Chapter 15 Ionic Bonding Valence Electrons Do the

Chapter 15 Ionic Bonding

Valence Electrons Ø Do the electron configuration for the following elements • Li • Be • B • O • F • Ne

Valence Electrons • Now look at the outermost electrons by looking at the last s and p orbital • Circle and count these electrons • These are the valence electrons

Valence electrons – Valence electrons are in the highest energy level of an element’s atom – Valence electrons are the same as group # (roman numeral) for the group A representative elements – Valence electrons are usually the only electrons used in chemical bonds – We use valence e- to write Lewis Dot Structures

Valence electrons (cont. ) A. Lewis Dot Structure Ex: bromine ● ● ● Br ● ●

Electron configurations for cations & anions A. Octet Rule: when forming compounds atoms want to have 8 electrons (s 2 p 6) like the noble gases (except He)

Cations (metals) tend to lose electrons, positively charged ion lose 1 e+ 2 2 6 1 2 2 6 Na 1 s 2 s 2 p 3 s Na (atom) (cation) 1 s 2 s 2 p octet Using the Dot Structure ● lose 1 e- Na (cation) (neutral atom) ● Mg (neutral atom) Na + ● lose 2 e- Mg (cation) 2+

Anions (Nonmetals) have negative charges because they gain electrons Cl gain 1 e- 1 s 22 p 63 s 23 p 5 (atom) Using the Dot Structure ●● ● Cl gain 1 O ● ● ● (neutral atom) octet ● ● e- ● ● Cl ● ● - ●● (chloride ion) (neutral atom) ● 1 s 22 p 63 s 23 p 6 (chloride ion) valence ● ● ● - gain 2 e- ● ● O ● ● ●● (oxide ion) 2 -
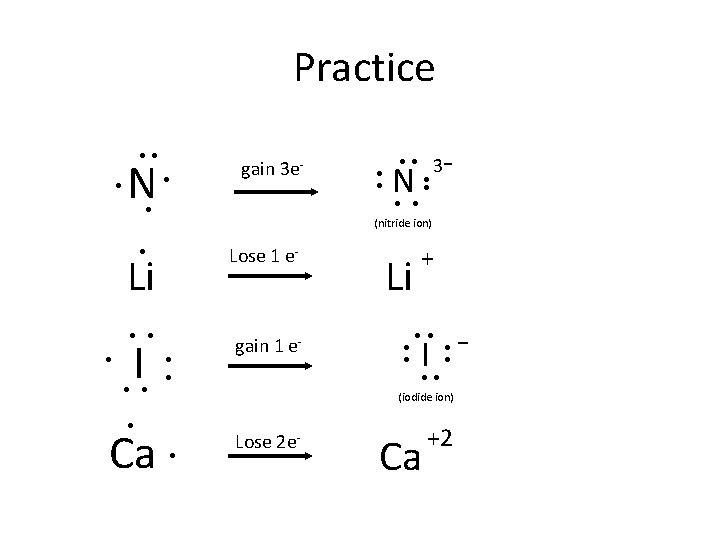
Practice ●● ● N ● gain 3 e- Lose 1 e- Li ● ● ● 3 - gain 1 e- Li ● ● + I - ● ● ●● (iodide ion) ● Ca N ● ● (nitride ion) ● I ●● ● ● ● Lose 2 e- Ca +2

Ionic Compound • Composed of a metal (cation) and nonmetal (anion) • Ionic bond – oppositely charged ions attract • Electrically neutral (+) = (-) A. Properties 1. ionic compounds form crystals 2. high melting and boiling points 3. hard and brittle 4. conduct electricity when dissolved in water or melted
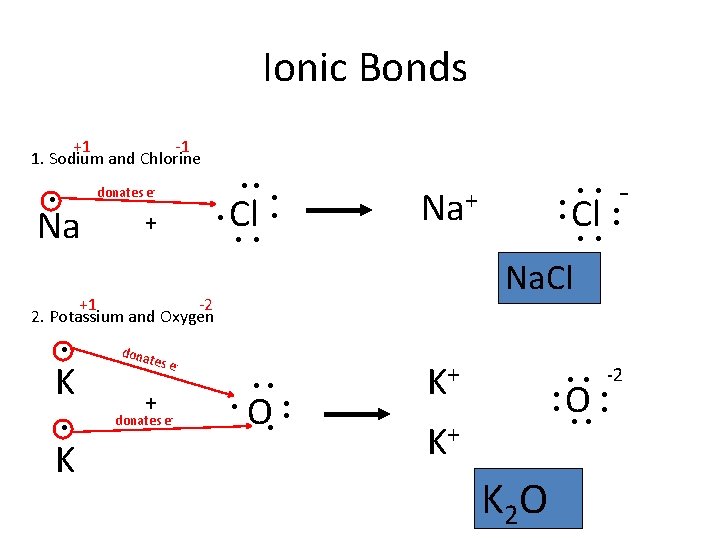
Ionic Bonds +1 -1 1. Sodium and Chlorine ●● donates e- ● Na + ● Cl Na+ ● ● K donates e- Cl ● ● Na. Cl dona tes e - + - ● ● ● +1 -2 2. Potassium and Oxygen ● ● ● O ● ● ● K+ K 2 O ● ● -2 ● ●

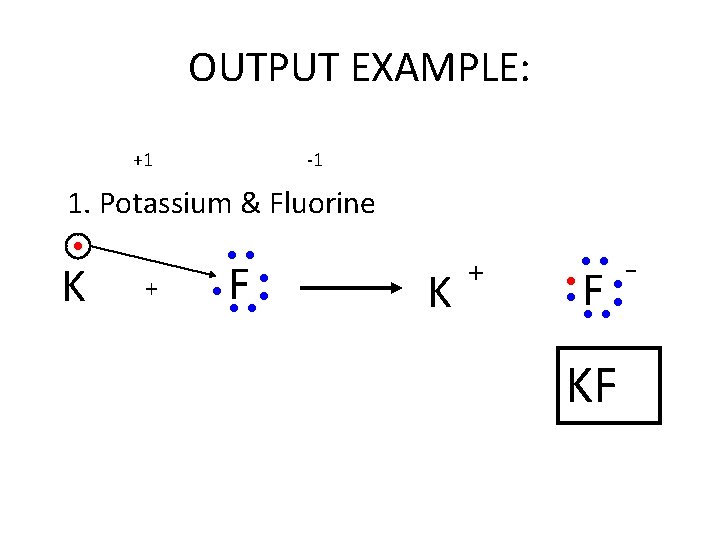
OUTPUT EXAMPLE: +1 -1 1. Potassium & Fluorine ● K ●● + ● F ●● ● ● K + ● ● ●● F ●● ● ● KF -
- Slides: 13