the reactants remain in the final product – Does not account for solvents, reagents, reaction yield, and reactant molar excess

Green Chemistry Metrics (cont) • E-Factor – Smaller is better
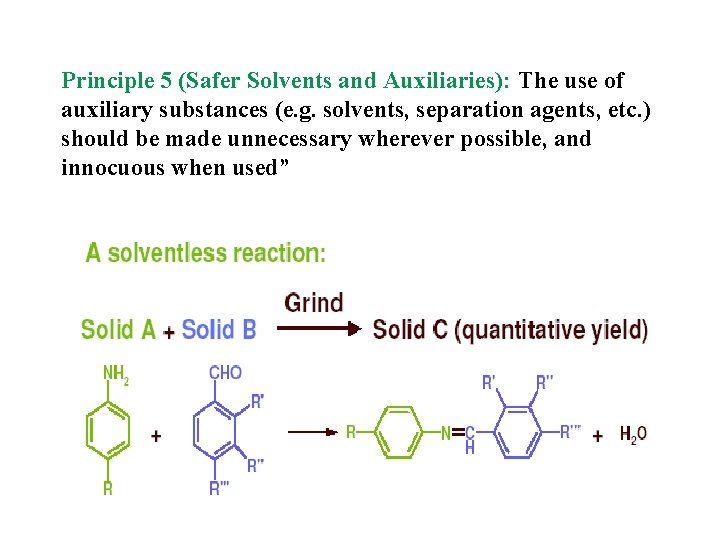
Principle 5 (Safer Solvents and Auxiliaries): The use of auxiliary substances (e. g. solvents, separation agents, etc. ) should be made unnecessary wherever possible, and innocuous when used”
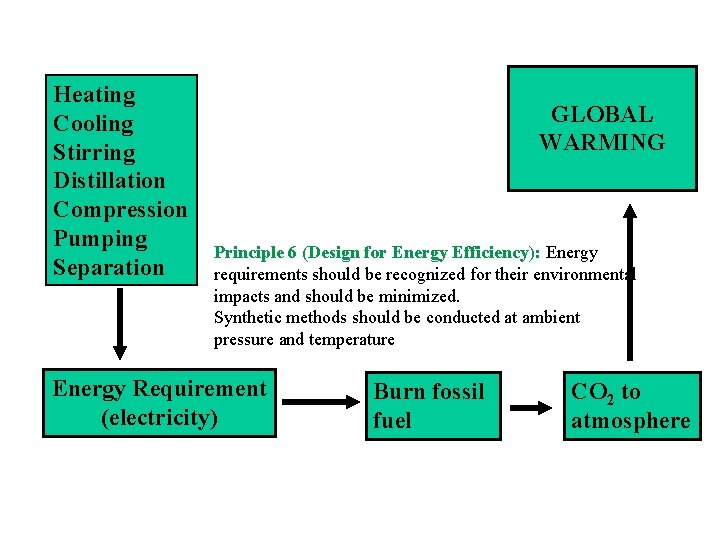
Heating Cooling Stirring Distillation Compression Pumping Separation GLOBAL WARMING Principle 6 (Design for Energy Efficiency): Energy requirements should be recognized for their environmental impacts and should be minimized. Synthetic methods should be conducted at ambient pressure and temperature Energy Requirement (electricity) Burn fossil fuel CO 2 to atmosphere

Principle 7 (Use of renewable feedstock): A raw material of feedstock should be renewable rather than depleting wherever technically and economically practical” Non-renewable Renewable
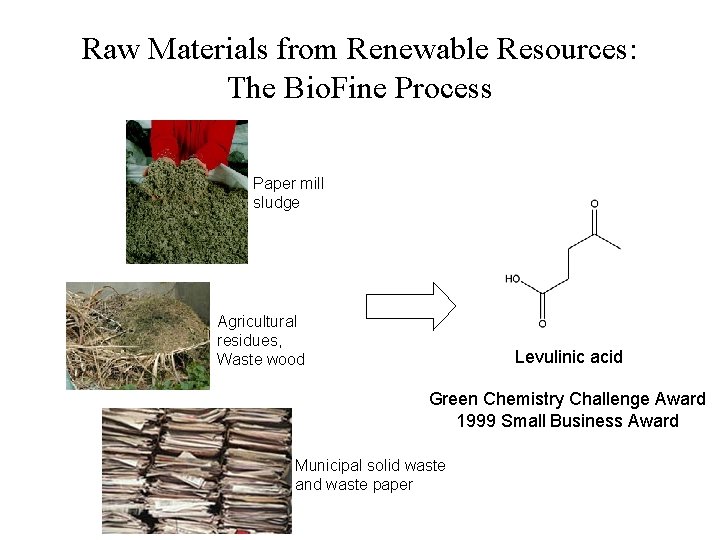
Raw Materials from Renewable Resources: The Bio. Fine Process Paper mill sludge Agricultural residues, Waste wood Levulinic acid Green Chemistry Challenge Award 1999 Small Business Award Municipal solid waste and waste paper
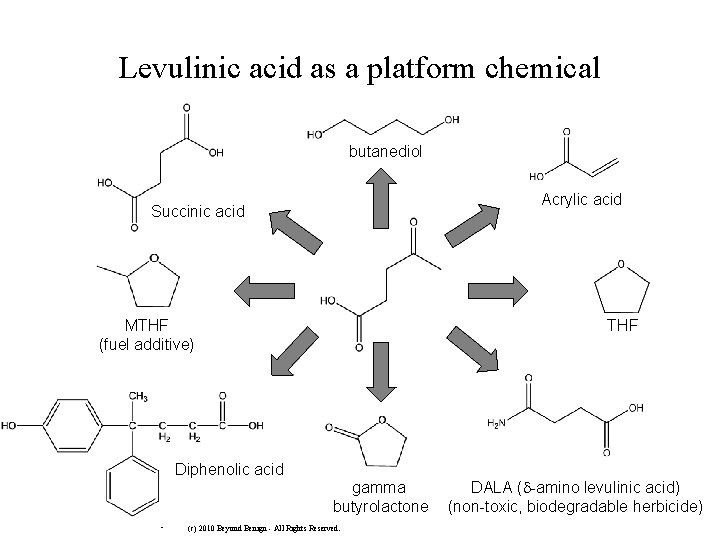
Levulinic acid as a platform chemical butanediol Acrylic acid Succinic acid MTHF (fuel additive) THF Diphenolic acid gamma butyrolactone • (c) 2010 Beyond Benign - All Rights Reserved. DALA ( -amino levulinic acid) (non-toxic, biodegradable herbicide)

Principle 9 (Catalysis) Catalytic reagents (as selective as possible) are superior to stoichiometric reagents.

Heterogeneous vs Homogenous • • • Distinct solid phase Readily separated Readily regenerated & recycled Rates not as fast Diffusion limited Sensitive to poisons Lower selectivity Long service life High energy process Poor mechanistic understanding • Same phase as rxn medium • Difficult to separate • Expensive and/or difficult to separate • Very high rates • Not diffusion controlled • Robust to poisons • High selectivity • Short service life • Mild conditions • Mechanisms well understood
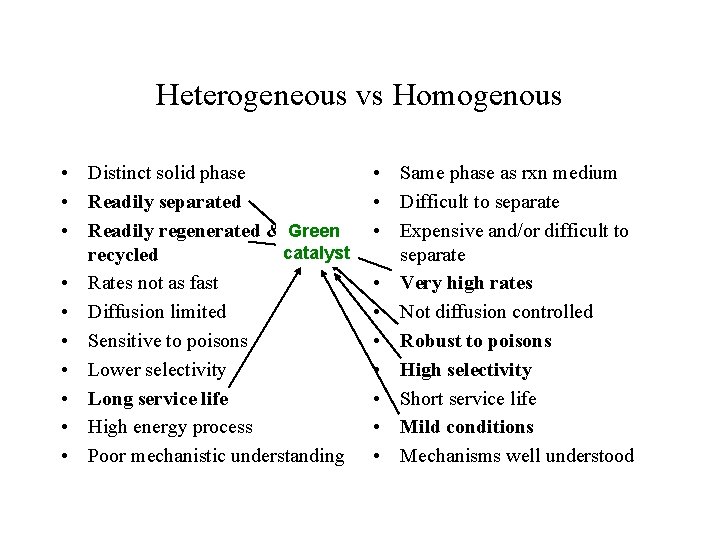
Heterogeneous vs Homogenous • Distinct solid phase • Readily separated • Readily regenerated & Green catalyst recycled • Rates not as fast • Diffusion limited • Sensitive to poisons • Lower selectivity • Long service life • High energy process • Poor mechanistic understanding • Same phase as rxn medium • Difficult to separate • Expensive and/or difficult to separate • Very high rates • Not diffusion controlled • Robust to poisons • High selectivity • Short service life • Mild conditions • Mechanisms well understood

Biocatalysis • Enzymes or whole-cell microorganisms • Benefits – Fast rxns due to correct orientations – Orientation of site gives high stereospecificity – Substrate specificity – Water soluble – Naturally occurring – Moderate conditions – Possibility for tandem rxns (onepot)

Principle 10 (Design for Degradation) Chemical products should be designed so that at the end of their function they do not persist in the environment and instead break down into innocuous degradation products.

Degradation of Polymers: Polylactic Acid u u u Manufactured from renewable resources n Corn or wheat; agricultural waste in future Uses 20 -50% fewer fossil fuels than conventional plastics PLA products can be recycled or composted
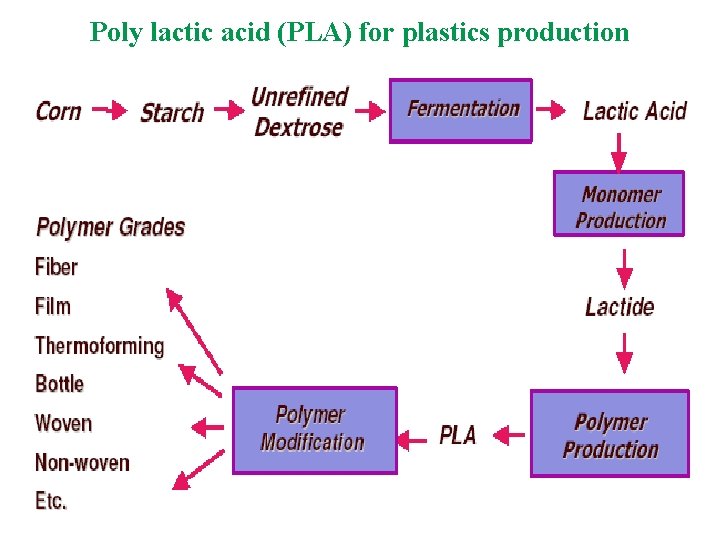
Poly lactic acid (PLA) for plastics production
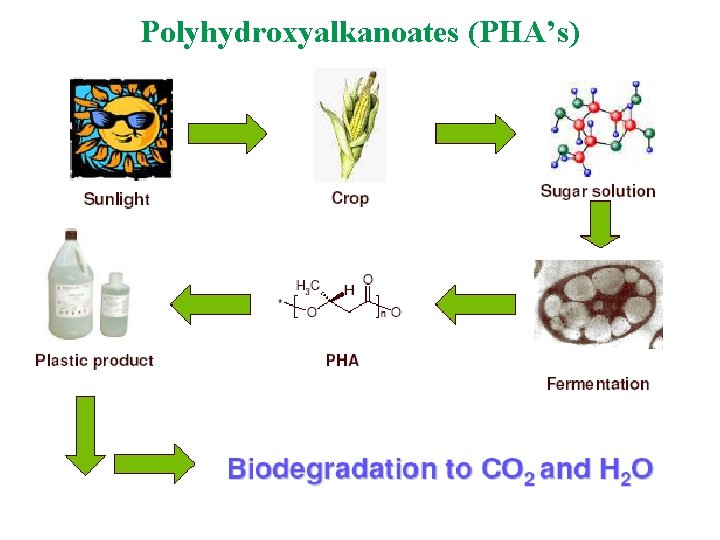
Polyhydroxyalkanoates (PHA’s)
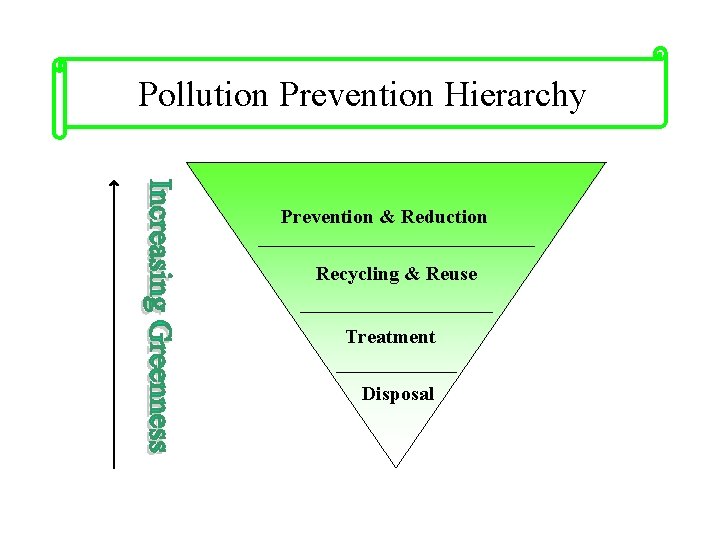
Pollution Prevention Hierarchy Prevention & Reduction Recycling & Reuse Treatment Disposal

Conclusion Not a solution to all environmental problems But Green chemistry the most fundamental approach to preventing pollution.

Thank you
- Slides: 37