Chapter 16 1 Electric Charge and Electric Force











- Slides: 16

Chapter 16 -1 Electric Charge and Electric Force

Quick Note: • Most of this is not in the textbook.

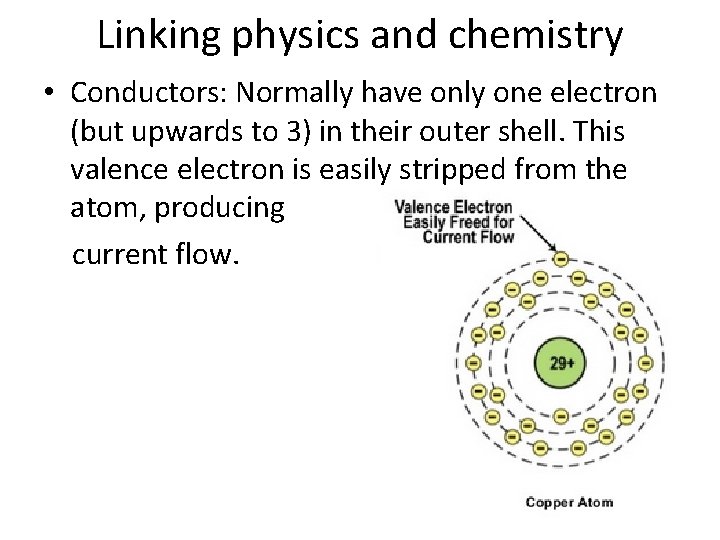
Linking physics and chemistry • Conductors: Normally have only one electron (but upwards to 3) in their outer shell. This valence electron is easily stripped from the atom, producing current flow.

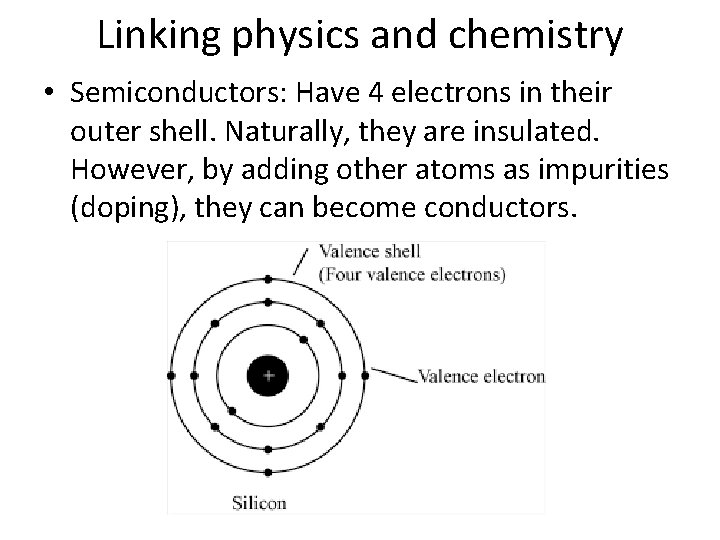
Linking physics and chemistry • Semiconductors: Have 4 electrons in their outer shell. Naturally, they are insulated. However, by adding other atoms as impurities (doping), they can become conductors.

Insulators • Insulators will generally have 5 -8 electrons (a full outer shell) and a high electronegativity (desire for electrons).

Semi-Conductors and Doping 2 types of doping are used: • N-Type: phosphorous or arsenic is added to silicon. Both have 5 valence electrons. When added to the lattice of silicon the 5 th electron cannot bond and is free to move around. • P-Type: boron or gallium is added. These have only 3 outer electrons, so when added to the silicon lattice, a hole exists.

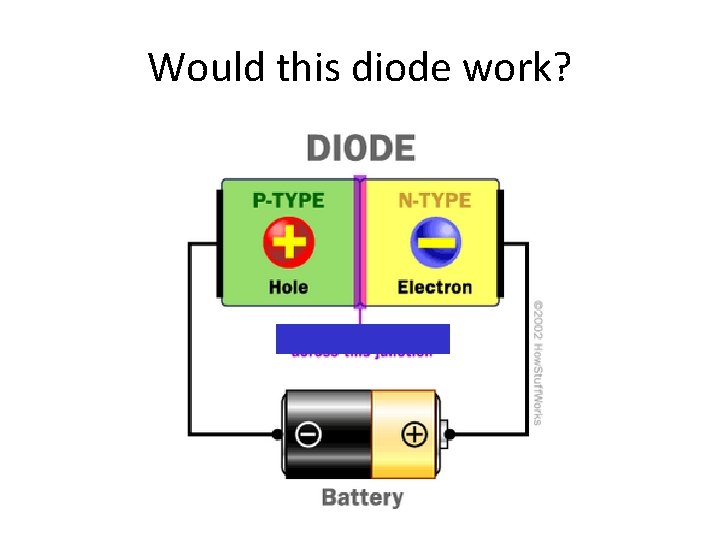
Using P- and N-Type Semi-Conductors • On their own, neither type of semi-conductor is that great. • However, when combined, the junction between the two is very interesting. • Combining the two creates a diode • A diode allows current to flow in one direction, but not the other.

Would this diode work?

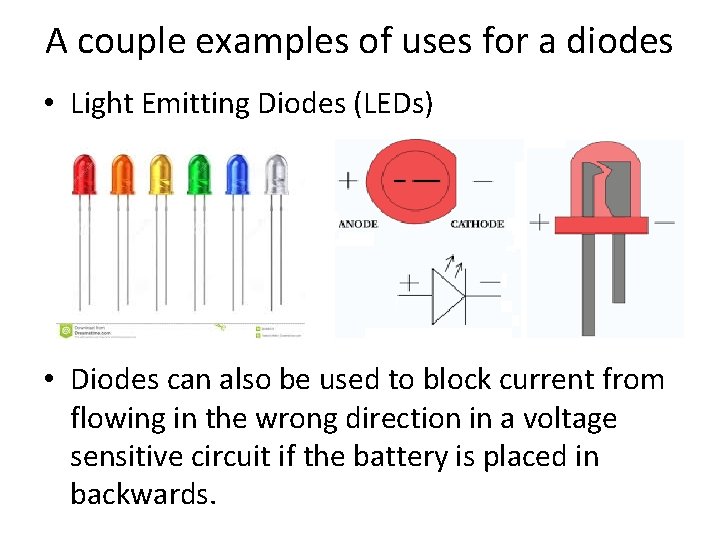
A couple examples of uses for a diodes • Light Emitting Diodes (LEDs) • Diodes can also be used to block current from flowing in the wrong direction in a voltage sensitive circuit if the battery is placed in backwards.

Superconductors • Superconductors conduct electricity with no resistance. • This means they could carry a current indefinitely without losing any energy. • First discovered in 1911 by a Dutch scientist named Heike Kammerling Onnes, who was the first person to liquefy helium and reach temperatures as low as 1. 7 Kelvin (-271. 45°C)

Superconductors • The most recent research is using hydrogen sulfide (H 2 S), which gives eggs the rotten smell. • The liquid is super-cooled and placed between two diamond plates. The mixture is subjected to 1. 5 megabars (10, 000 k. Pa) of pressure and will superconduct at -70°C. • This pressure is equivalent to half of the pressure found at Earth’s core.

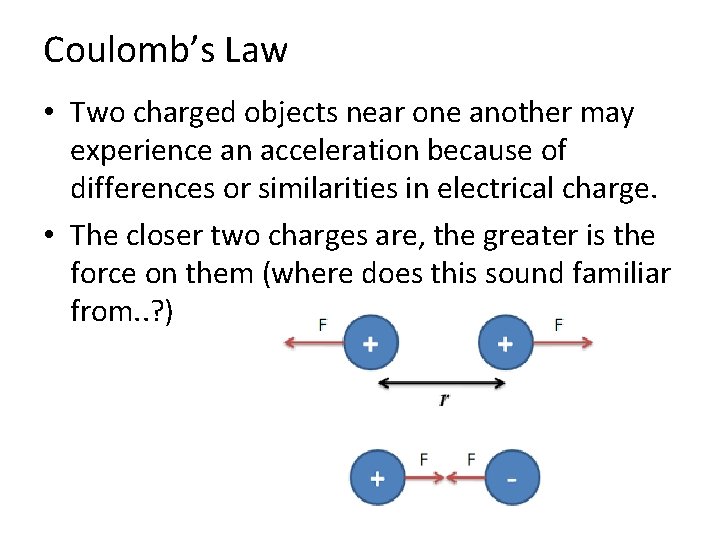
Coulomb’s Law • Two charged objects near one another may experience an acceleration because of differences or similarities in electrical charge. • The closer two charges are, the greater is the force on them (where does this sound familiar from. . ? )

Formula Symbol Description Unit Name Unit Felectric Electrical Force between two particles Newton N k. C Coulomb’s Constant Newton meters squared per Coulomb squared Nm 2/C 2 q 1 and q 2 Charge 1 and charge 2 Coulombs C r 2 meters squared m 2 radius squared

Example • The electron and proton of a hydrogen atom are separated, on average, by a distance of about 5. 3 x 10 -11 m. Find the magnitudes of the elctric force and the gravitational force that each particle exerts on each other. melec = 9. 109 x 10 -31 kg mprot = 1. 673 x 10 -27 kg qe = -1. 60 x 10 -19 C qp = +1. 60 x 10 -19 C

Solution

Practice Problems • Page 556 #1 -3