Chapter 15 Water and Aqueous Systems Part 1
















- Slides: 28

Chapter 15: Water and Aqueous Systems Part 1 15. 1 Liquid Water and its Properties

Water Molecule Warm-up n Draw your water molecule in the space provided. n Label the hydrogen and oxygen on your water molecule. Describe what happens when your water molecule is brought near the water molecule of someone near you. n n Draw a picture of three water molecules interacting in the space provided.

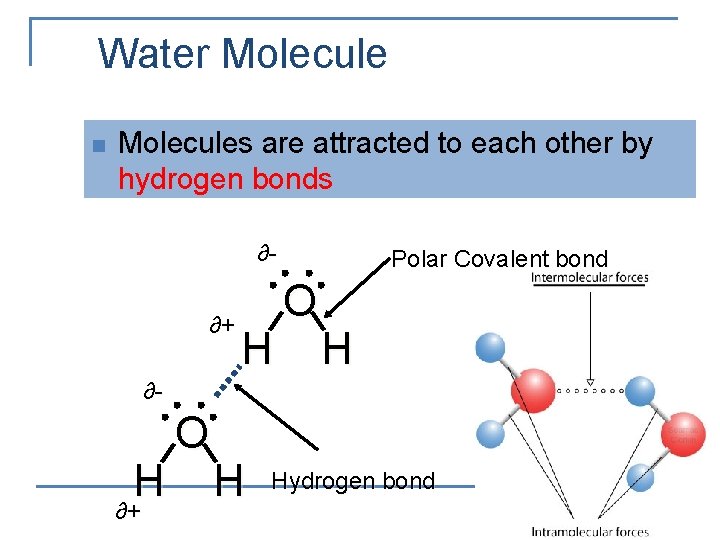
Water Molecule n n Water has polar bonds due to a difference in electronegativity of 1. 4. Water is a polar molecule and has a bent shape due to lone pair electrons on the central element, oxygen

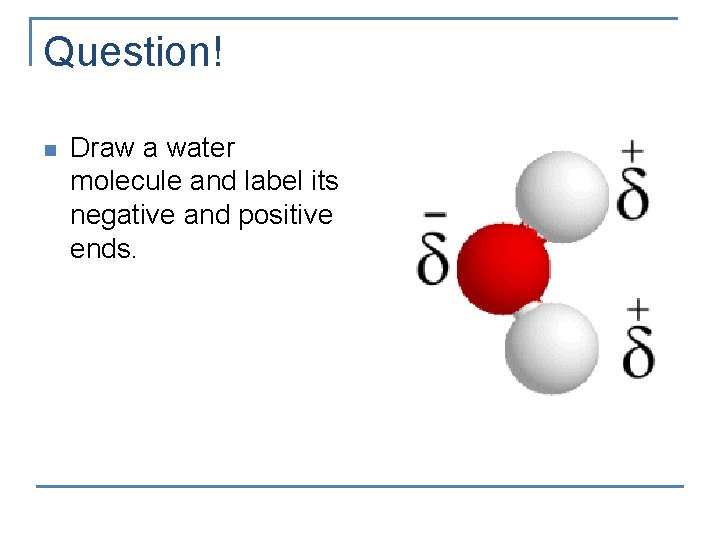
Question! n Draw a water molecule and label its negative and positive ends.

Water Molecule n Molecules are attracted to each other by hydrogen bonds ∂∂+ H Polar Covalent bond O H ∂- H ∂+ O H Hydrogen bond

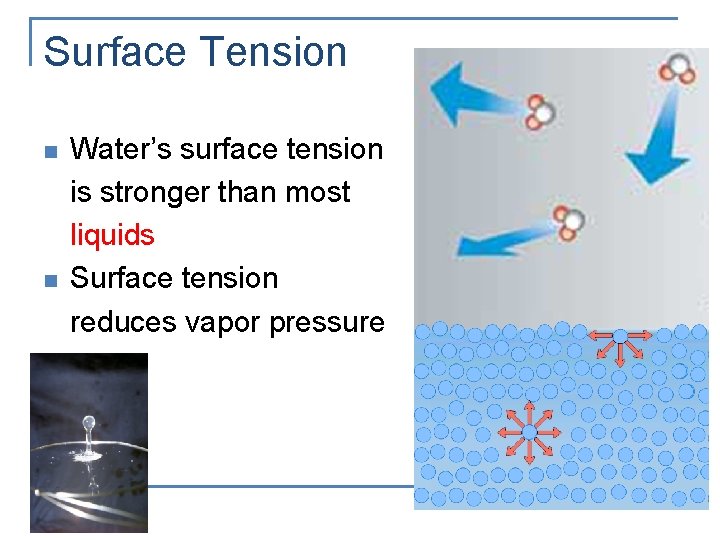
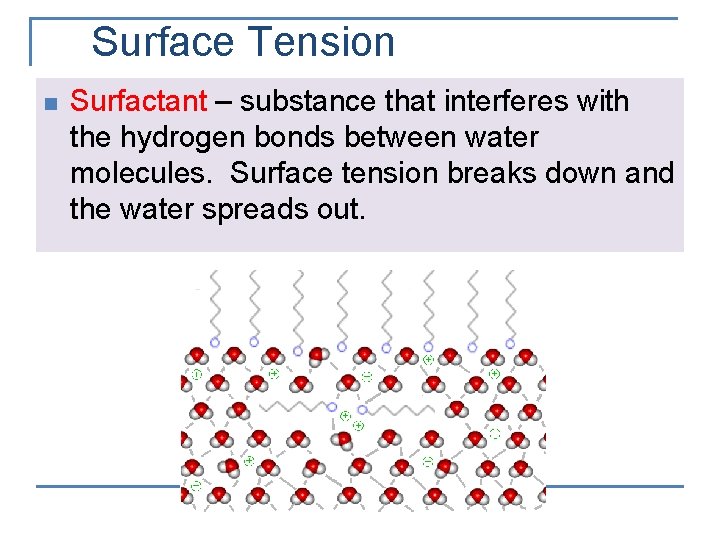
Surface Tension n The inward force, or pull, that tends to minimize the surface area of a liquid Tends to hold a drop of liquid in a spherical shape A sphere has the smallest surface area for a given volume

Question! n Draw arrows showing the pull of surface tension on the drop of water below.

Surface Tension n n Water’s surface tension is stronger than most liquids Surface tension reduces vapor pressure

Surface Tension n Surfactant – substance that interferes with the hydrogen bonds between water molecules. Surface tension breaks down and the water spreads out.

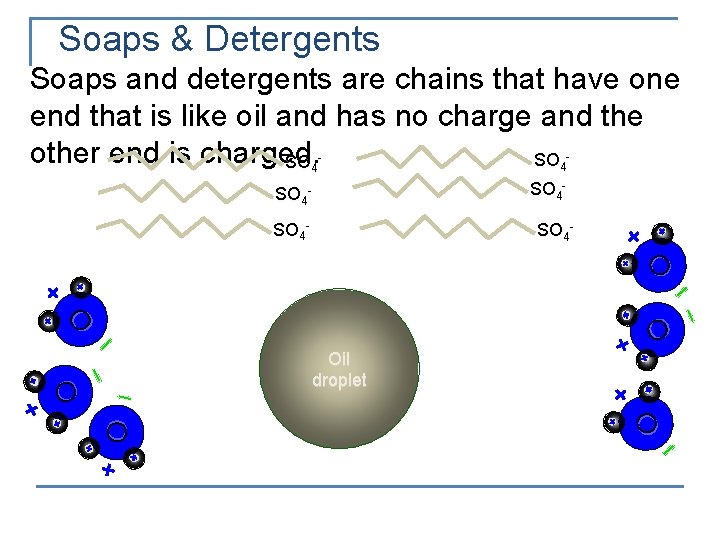
Soaps & Detergents Soaps and detergents are chains that have one end that is like oil and has no charge and the other end is charged. SO SO 4 SO 4 - 4 SO 4 - O O Oil droplet O O

Thermochemistry Review n n Specific heat capacity – the amount of energy required to raise the temperature of 1. 0 g of water by 1 C. q = m C T q C = specific heat capacity n q q q 1. 00 cal/g°C or 4. 18 J/g°C m = mass of substance in grams ∆T = change in temperature in °C q = heat in joules or calories

Question! n Why would a burn from steam at 100 C be worse than a burn from water at 100 C?

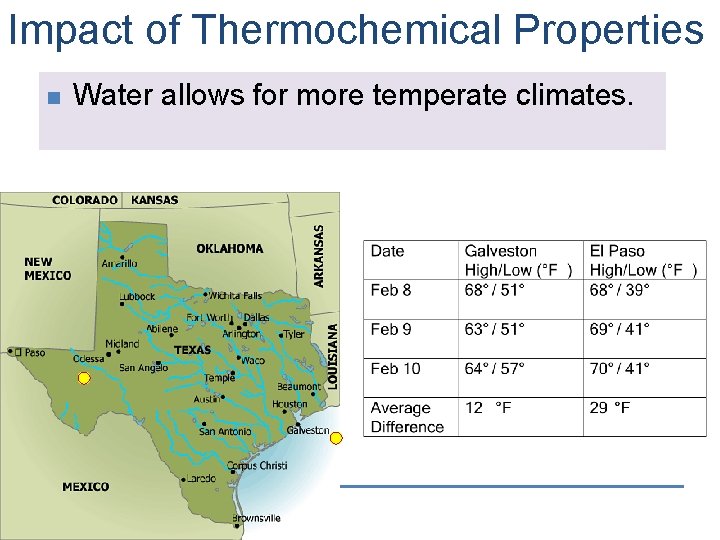
Impact of Thermochemical Properties n Water allows for more temperate climates.


Water Vapor n Evaporation – the change of state from liquid to gas at the surface of a liquid that is not boiling

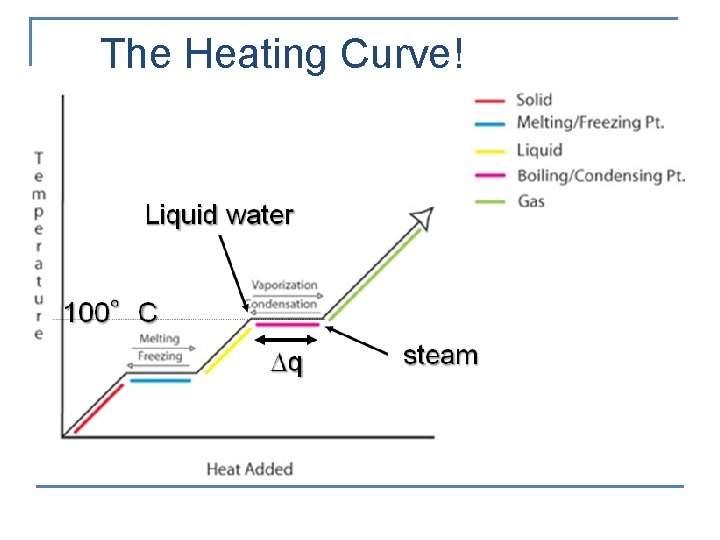
Water Vapor n Condensation – the change of state from a gas to a liquid n n n Water has a high heat of vaporization (2260 J/g) 2260 J of energy are needed to change 1. 0 g of water from a liquid to a gas or a gas to a liquid Water’s high boiling and melting points are caused by hydrogen bonding. When compared to ammonia (NH 3) the hydrogen bonds are stronger. It takes more energy to disrupt these bonds, giving water a higher boiling and melting point.

The Heating Curve!

Ice n Freezing – the change of state from a liquid to a solid q q Water has a high heat of fusion (334 J/g) 334 J of energy is needed to change 1. 0 g of water from a solid to a liquid or a liquid to a solid

Ice n Density – the ratio of the mass of an object to its volume M D= V

Question! n What happens when water freezes?

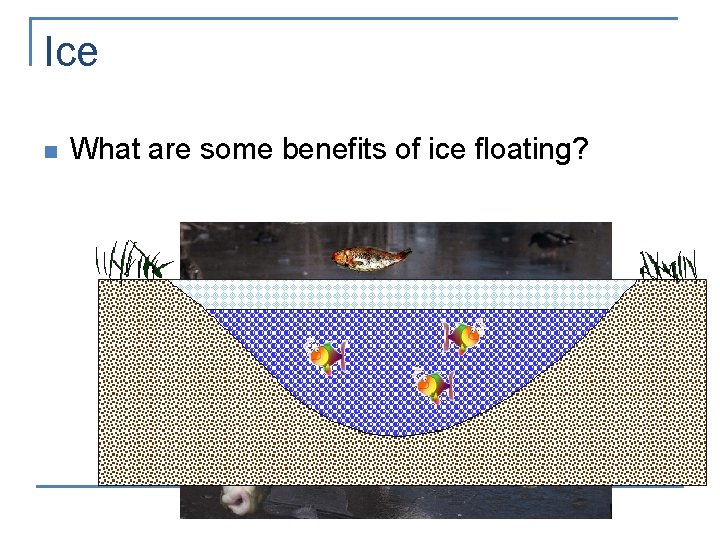
Ice n What are some benefits of ice floating?

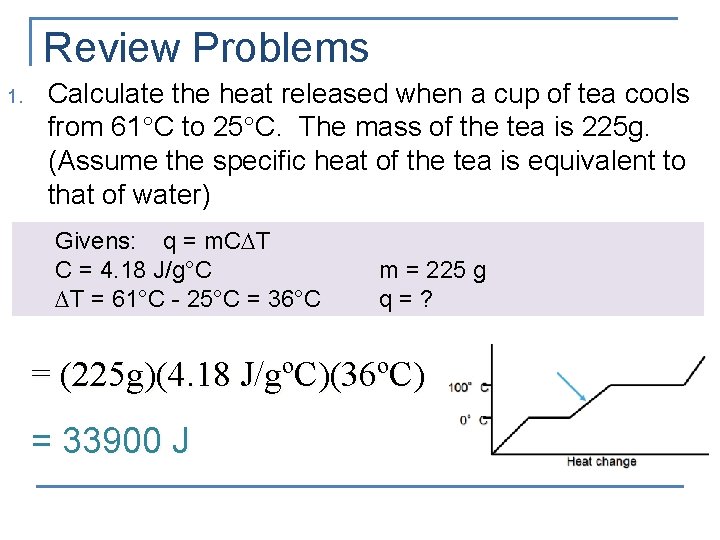
Review Problems 1. Calculate the heat released when a cup of tea cools from 61 C to 25 C. The mass of the tea is 225 g. (Assume the specific heat of the tea is equivalent to that of water) Givens: q = m. C T C = 4. 18 J/g°C ∆T = 61°C - 25°C = 36°C m = 225 g q=? = (225 g)(4. 18 J/gºC)(36ºC) = 33900 J

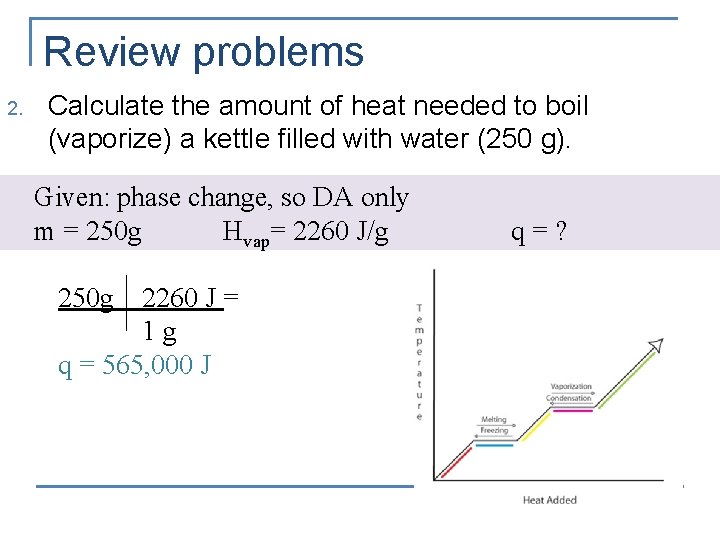
Review problems 2. Calculate the amount of heat needed to boil (vaporize) a kettle filled with water (250 g). Given: phase change, so DA only m = 250 g Hvap= 2260 J/g 250 g 2260 J = 1 g q = 565, 000 J q=?

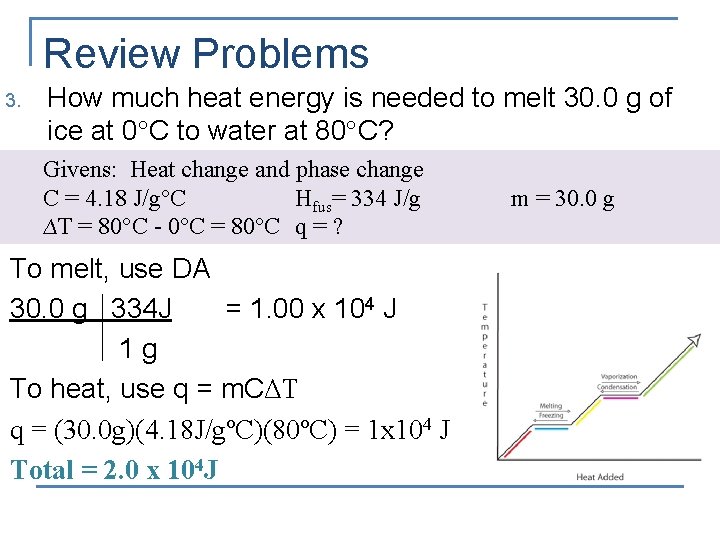
Review Problems 3. How much heat energy is needed to melt 30. 0 g of ice at 0 C to water at 80 C? Givens: Heat change and phase change C = 4. 18 J/g°C Hfus= 334 J/g ∆T = 80°C - 0°C = 80°C q = ? To melt, use DA 30. 0 g 334 J = 1. 00 x 104 J 1 g To heat, use q = m. CΔT q = (30. 0 g)(4. 18 J/gºC)(80ºC) = 1 x 104 J Total = 2. 0 x 104 J m = 30. 0 g

Review 4. Which atom in the water molecule is most electronegative? A. B. C. 5. Hydrogen Oxygen Neither How are water molecules held together? A. B. C. D. magic dispersion forces covalent bonds hydrogen bonds

Review 6. Water has a ________ vapor pressure due to _________. A. B. C. D. 7. low; low surface tension high; london dispersion forces low; hydrogen bonds high; hydrogen bonds Water has a ______ heat capacity and a _______ heat of vaporization. A. B. C. D. high; low high; high low; low

Review Due to the high heat capacity and vaporization energy of H 2 O, coastal areas have ___ temperatures. 8. A. B. C. D. 9. moderate extreme low high Which of the following are characteristics of surface tension? A. B. C. D. E. reduces vapor pressure holds liquids in spherical shapes is caused by hydrogen bonding is very strong in water all of the above

Review 10. A surfactant is a substance that interferes with ________ and decreases the _________. A. free time; productivity hydrogen bonding; surface tension intramolecular forces; vapor pressure B. C.