Chapter 15 Overview Equilibrium Reactions Equilibrium Constants Kc

Chapter 15

Overview • Equilibrium Reactions • Equilibrium Constants Kc & Kp • Equilibrium Expression product/reactant • Reaction Quotient Q • Calculations • Le Chatelier’s Principle – disturbing the equilibrium

Overview, cont’d • All reactions are reversible • Dynamic Equilibrium – When the rates of the forward and – reverse reactions are equal • Reactions do not “go to completion” • Cannot use stoichiometric methods to calculate amount of products formed
![Equilibrium and Rates A B kf [A] = kr [B] forward rate = reverse Equilibrium and Rates A B kf [A] = kr [B] forward rate = reverse](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-4.jpg)
Equilibrium and Rates A B kf [A] = kr [B] forward rate = reverse rate [B ] [A] = kf = constant kr

Equilibrium Constant Expression • Ratio of Products to Reactant – raised to each coefficient • for example, – N 2 + –K = [NH 3]2 [N 2][H 2]3 3 H 2 2 NH 3
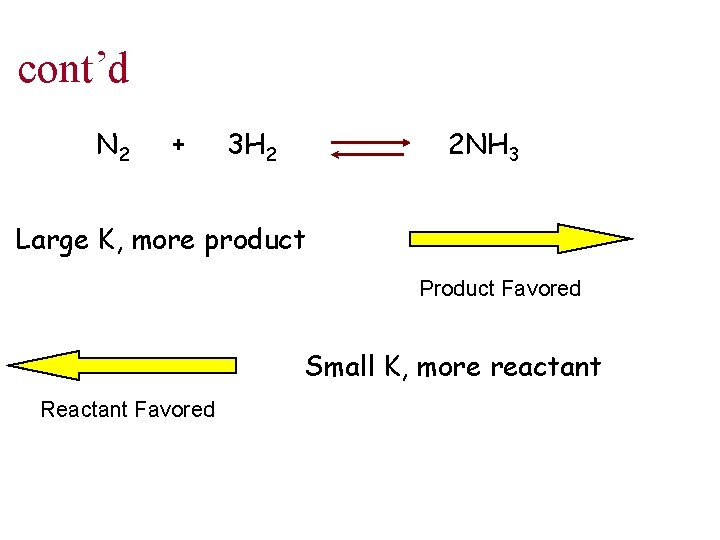
cont’d N 2 + 3 H 2 2 NH 3 Large K, more product Product Favored Small K, more reactant Reactant Favored
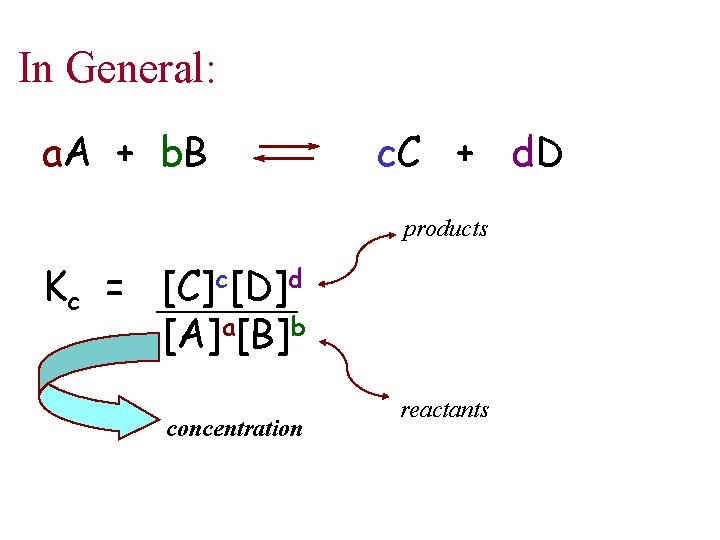
In General: a. A + b. B c. C + d. D products Kc = [C]c[D]d [A]a[B]b concentration reactants
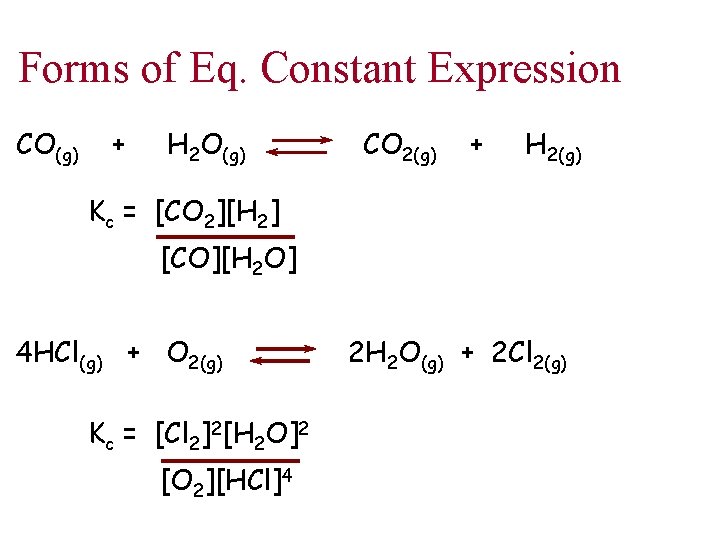
Forms of Eq. Constant Expression CO(g) + H 2 O(g) CO 2(g) + H 2(g) Kc = [CO 2][H 2] [CO][H 2 O] 4 HCl(g) + O 2(g) Kc = [Cl 2]2[H 2 O]2 [O 2][HCl]4 2 H 2 O(g) + 2 Cl 2(g)
![Cont’d 2 HI H 2 + I 2 Kf = [H 2][I 2] [HI]2 Cont’d 2 HI H 2 + I 2 Kf = [H 2][I 2] [HI]2](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-9.jpg)
Cont’d 2 HI H 2 + I 2 Kf = [H 2][I 2] [HI]2 H 2 + I 2 Kr 2 HI = [HI]2 [H 2][I 2] = 1 Kf
![Cont’d 2{ 2 HI H 2 + I 2 } Kf = [H 2][I Cont’d 2{ 2 HI H 2 + I 2 } Kf = [H 2][I](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-10.jpg)
Cont’d 2{ 2 HI H 2 + I 2 } Kf = [H 2][I 2] [HI]2 4 HI 2 H 2 + 2 I 2 K = Kf 2 = [H 2]2[I 2]2 [HI]4

Example N 2 O 4 2 NO 2 Initial N 2 O 4 Initial NO 2 Equilibrium N 2 O 4 0. 02 0. 213 0. 02 0. 03 0. 04 0. 0014 0. 0028 0. 0045 Kc = [NO 2]2 [N 2 O 4] 0. 0 Equilibrium NO 2 0. 0172 0. 0243 0. 0310 0. 0045 Kc 0. 211 0. 213 0. 0310 generally unitless
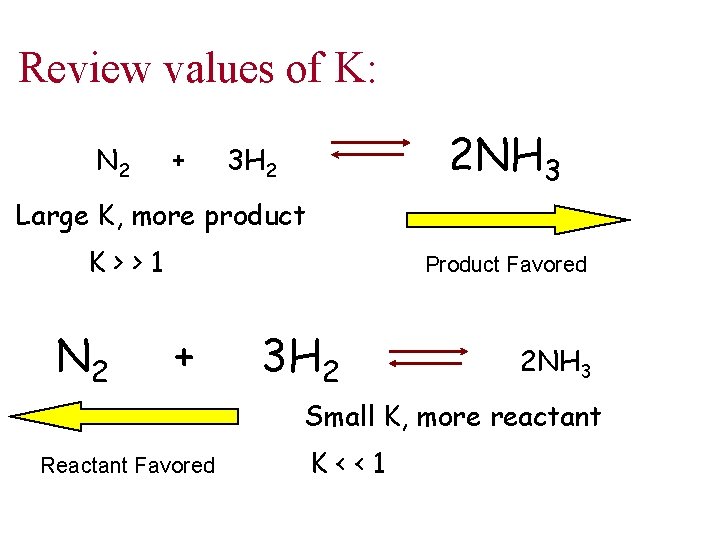
Review values of K: N 2 + 2 NH 3 3 H 2 Large K, more product K>>1 N 2 Product Favored + 3 H 2 2 NH 3 Small K, more reactant Reactant Favored K<<1
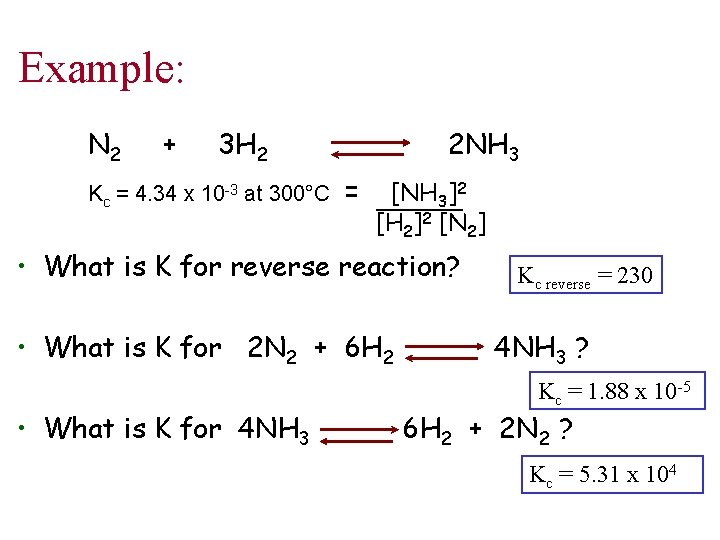
Example: N 2 + 3 H 2 Kc = 4. 34 x 10 -3 at 300°C 2 NH 3 = [NH 3]2 [H 2]2 [N 2] • What is K for reverse reaction? • What is K for 2 N 2 + 6 H 2 • What is K for 4 NH 3 Kc reverse = 230 4 NH 3 ? Kc = 1. 88 x 10 -5 6 H 2 + 2 N 2 ? Kc = 5. 31 x 104

Heterogeneous Equilibria: • When pure solid or liquid is involved – Pure solids & liquids do not appear in the equilibrium constant expression • When H 2 O is a reactant or product and is the solvent – H 2 O does not appear in the equilibrium constant expression

Examples: • Ca. CO 3(s) Ca. O(s) + CO 2(g) – conc. = mol = g/cm 3 = density cm 3 g/mol MM • Kc = [Ca. O] [CO 2] = (constant 1) [CO 2] [Ca. CO 3] (constant 2) • Kc’ = Kc (constant 2) = [CO 2] (constant 1) both are constant
![Multi-Step Equilibria Ag. Cl(s) Ag+(aq) + 2 NH 3(aq) [Ag(NH 3)2+] Cl-(aq) K 1 Multi-Step Equilibria Ag. Cl(s) Ag+(aq) + 2 NH 3(aq) [Ag(NH 3)2+] Cl-(aq) K 1](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-16.jpg)
Multi-Step Equilibria Ag. Cl(s) Ag+(aq) + 2 NH 3(aq) [Ag(NH 3)2+] Cl-(aq) K 1 = [Ag+][Cl-] Ag(NH 3)2+(aq) K 2 = [Ag+][NH 3]2 Ag. Cl(s) + 2 NH 3(aq) Ag(NH 3)2+(aq) + Cl-(aq) Ktot = K 1 K 2 = [Ag(NH 3)2+][Cl-] [NH 3]2

Problem: 1. 00 mole of H 2 & 1. 00 mole of I 2 are placed in a 1. 0 L container at 520 K and allowed to come to equilibrium. Analysis reveals 0. 12 mol of HI present at equilibrium. Calculate Kc. H 2(g) + I 2(g) 2 HI(g) initial 1. 00 change -0. 06 equil. Kc = 0. 94 mol (0. 12)2 = (0. 94) 1. 00 -0. 06 0 +0. 12 0. 94 mol +0. 12 1. 6 x 10 -2

Conversion between Kp and Kc • Kc – Equilibrium constant using concentrations • Kp – Equilibrium constant using partial pressures Kp = Kc (RT)Dn P = n RT V R = 0. 0821 L atm/mol K T = Temperature in K Dn = tot. mol product - tot. mol reactant

Problem: For 2 SO 3(g) 2 SO 2(g) + O 2(g) Kc = 4. 08 x 10 -3 at 1000 K. Calculate Kp. Kp = Kc(RT)Dn = 4. 08 x 10 -3 (0. 0821 x 1000)1 Kp = 0. 0335

Problem: For 3 H 2(g) + N 2(g) 2 NH 3(g) Kc = 0. 105 at 472°C. Calculate Kp. Kp = Kc(RT)Dn = 0. 105 (0. 0821 x 745)-2 Kp = 2. 81 x 10 -5

Applications of Eq. Constants • Reaction Quotient – Non-equilibrium concentrations used in the equilibrium constant expression Q = K Reaction is at equilibrium Q > K Reaction will shift left to equilibrium Q < K Reaction will shift right to equilibrium

Problem: Kc = 5. 6 x 10 -12 at 500 K for I 2(g) 2 I(g) [I 2] = 0. 020 M & [I] = 2. 0 x 10 -8 M. Is the reaction at equilibrium? Which direction will it shift to reach equilibrium? Q = [I ]2 [I 2] = Q (2. 0 x 10 -8)2 (0. 020) < (2. 0 x 10 -14) < = 2. 0 x 10 -14 not at equilibrium because Q K K (5. 6 x 10 -12) Reaction Shifts Right to get to equilibrium

Calculation of Eq. Concentrations • use the stoichiometry of reaction • initial concentration of all species • change that occurs to all species • equilibrium concentration of all species • reaction will occur to reach the equilibrium point no matter the direction of reaction
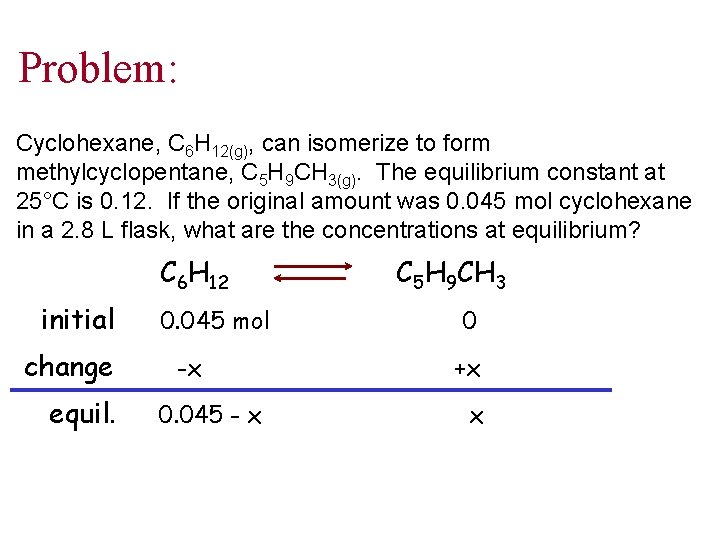
Problem: Cyclohexane, C 6 H 12(g), can isomerize to form methylcyclopentane, C 5 H 9 CH 3(g). The equilibrium constant at 25°C is 0. 12. If the original amount was 0. 045 mol cyclohexane in a 2. 8 L flask, what are the concentrations at equilibrium? C 6 H 12 initial change equil. 0. 045 mol -x 0. 045 - x C 5 H 9 CH 3 0 +x x

Cont’d 0. 12 = (x) (0. 045 - x) Solve for x which is the equilibrium concentration of methylcyclopentane or the product x = 4. 8 x 10 -3 mol C 5 H 9 CH 3 in 2. 8 L flask [C 5 H 9 CH 3] = 1. 7 x 10 -3 M [C 6 H 12] = 1. 4 x 10 -2 M
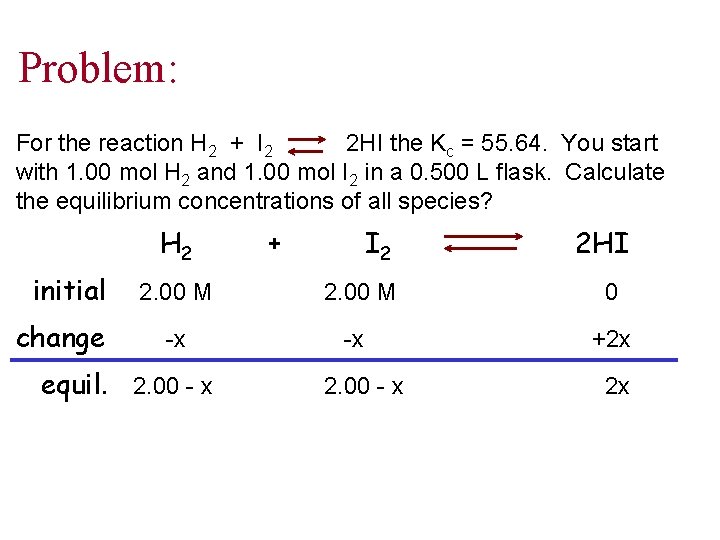
Problem: For the reaction H 2 + I 2 2 HI the Kc = 55. 64. You start with 1. 00 mol H 2 and 1. 00 mol I 2 in a 0. 500 L flask. Calculate the equilibrium concentrations of all species? H 2 initial change + I 2 2 HI 2. 00 M 0 -x -x +2 x equil. 2. 00 - x 2 x

Cont’d perfect square 55. 64 = (55. 64)½ = (2 x)2 (2. 00 -x)2 ½ x = 1. 58 M [HI] = 3. 16 M = 2 x [H 2] = 0. 42 M = 2. 00 - x [I 2] = 0. 42 M = 2. 00 - x
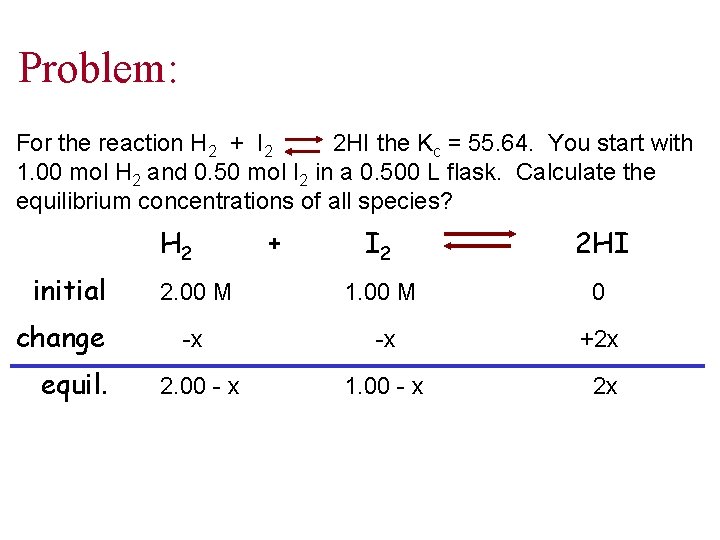
Problem: For the reaction H 2 + I 2 2 HI the Kc = 55. 64. You start with 1. 00 mol H 2 and 0. 50 mol I 2 in a 0. 500 L flask. Calculate the equilibrium concentrations of all species? H 2 initial change equil. + I 2 2 HI 2. 00 M 1. 00 M 0 -x -x +2 x 2. 00 - x 1. 00 - x 2 x
![Cont’d Kc 55. 64 = = [HI]2 [H 2][I 2] not a perfect square Cont’d Kc 55. 64 = = [HI]2 [H 2][I 2] not a perfect square](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-29.jpg)
Cont’d Kc 55. 64 = = [HI]2 [H 2][I 2] not a perfect square (2 x)2 (2. 00 -x)(1. 00 -x) reduces to a quadratic equation: x 2 - 3. 232 x + 2. 155 = 0

Quadratic Equation x =-b± b 2 - 4 ac 2 a ½ for ax 2 + bx + c = 0

Cont’d x = +3. 232 ± 10. 446 - 8. 62 2 ½ x = 1. 616 ± 0. 6755 [HI] = 1. 88 M = 2 x [H 2] = 1. 06 M = 2. 00 - x [I 2] = 0. 060 M = 1. 00 - x

Le Chatelier’s Principle • When a stress is applied to an equilibrium reaction, the equilibrium shifts to reduce the stress • Types of Stress – Addition or removal of reactant – Addition or removal or product – Increase or decrease of temperature – Change in pressure or volume
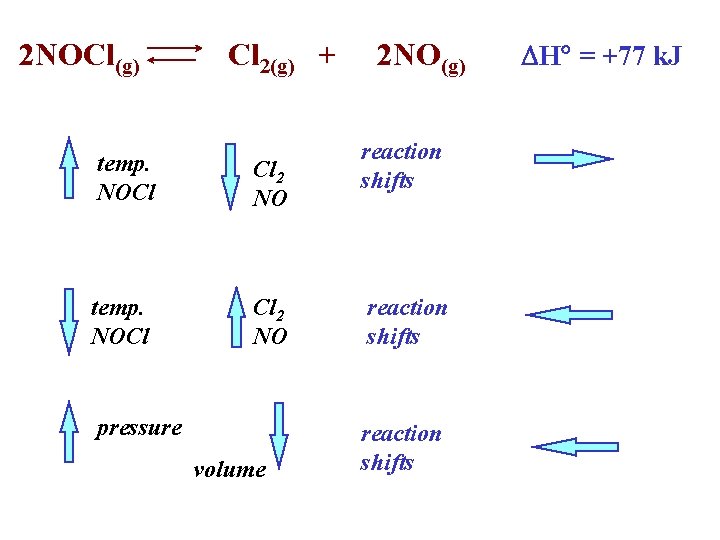
2 NOCl(g) Cl 2(g) + temp. NOCl Cl 2 NO pressure volume 2 NO(g) reaction shifts DH° = +77 k. J
![Addition of Reactant or Product [C 6 H 12] [C 5 H 9 CH Addition of Reactant or Product [C 6 H 12] [C 5 H 9 CH](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-34.jpg)
Addition of Reactant or Product [C 6 H 12] [C 5 H 9 CH 3] initial 1. 4 x 10 -2 + 1. 0 x 10 -2 M 1. 7 x 10 -3 M change -x equil. 2. 4 x 10 -2 - x 0. 12 = (1. 7 x 10 -3 + x) (2. 4 x 10 -2 - x) [C 6 H 12] = 0. 023 M +x 1. 7 x 10 -3 + x x = 1. 05 x 10 -3 M [C 5 H 9 CH 3] = 0. 0028 M

Changes in Temperature • will change K • for an endothermic reaction – increasing T increases K • for an exothermic reaction – increasing T decreases K
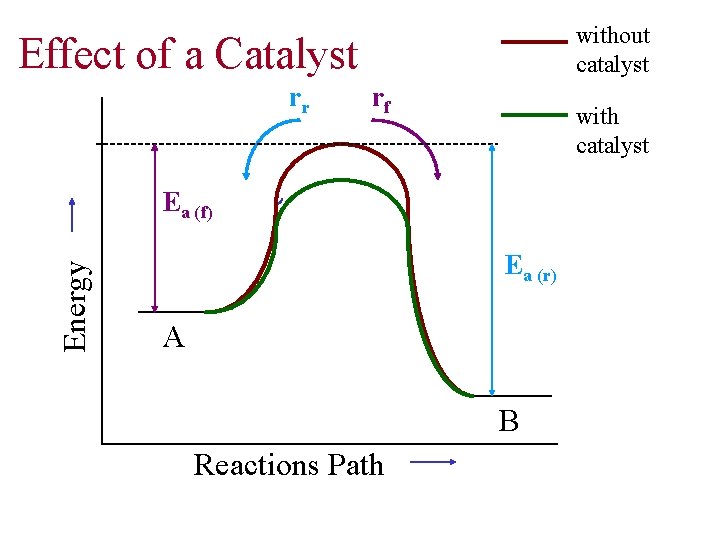
without catalyst Effect of a Catalyst rr rf with catalyst Energy Ea (f) Ea (r) A B Reactions Path

Reaction Mechanisms & Equilibria 2 O 3(g) O(g) k 1 k 2 + 3 O 2(g) overall rxn O 2(g) + O(g) O 3(g) k 3 fast equil. rate 1 = k 1[O 3] rate 2 = k 2[O 2][O] 2 O 2(g) slow rate 3 = k 3[O][O 3] rate 3 includes the conc. of an intermediate and the exptl. rate law will include only species that are present in measurable quantities
![Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-38.jpg)
Substitution Method at equilibrium k 1[O 3] = k 2[O 2][O] rate 3 =k 3[O][O 3] [O] = substitute rate 3 = k 3 k 1 [O 3]2 k 2 [O 2] overall rate or = k’ [O 3]2 [O 2] k 1 [O 3] k 2 [O 2]

Problem: Derive the rate law for the following reaction given the mechanism step below: OCl - (aq) + I + HOCl HOI + OH k 2 - OI (aq) k 1 OCl - + H 2 O I- - HOCl - + Cl (aq) + OH - k 3 HOI + k 4 H 2 O + OI - Cl - - (aq) fast slow fast
![Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 = Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 =](http://slidetodoc.com/presentation_image_h2/c65fe0541ff86871a4ce424722ecebbd/image-40.jpg)
Cont’d rate 1 = k 1 [OCl -][H 2 O] = rate 2 = k 2 [HOCl][OH -] [HOCl] = k 1[OCl -][H 2 O] k 2[OH -] rate 3 = k 3 [HOCl][I -] rate 3 = k 3 k 1[OCl -][H 2 O][I -] k 2 [OH -] overall rate = k’ [OCl -][I -] [OH -] solvent
- Slides: 40