Chapter 16 Acids Bases Acids vs Bases Section

Chapter 16 Acids & Bases

Acids vs. Bases Section 16. 1 ¢ Arrhenius model l ¢ Brønsted-Lowry model l ¢ Oldest model; only applies to compounds that contain H+ or OHions Refers to a compound’s ability to donate or accept an H+ ion Ex: HCl NH 3 H 2 O
Brønsted-Lowry Acids and Bases Section 16. 2 ¢ Water as Brønsted. Lowry acid/base: l No such thing as H+ ion in solution (too unstable) • Only H 3 O+ ¢ Proton transfer reactions:
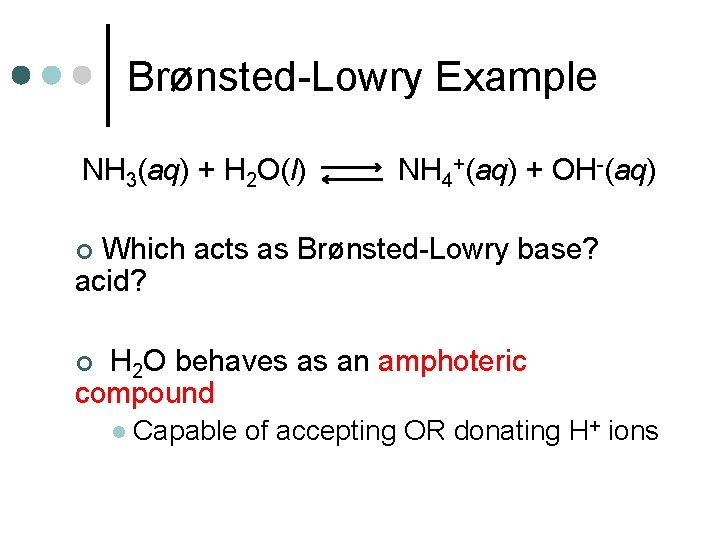
Brønsted-Lowry Example NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq) Which acts as Brønsted-Lowry base? acid? ¢ H 2 O behaves as an amphoteric compound ¢ l Capable of accepting OR donating H+ ions
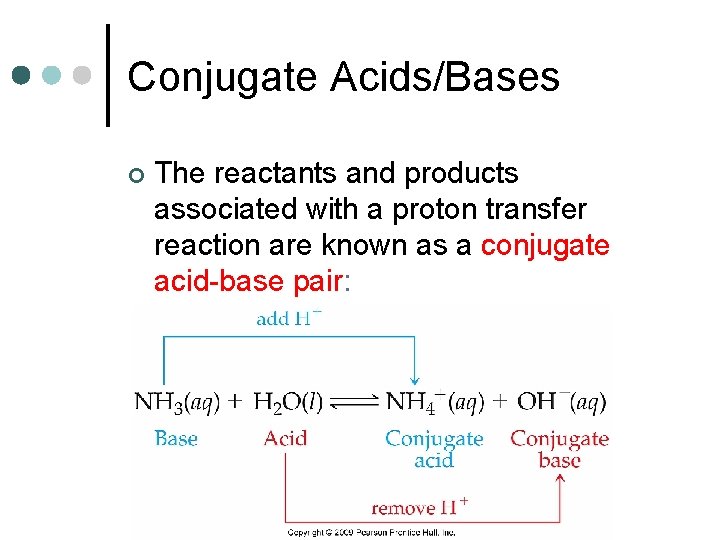
Conjugate Acids/Bases ¢ The reactants and products associated with a proton transfer reaction are known as a conjugate acid-base pair:

Relative Strength of Acids and Bases ¢ The strength of an acid depends primarily on the willingness to donate or accept electrons l l i. e. strength of conjugate acid/base Equilibrium for strong acids lies heavily on the side of the deprotonated form and vice versa

The Autoionization of Water Section 16. 3 ¢ Water has a very interesting property due to its amphoterism l Capable of autoionization:
![Calculating the [H 3 O+] An acid is added to water so that the Calculating the [H 3 O+] An acid is added to water so that the](http://slidetodoc.com/presentation_image/68575d1e9839f0f62dff96fa20131b86/image-8.jpg)
Calculating the [H 3 O+] An acid is added to water so that the hydrogen ion concentration is 0. 25 M. Calculate the hydroxide ion concentration. ¢ See Sample Exercise 16. 4 (Pg. 674)

The p. H Scale Section 16. 4 ¢ Concentrations of either H 3 O+ or OH- are typically very small and therefore cumbersome l The p. H scale is a logarithmic scale and is much more convenient: p. H = -log[H 3 O+] p. OH = -log[OH-] p. H + p. OH = 14

p. H of Common Substances

Strong Acids and Bases Section 16. 5 ¢ For a compound to be classified as a strong acid or base it must completely dissociate into ions when placed into aqueous solution l l ¢ ¢ Very weak conjugate bases No equilibrium Ex: HCl(g) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq) There are 7 strong acids which you will have to remember: l HCl, HBr, HI, HNO 3, HCl. O 4, H 2 SO 4

Weak Acids Section 16. 6 Weak acids have a relatively strong conjugate base (compared to strong acids) and therefore some competition exists for the proton HF(g) + H 2 O(l) H 3 O+(aq) + F-(aq) ¢ ¢ As a result, the equilibrium between conjugate acid/base is treated as if it were any other equilibrium

Calculating Ka for a Weak Acid Picric acid, a weak acid, is dissolved in water to prepare a 0. 100 M solution. The p. H of the solution was found to be 1. 09. Calculate Ka for picric acid at this temperature. See Sample Exercise 16. 10 (Pg. 682)

Using Ka to Calculate p. H Calculate the p. H in a 0. 100 M solution of the weak acid naphthol, for which Ka is 1. 7 10 -10 ¢ See Sample Exercise 16. 12 (Pg. 685)

Using Ka to Calculate p. H Calculate the p. H of 0. 017 M C 6 H 5 COOH. The Ka of C 6 H 5 COOH is 6. 3 x 10 -5. ¢ See Sample Exercise 16. 12 (Pg. 685)

p. H of Weak Acids ¢ Calculate the p. H of a 0. 10 M HF solution

Percent Ionization ¢ Weak acids, by definition, do not ionize 100% when placed in aqueous solution l It is therefore possible to calculate the extent of ionization (percent ionization)

Calculating Percent Ionization Calculate the percent of HF molecules ionized in a 0. 010 M HF solution. ¢ See Sample Exercise 16. 13 (Pg. 687)
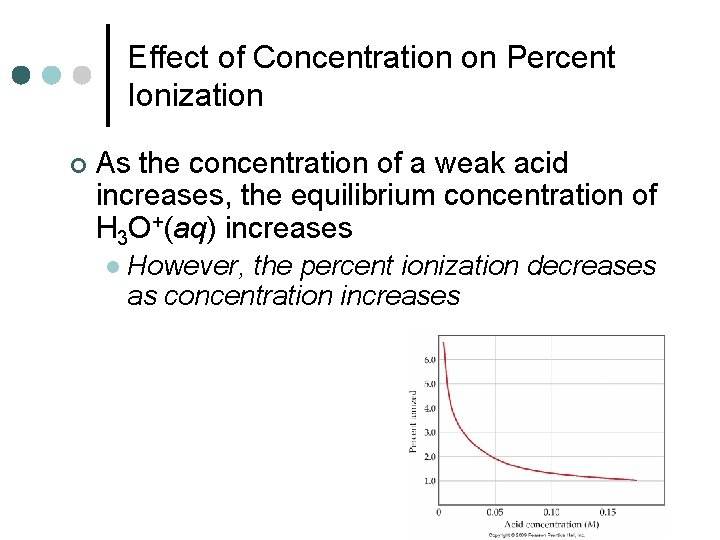
Effect of Concentration on Percent Ionization ¢ As the concentration of a weak acid increases, the equilibrium concentration of H 3 O+(aq) increases l However, the percent ionization decreases as concentration increases
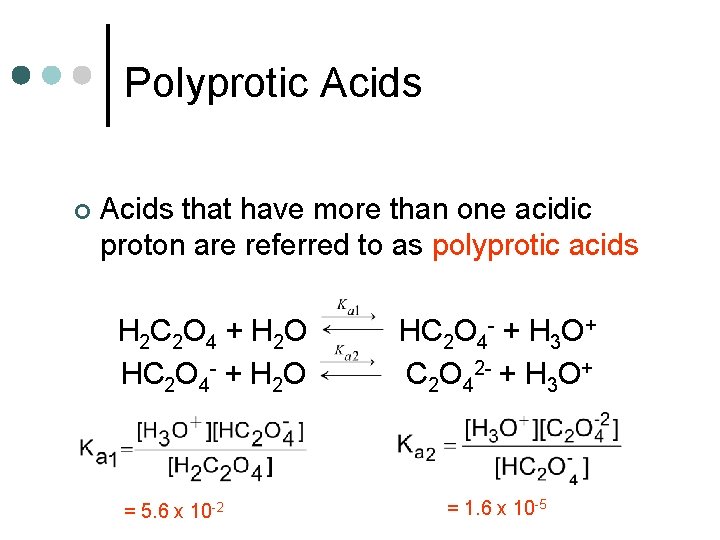
Polyprotic Acids ¢ Acids that have more than one acidic proton are referred to as polyprotic acids H 2 C 2 O 4 + H 2 O HC 2 O 4 - + H 2 O = 5. 6 x 10 -2 HC 2 O 4 - + H 3 O+ C 2 O 42 - + H 3 O+ = 1. 6 x 10 -5

Calculating p. H for Polyprotic Acid Solutions ¢ ¢ Because the acidic protons of a polyprotic acid are removed in discreet steps, we must calculate the [H 3 O+] for each step However, because Ka 1 is typically very large compared to Ka 2 or Ka 3, we may initially assume that the [H 3 O+] is a result of the first ionization

Calculating p. H and Concentration of All Species for a Polyprotic Acid Calculate the p. H of a 0. 0037 M H 2 CO 3 solution. For super duper fun, calculate the [CO 32 -] in solution. ¢ See Sample Exercise 16. 14 (Pg. 689)

Weak Bases Section 16. 7 ¢ There are fewer weak bases compared to the number of weak acids l Equilbria are the same (only involve OHas product as opposed to H 3 O+)

Calculating p. H of Weak Base Solutions ¢ What is the p. H of a 1. 44 M (concentration of household ammonia) solution of NH 3? Kb = 1. 8 x 10 -5 ¢ See Sample Exercise 16. 15 (Pg. 691)

Relationship Between Ka and Kb Section 16. 8 ¢ Examine the following equilibria: NH 4+(aq) NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq)

Converting Between Ka and Kb ¢ The product of the dissociation constants for any conjugate acid/base pair is always equal to the dissociation constant for H 2 O: Ka x Kb = Kw = 1. 0 x 10 -14 l Likewise: p. Ka + p. Kb = p. Kw = 14. 00 l

Calculating p. H of a Weak Base (Two Approaches) ¢ Calculate the p. H of a solution that is 0. 0500 M in ammonium ion. Kb = 1. 8 x 10 -5

Acid-Base Properties of Salt Solutions Section 16. 9 ¢ Hydrolysis reactions l The conjugate base of weak acids are capable of producing hydroxide ions in solution • Raises p. H l Conjugate acid of weak bases are capable of producing hydronium ions in solution • Lowers p. H

Acid-Base Behavior and Chemical Structure Section 16. 10 ¢ The chemical structure of a compound is what ultimately determines acid/base behavior l ¢ Ex: Why does Na. OH act as a base whereas CH 3 OH acts as a weak acid? Why does KH act as a strong base? Factors that affect? l l l Polarization of H—X bond (Na. H vs. CH 4) Bond strength (HF vs. HCl) Stability of conjugate base (HNO 3 vs. HNO 2)

Oxyacids ¢ ¢ Special attention is paid to oxyacids because the majority of weak acids fall into this category The strength of these acids is governed by one overriding principle: charge stabilization l Either through electronegativity or resonance

Effect of Additional Oxygens ¢ Because oxygen is a very electronegative element, the addition of more oxygen leads to a stronger acid:
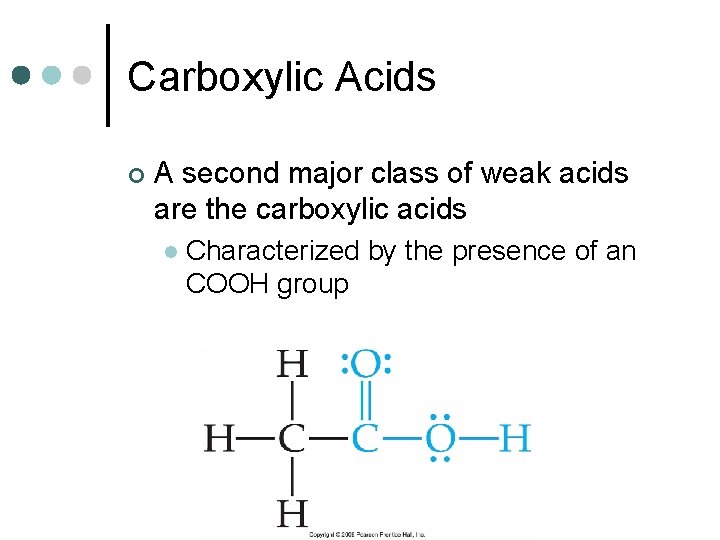
Carboxylic Acids ¢ A second major class of weak acids are the carboxylic acids l Characterized by the presence of an COOH group
- Slides: 32