Chapter 15 Structure Determination by Spectroscopy II UVVis

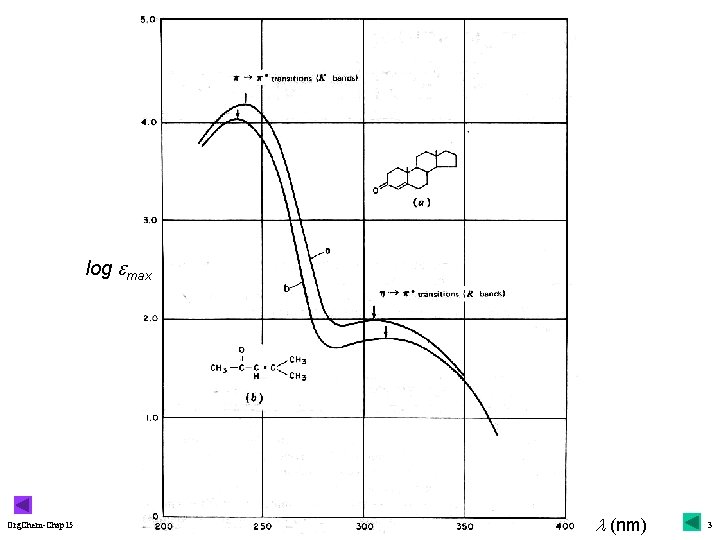
Chapter 15 Structure Determination by Spectroscopy (II): UV-Vis & Mass r Ultraviolet-Visible: u 200~800 u. A nm (143~36 kcal/mol): 610 Figure 15. 1 = e cl at lmax (e max, solvent ); 611 Figure 15. 2 uchromophore Org. Chem-Chap 15 electronic transitions (absorption part): 613 Figure 15. 3 malkanes: s s*, lmax< 200 nm, used as solvents mallowed: p p* (e >1, 000), forbidden: n p* (e <1, 000) 1

v Interpretation of UV-Vis Spectra r color: absorption in the visible region ucomplementary r indication color: 614 Table 15. 1 & bottom of conjugated double bonds usimilar absorption for the same chromophore: for structural identification: 616 top r UV-B/A: Org. Chem-Chap 15 295~320/320 -340 nm (sun-burn) usunscreen: ozone & PABA, 617 Focus On upractice: 616 Problem 15. 6 2
log emax Org. Chem-Chap 15 l (nm) 3

v Mass Spectrometry: Mw & Formula r molecular weight: molecular ion (M • +), 618 uspectrometer: uspectrum: r molecular u. HRMS: m. Mw m/z & base peak, 619 Figure 15. 5 formula: HRMS or isotope intensity atomic wt. vs exact mass, 619 Table 15. 2 44: CO 2, C 2 H 4 O, C 3 H 8 - different exact mass urelative intensity of isotope peaks: 620 Table 15. 3 m%[M+1] 1. 1 C + 0. 4 N; %[M+2] (1. 1 C)2/200 + 0. 2 O upractice: Org. Chem-Chap 15 only cation, 618 Figure 15. 4 621 -2 & 623 Problem 15. 10 4
![v Fragmentation Pattern (I): Stability r alkanes: [R-R’] • + [R]+ + [R’] • v Fragmentation Pattern (I): Stability r alkanes: [R-R’] • + [R]+ + [R’] •](http://slidetodoc.com/presentation_image_h2/50e4a53c5427c0c657dc840382b69223/image-5.jpg)
v Fragmentation Pattern (I): Stability r alkanes: [R-R’] • + [R]+ + [R’] • or [R] • + [R’]+ uoctane/2 -methylpentane: 625 -7 Figure 15. 8 -15. 9 r alkenes: allyl cation (m/z=41), 629 Figure 15. 10 r benzylic cleavage (m/z=91): 629 Figure 15. 11 upractice: Org. Chem-Chap 15 631 Practice 15. 2 5

v Fragmentation Pattern (II): Stability r alcohols: M-18(H 2 O) peak, 625 top ua-fission: 2 -butanol, 630 middle & Figure 15. 12 a-fission & Mc. Lafferty rearrangement, 630 & 631 & Figure 15. 13 r carbonyls: upractice: u. Focus Org. Chem-Chap 15 632 Problem 15. 14 On: GC/MS, 633 6
- Slides: 6