Chapter 15 Holt Chemical Reactions Using Vocabulary 1

Chapter 15 Holt Chemical Reactions

Using Vocabulary • 1. Adding a(n) inhibitor will slow down a chemical reactions. • 2. A chemical reaction that gives off light is called exothermic.

• 3. A chemical reaction that forms one compound from two or more substances is called a synthesis reaction.

• 4. The 2 in the formula Ag 2 S is a subscript. • 5. The staring materials in a chemical reaction are reactants.

Understanding Concepts • 6. Balancing a chemical equation so that the same number of atoms of each element is found in both the reactants and the products is an illustration of • C) the law of conservations of mass.

• 7. What is the correct chemical formula for calcium chloride? • B) Ca. Cl 2 • Ca has a valence of 2, so 2+ charge • Cl has a valence of 7, so -1 charge • So 2 Cl- with Ca++ makes Ca. Cl 2
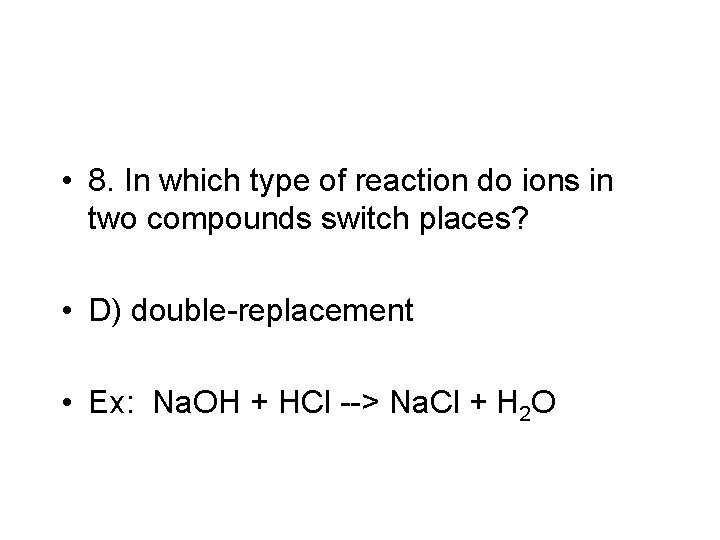
• 8. In which type of reaction do ions in two compounds switch places? • D) double-replacement • Ex: Na. OH + HCl --> Na. Cl + H 2 O

• 9. Which is an example of the use of activation energy? • C) holding a match to paper

• 10. Enzymes in your body act as catalysts. The role of enzymes is to • A) Increase the rate of chemical reactions (make the reactions happen faster)

Short Answers • 11. Classify each of the follwing reactions: • A) Fe + O 2 --> Fe 2 O 3 synthesis • Balanced this would be • 4 Fe + 3 O 2 --> 2 Fe 2 O 3 • Iron + oxygen --> rust

• • • B) Al + Cu. SO 4 --> Al 2(SO 4)3 + Cu single replacement Balanced this is 2 Al + 3 Cu. SO 4 --> Al 2(SO 4)3 + 3 Cu Aluminum + cupric sulfide --> aluminum sulfide plus copper

• C) Ba(CN)2 + H 2 SO 4 --> Ba. SO 4 +HCN • Double replacement • Balanced this is • Ba(CN)2 + H 2 SO 4 --> Ba. SO 4 + 2 HCN

• 12. Name two ways that you could increase the rate of a chemical reaction. • 1) raise the temperature • ( this increases the concentration and/or the surface area) • 2) add a catalyst

• 13. The reactants are acetic acid (vinegar) and baking soda (sodium bicarbonate). • The products are carbon dioxide CO 2 , water H 2 O, and sodium acetate.

• • 14. Concept map is on the white board. Terms are: Chemical reaction Chemical equation Chemical formulas Reactants, products, Coefficients, subscripts

Critical Thinking & Problem Solving • 15. Your friend is very worried by rumors he has heard about a substance called dihydrogen monoxide. What could you say to your friend to calm his fears? • Dihydrogen H 2 • Monoxide O • So H 2 O - not to worry - its water !

• 16. �As long as proper safety precautions have been taken, why can explosives be transported long distances without exploding? • Explosives need to absorb energy to being the reaction. Without this absorption, they can be transported safely.

Math in Science • 17. Calculate the number of atoms of each element shown in each of the following: • A) Ca. SO 4 • Ca - Calcium - 1 atom • S - Sulfur - 1 atom • O - Oxygen - 4 atoms

• • B) 4 Na. OCl Na - sodium - 4 atoms O - oxygen - 4 atoms Cl - chlorine - 4 atoms

• • C) Fe(NO 3)2 Fe - iron - 1 atom N - nitrogen - 2 atoms O - oxygen - 6 atoms

• D) 2 Al 2(CO 3)3 • Al - Aluminum - 4 atoms • C - carbon - 6 atoms • O - oxygen - 18 atoms

• 18. Write balanced equations for the following: • A) Fe + O 2 --> Fe 2 O 3

• 18. Write balanced equations for the following: • A) Fe + O 2 --> Fe 2 O 3 • Answer: • 4 Fe + 3 O 2 --> 2 Fe 2 O 3

• B) Al + Cu. SO 4 --> Al 2(SO 4)3 + Cu • Answer

• B) Al + Cu. SO 4 --> Al 2(SO 4)3 + Cu • Answer • 2 Al + 3 Cu. SO 4 --> Al 2(SO 4)3 + 3 Cu
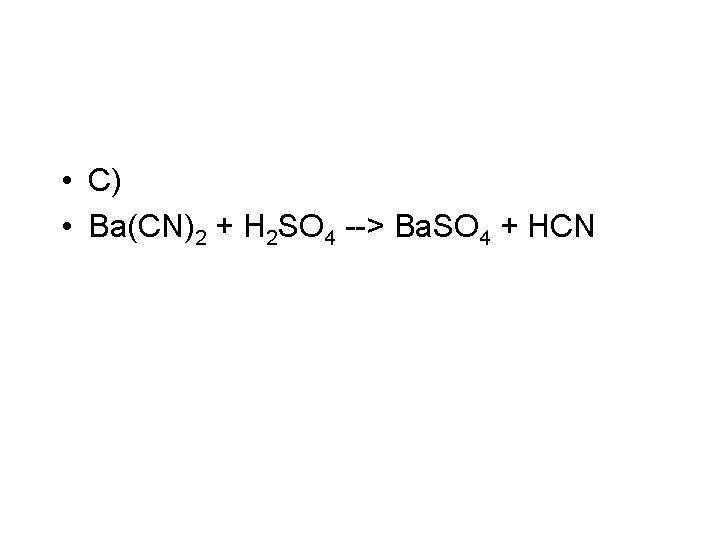
• C) • Ba(CN)2 + H 2 SO 4 --> Ba. SO 4 + HCN
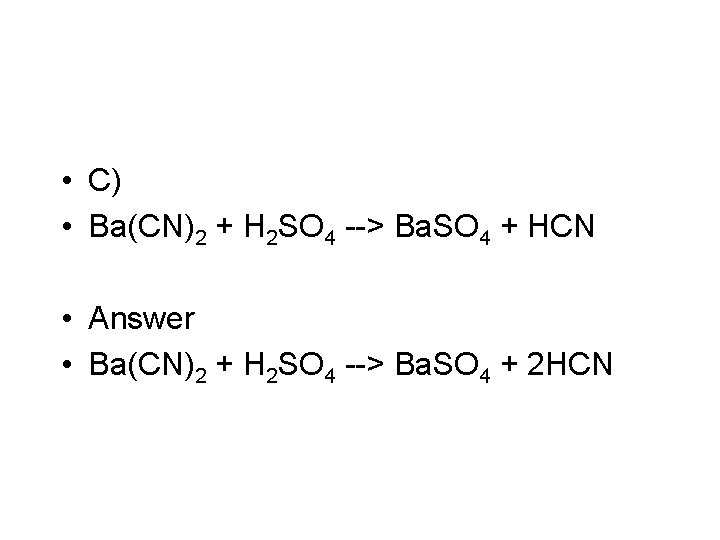
• C) • Ba(CN)2 + H 2 SO 4 --> Ba. SO 4 + HCN • Answer • Ba(CN)2 + H 2 SO 4 --> Ba. SO 4 + 2 HCN
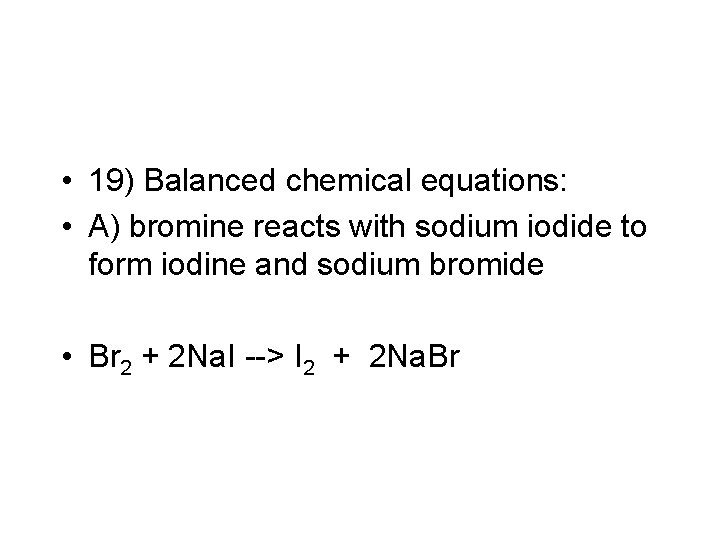
• 19) Balanced chemical equations: • A) bromine reacts with sodium iodide to form iodine and sodium bromide • Br 2 + 2 Na. I --> I 2 + 2 Na. Br
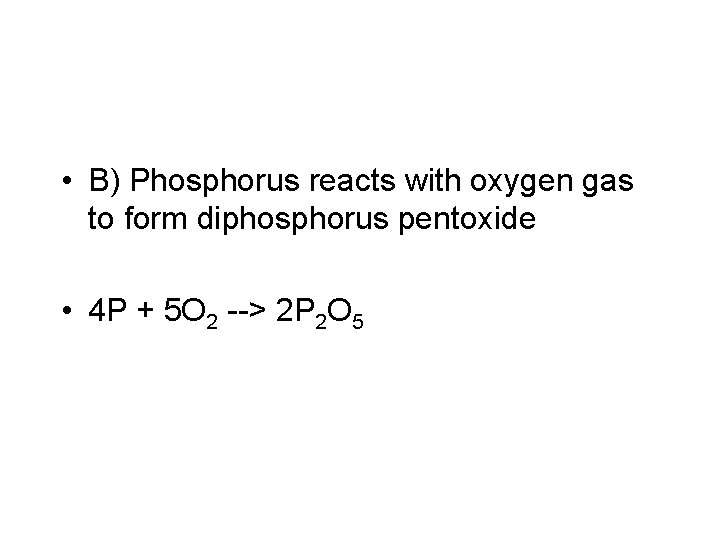
• B) Phosphorus reacts with oxygen gas to form diphosphorus pentoxide • 4 P + 5 O 2 --> 2 P 2 O 5

• C) Lithium oxide decomposes to form lithium and oxygen • 2 Li 2 O --> 4 Li + O 2

Interpreting Graphics • 20. In the photo on page 393, what evidence supports that this is a chemical reaction? • Answer • - Bubbles of gas are produced • - Light is given off

• 21. Using the energy diagram on page 393, answer q’s a, b, c • A) “C” represents the energy of the products (far right section of graph) • B) “B” represents the activation energy of the reaction (increases then peaks) • C) Energy is “given off” by this reaction. • That’s the decline of energy below starting energy mark - “D” on the chart)

• The end
- Slides: 33