Chapter 16 Immunity to Microbes Immunity to Microbes

Chapter 16 Immunity to Microbes

Immunity to Microbes v The key events during infection include entry of the microbe, invasion and colonization of host tissues, evasion of host immunity, and tissue injury or functional impairment. v Microbes produce disease by killing the host cells they infect or by liberating toxins that can cause tissue damage and functional derangements in neighboring or distant cells and tissues that are not infected.

Important General Features of Immunity to Microbes Defense against microbes is mediated by the effector mechanisms of innate and adaptive immunity The immune system responds in specialized and distinct ways to different types of microbes to most effectively combat these infectious agents. Survival and pathogenicity of microbes in a host are critically influenced by the ability of the microbes to evade or resist the effector mechanisms of immunity. The balance between host immune responses and microbial strategies for resisting immunity often determines the outcome of infections.

v Many microbes establish latent, or persistent, infections in which the immune response controls but does not eliminate the microbe and the microbe survives without propagating the infection. v Tissue injury and disease may be caused by the host response v Analysis of immune responses is a valuable clinical assay for infections. The most useful test is measurement of serum antibodies specific for particular microbes. v The presence of Ig. M is indicative of recent infection, whereas the presence of only Ig. G suggests past infection. Other tests include assays for T cell responses

IMMUNITY TO EXTRACELLULAR BACTERIA
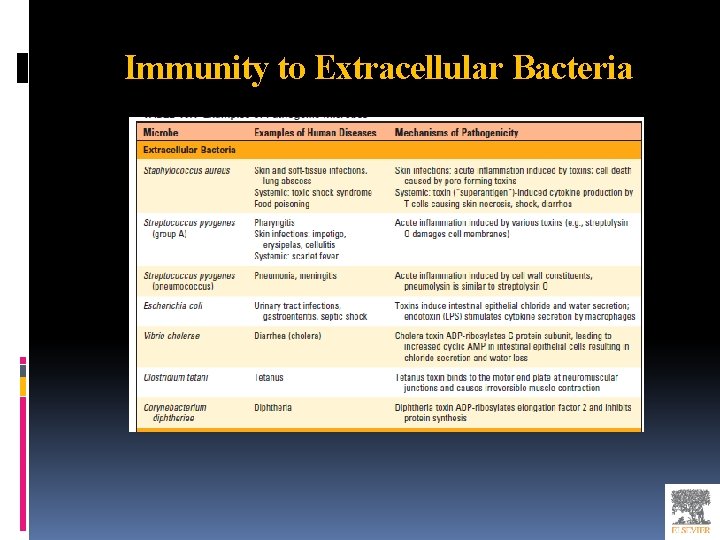
Immunity to Extracellular Bacteria

Innate Immunity to Extracellular Bacteria Extracellular bacteria are capable of replicating outside host cells, for example, in the blood, in connective tissues, and in tissue spaces such as the lumens of the airways and gastrointestinal tract. These bacteria induce inflammation, which results in tissue destruction at the site of infection. Second, bacteria produce toxins, which have diverse pathologic effects. The toxins are traditionally classified as endotoxins (LPS), which are components of bacterial cell walls, and exotoxins, which are secreted by the bacteria. Many toxins are cytotoxic, and others disrupts several critical biochemical signaling pathways in infected cells, stimulate the production of cytokines that cause disease.

Innate Immunity to Extracellular Bacteria v Principal mechanisms of innate immunity to extracellular bacteria are complement activation, phagocytosis, and the inflammatory response. v Complement activation: Alternative pathway and lectin pathway Opsonization and enhanced phagocytosis of the bacteria, recruiting and activating leukocytes v Activation of phagocytes and inflammation: Phagocytes (neutrophils and macrophages) use surface receptors, to recognize extracellular bacteria, and use Fc receptors and complement receptors to recognize bacteria opsonized. v Toll-like receptors (TLRs) of phagocytes participate in the activation of the phagocytes as a result of encounter with microbes. v Secretion of cytokines that induce leukocyte infiltration into sites of infection (inflammation)
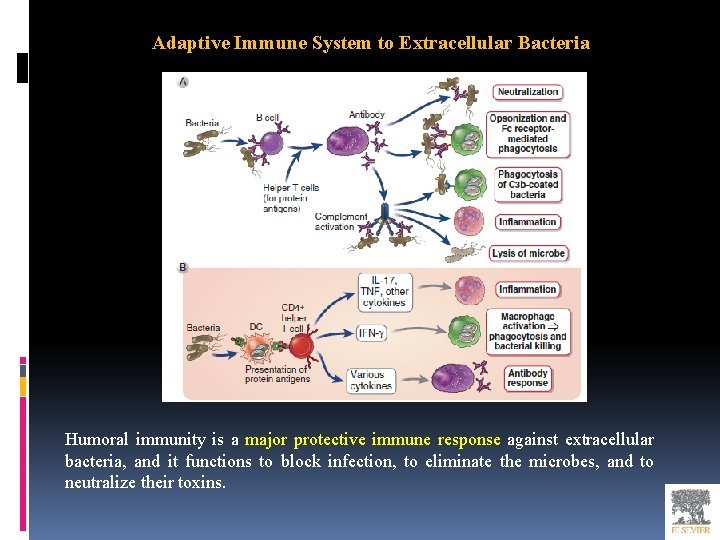
Adaptive Immune System to Extracellular Bacteria Humoral immunity is a major protective immune response against extracellular bacteria, and it functions to block infection, to eliminate the microbes, and to neutralize their toxins.

Injurious Effects of Immune Responses The principal injurious consequences of host responses to extracellular bacteria are inflammation and sepsis. Cytokines secreted by leukocytes in response to bacterial products also stimulate the production of acute-phase proteins and cause the systemic manifestations of the infection. Sepsis is a pathologic consequence of severe infection by some bacteria (as well as some fungi), in which viable microbes or microbial products are present in the blood. Septic shock is the most severe and frequently fatal form of sepsis. Tumor necrosis factor (TNF), IL-6, and IL-1 are the principal cytokine mediators of sepsis, but IFN-γ and IL-12 may also contribute (cytokine storm).
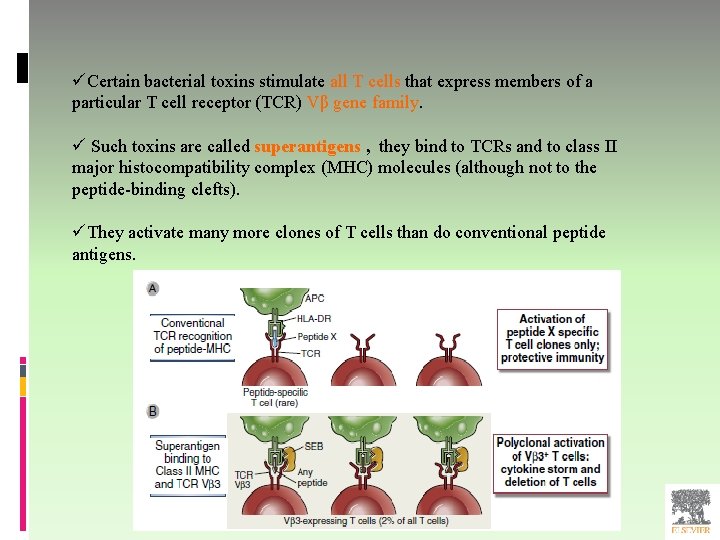
üCertain bacterial toxins stimulate all T cells that express members of a particular T cell receptor (TCR) Vβ gene family. ü Such toxins are called superantigens , they bind to TCRs and to class II major histocompatibility complex (MHC) molecules (although not to the peptide-binding clefts). üThey activate many more clones of T cells than do conventional peptide antigens.

üA late complication of the humoral immune response to bacterial infection may be the generation of disease-producing antibodies. üRheumatic fever is a sequel to pharyngeal infection with some serologic types of group A β-hemolytic streptococci. Infection leads to the production of antibodies against a bacterial cell wall protein. üSome of these antibodies cross-react with myocardial proteins and are deposited in the heart, where they cause inflammation (carditis).
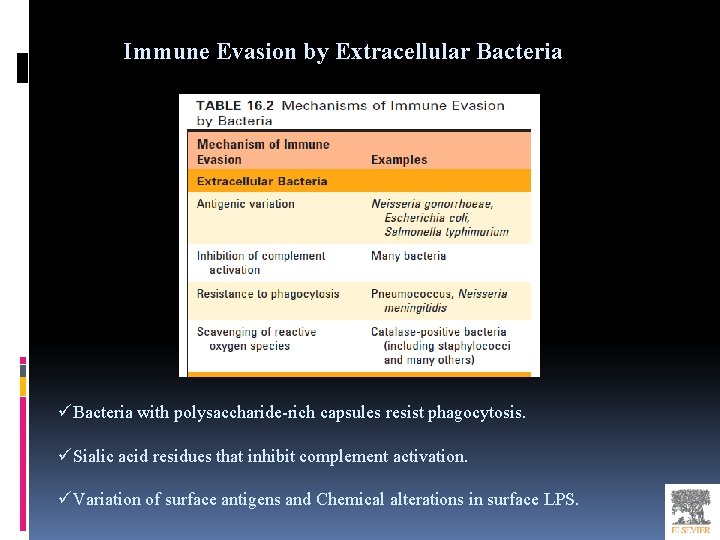
Immune Evasion by Extracellular Bacteria üBacteria with polysaccharide-rich capsules resist phagocytosis. üSialic acid residues that inhibit complement activation. üVariation of surface antigens and Chemical alterations in surface LPS.
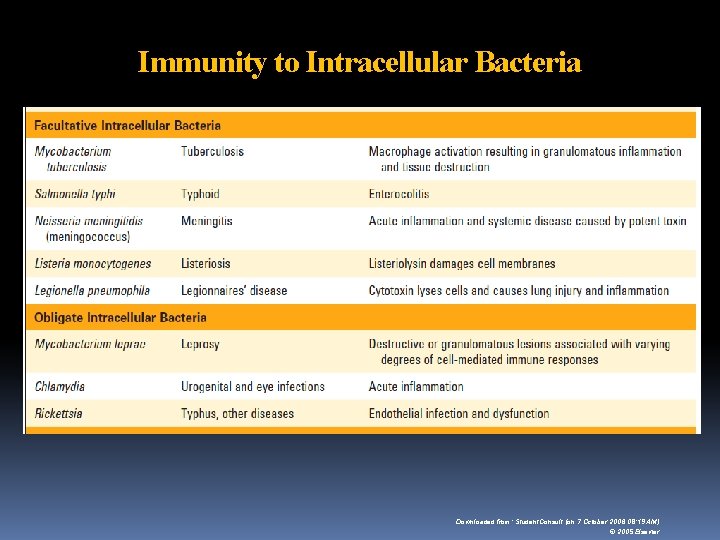
Immunity to Intracellular Bacteria Downloaded from: Student. Consult (on 7 October 2006 08: 19 AM) © 2005 Elsevier
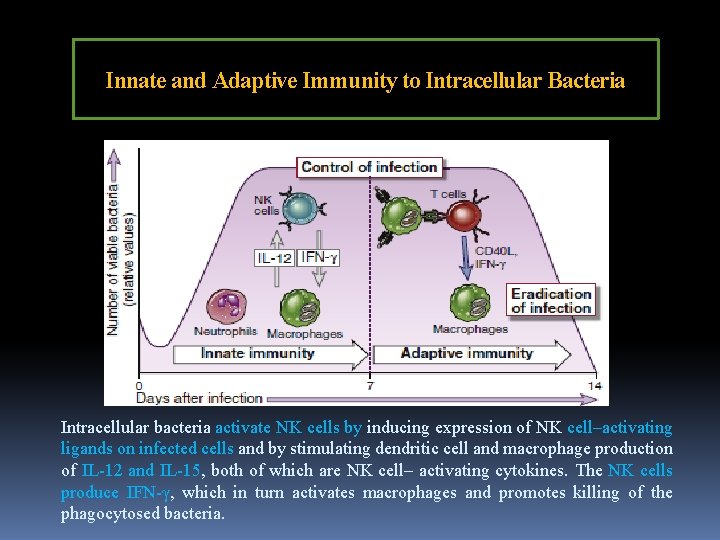
Innate and Adaptive Immunity to Intracellular Bacteria Intracellular bacteria activate NK cells by inducing expression of NK cell–activating ligands on infected cells and by stimulating dendritic cell and macrophage production of IL-12 and IL-15, both of which are NK cell– activating cytokines. The NK cells produce IFN-γ, which in turn activates macrophages and promotes killing of the phagocytosed bacteria.
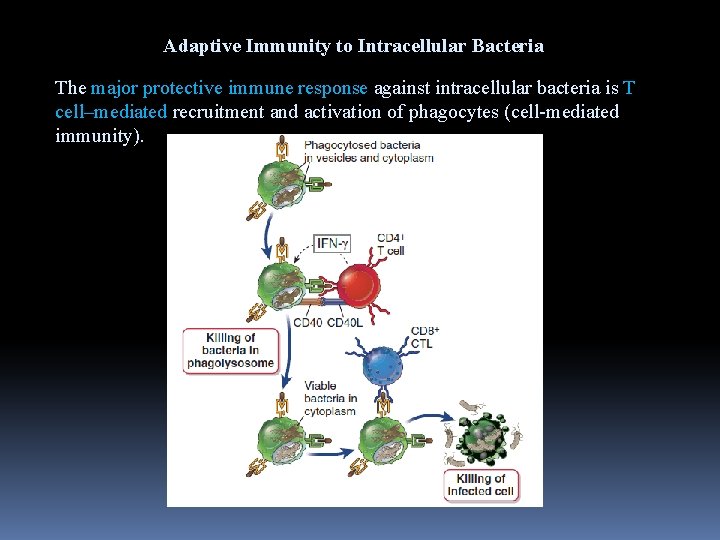
Adaptive Immunity to Intracellular Bacteria The major protective immune response against intracellular bacteria is T cell–mediated recruitment and activation of phagocytes (cell-mediated immunity).

v Intracellular bacteria have evolved to resist killing within phagocytes, persist for long periods and cause chronic T cell and macrophage activation, which may result in the formation of granulomas surrounding the microbes. v Granulomatous inflammation may serve to localize and prevent spread of the microbes, but it is also associated with severe functional impairment caused by tissue necrosis and fibrosis.
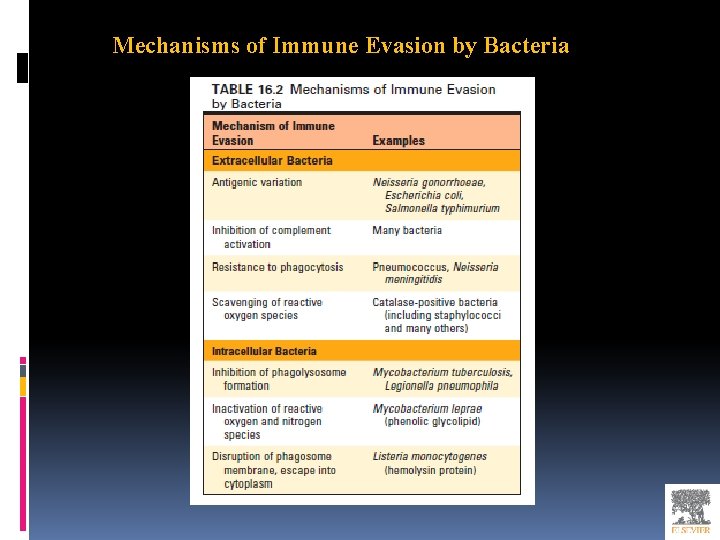
Mechanisms of Immune Evasion by Bacteria

Immunity to Fungi

Fungal infections, also called mycoses, are important causes of morbidity and mortality in humans. Compromised immunity is the most important predisposing factor (opportunistic infection). Different fungi infect humans and may live in extracellular tissues and within phagocytes. Therefore, the immune responses to these microbes are often combinations of the responses to extracellular and intracellular microbes.
Innate and Adaptive Immunity to Fungi The principal mediators of Innate Immunity are neutrophils and macrophages. Patients with neutropenia are extremely susceptible to opportunistic fungal infections. Phagocytes and dendritic cells sense fungal organisms by TLRs and lectin-like receptors called dectins Neutrophils presumably liberate fungicidal substances, such as reactive oxygen species and lysosomal enzymes, and phagocytose fungi for intracellular killing

Cell-mediated immunity is the major mechanism of adaptive immunity against intracellular fungal infections. (macrophages/TH 1/TCD 8+) Many extracellular fungi elicit strong TH 17 responses Individuals with defective TH 17 responses are susceptible to chronic mucocutaneous Candida infections. Fungi also elicit specific antibody responses that may be of protective value.

Immunity to Viruses are obligatory intracellular microorganisms that use components of the nucleic acid and protein synthetic machinery of the host to replicate. Viral replication interferes with normal cellular protein synthesis and function and leads to injury and ultimately death of the infected cell Cytopathic effect of viruses Viruses can stimulate inflammatory responses that cause damage to tissues. Viruses may also cause latent infections, discussed later. Innate and adaptive immune responses to viruses are aimed at blocking infection and eliminating infected cells

ü Innate immunity to viruses: The principal mechanisms of innate immunity against viruses are inhibition of infection by type I interferons and NK cell–mediated killing of infected cells. ü Production of type I interferons (IFNs) by infected cells, and by dendritic cells, especially of the plasmacytoid type following recognition of viral RNA and DNA by endosomal TLRs. ü Type I IFNs function to inhibit viral replication in both infected and uninfected cells. ü Class I MHC expression is often shut off in virus-infected cells as an escape mechanism from CTLs. This enables NK cells to kill the infected cells because the absence of class I releases NK cells from a normal state of inhibition.


Mechanisms of Immune Evasion by Viruses
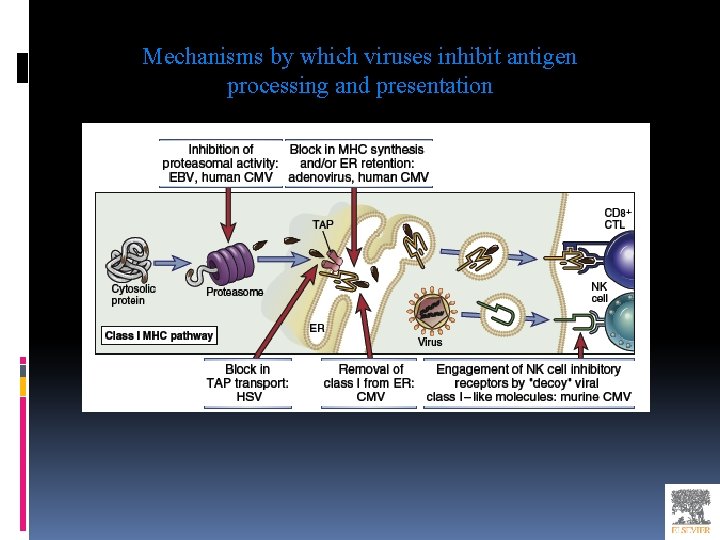
Mechanisms by which viruses inhibit antigen processing and presentation

Immunity to parasites

Parasitic infection such as protozoa, helminths, and ectoparasites (ticks and mites) Innate immunity to parasites: ü to protozoa and helminths is phagocytosis but may resist ü activate the alternative pathway of complement Adaptive immunity to parasites: ü to protozoa is macrophage activation by Th 1 cell-derived cytokines ü to helminths is Th 2 cells and Ig. E and activation of eosinophils then secrete MBP that is severe toxin for helminths Tissue injury after Adaptive immune responses (as fibrosis)
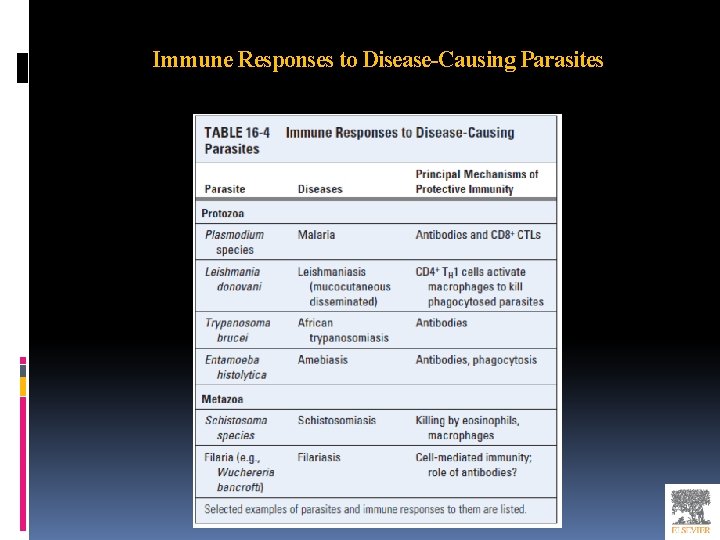
Immune Responses to Disease-Causing Parasites
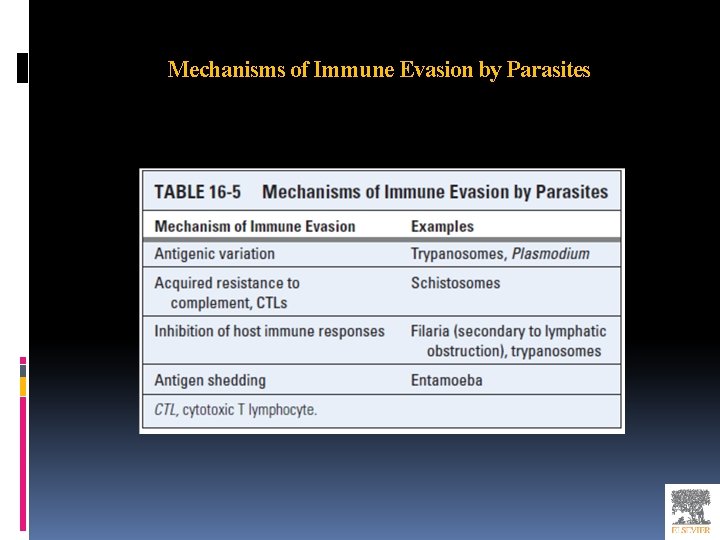
Mechanisms of Immune Evasion by Parasites

STRATEGIES FOR VACCINE DEVELOPMENT

The birth of immunology as a science dates from Edward Jenner's successful vaccination against smallpox in 1796 The success of active immunization in eradicating infectious disease is dependent on numerous factors: Vaccines are effective if the infectious agent does not establish latency, if it does not undergo much or any antigenic variation, and if it does not interfere with the host immune response limited to human hosts, and do not have animal reservoirs
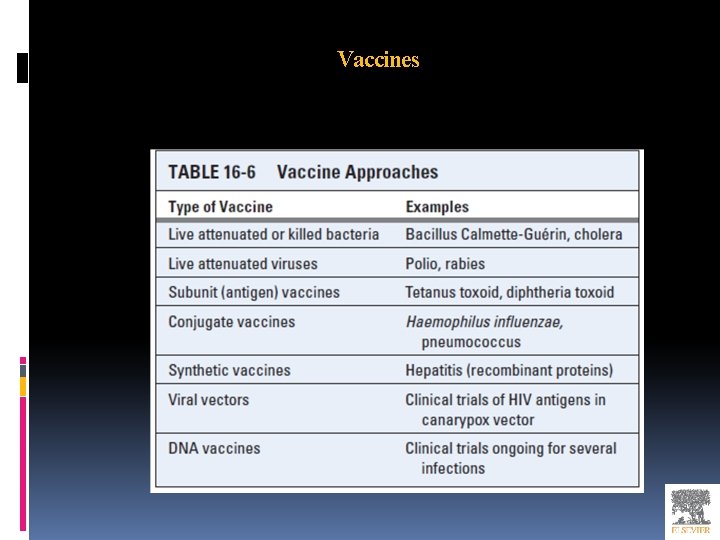
Vaccines

Adjuvants and Immunomodulators Most adjuvants elicit innate immune responses, with increased expression of costimulators and production of cytokines such as IL-12 that stimulate T cell growth and differentiation Heat-killed bacteria are powerful adjuvants that are commonly used in experimental animals with severe local inflammation Several are in clinical practice, including aluminum hydroxide gel (which appears to promote B cell responses) and lipid formulations that are ingested by phagocytes IL-12 incorporated in vaccines promotes strong CMI plasmid DNA has intrinsic adjuvant-like activities, and it is possible to incorporate costimulators (e. g. , B 7 molecules) or cytokines

Passive Immunization Passive immunity is short-lived because the host does not respond to the immunization and protection lasts only as long as the injected antibody persists In the clinical situation, passive immunization is most commonly used for rapid treatment of potentially fatal diseases caused by toxins, such as tetanus, and for protection from rabies and hepatitis Passive immunization does not induce memory, so an immunized individual is not protected against subsequent exposure to the toxin or microbe

- Slides: 37