Chapter 16 Acids Bases Acids vs Bases Section

Chapter 16 Acids & Bases

Acids vs. Bases Section 16. 1 ¢ Arrhenius model l ¢ Brønsted-Lowry model l ¢ Oldest model; only applies to compounds that contain H+ or OHions Refers to a compound’s ability to donate or accept an H+ ion Ex: HCl NH 3 H 2 O
Brønsted-Lowry Acids and Bases Section 16. 2 ¢ Water as Brønsted. Lowry acid/base: l No such thing as H+ ion in solution (too unstable) • Only H 3 O+ ¢ Proton transfer reactions:
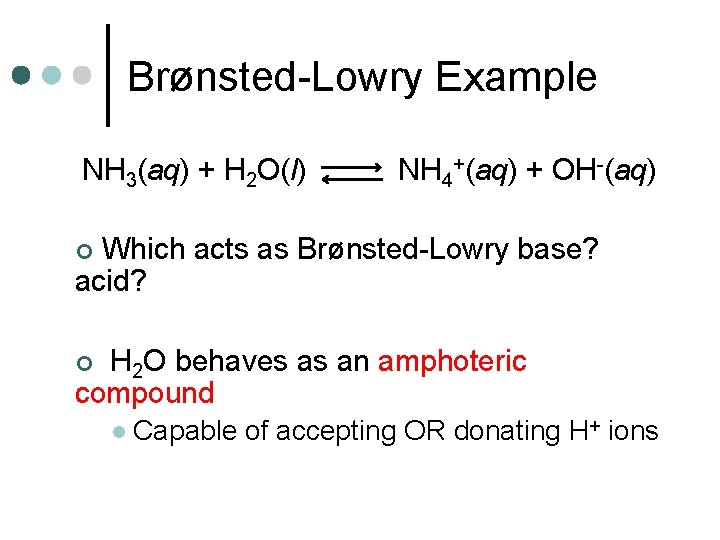
Brønsted-Lowry Example NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq) Which acts as Brønsted-Lowry base? acid? ¢ H 2 O behaves as an amphoteric compound ¢ l Capable of accepting OR donating H+ ions
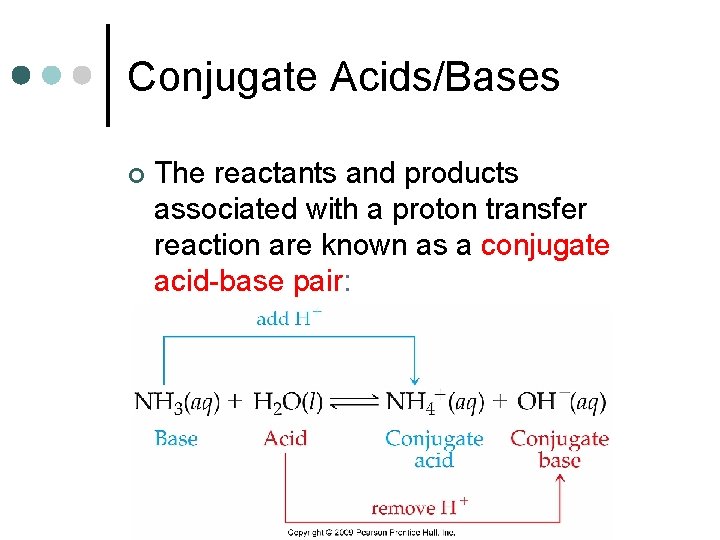
Conjugate Acids/Bases ¢ The reactants and products associated with a proton transfer reaction are known as a conjugate acid-base pair:

Relative Strength of Acids and Bases ¢ The strength of an acid depends primarily on the willingness to donate or accept electrons l l i. e. strength of conjugate acid/base Equilibrium for strong acids lies heavily on the side of the deprotonated form and vice versa

The Autoionization of Water Section 16. 3 ¢ Water has a very interesting property due to its amphoterism l Capable of autoionization:
![Calculating the [H 3 O+] An acid is added to water so that the Calculating the [H 3 O+] An acid is added to water so that the](http://slidetodoc.com/presentation_image_h/1406de43884d7291a1c6065a05639916/image-8.jpg)
Calculating the [H 3 O+] An acid is added to water so that the hydrogen ion concentration is 0. 25 M. Calculate the hydroxide ion concentration. ¢ See Sample Exercise 16. 4 (Pg. 674)

The p. H Scale Section 16. 4 ¢ Concentrations of either H 3 O+ or OH- are typically very small and therefore cumbersome l The p. H scale is a logarithmic scale and is much more convenient: p. H = -log[H 3 O+] p. OH = -log[OH-] p. H + p. OH = 14

p. H of Common Substances

Strong Acids and Bases Section 16. 5 ¢ For a compound to be classified as a strong acid or base it must completely dissociate into ions when placed into aqueous solution l l ¢ ¢ Very weak conjugate bases No equilibrium Ex: HCl(g) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq) There are 7 strong acids which you will have to remember: l HCl, HBr, HI, HNO 3, HCl. O 4, H 2 SO 4
- Slides: 11