CHAPTER 15 Electrochemistry 15 3 Balancing Redox Equations

CHAPTER 15 Electrochemistry 15. 3 Balancing Redox Equations

Redox reactions Zn(s) + Cu. SO 4 → Zn. SO 4(aq) + Cu(s) Cu. SO 4(aq) Zn(s) 2 Cu(s) deposit 15. 3 Balancing Redox Equations

Redox reactions Zn(s) + Cu. SO 4 → Zn. SO 4(aq) + Cu(s) We saw that this is a redox reaction in which: some elements lose electrons; they are oxidized other elements gain electrons; they are reduced We learned how to determine oxidation numbers 3 15. 3 Balancing Redox Equations

Redox reactions Zn(s) + Cu. SO 4 → Zn. SO 4(aq) + Cu(s) We saw that this is a redox reaction in which: some elements lose electrons; they are oxidized other elements gain electrons; they are reduced We learned how to determine oxidation numbers Now we look at how to balance redox reactions 4 15. 3 Balancing Redox Equations

Two methods At the end, both mass and charge have to be balanced There are two methods: 1) The oxidation number method 2) The half-reaction method 5 15. 3 Balancing Redox Equations

The oxidation number method Increase in oxidation number for oxidized atoms = Decrease in oxidation number for reduced atoms Step 1 Assign oxidation numbers for all atoms Step 2 Identify the atoms that are oxidized, and atoms that are reduced Step 3 Adjust coefficients for atoms whose oxidation numbers change, then make sure that the rule above is observed Step 4 6 Check the overall mass balance 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) 7 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 1 Assign oxidation numbers for all atoms H N O 3 + Cu 2 O → Cu (N O 3)2 + N O + H 2 O +1 +5 – 2 8 +1 – 2 +2 +5 – 2 +2 – 2 +1 – 2 15. 3 Balancing Redox Equations
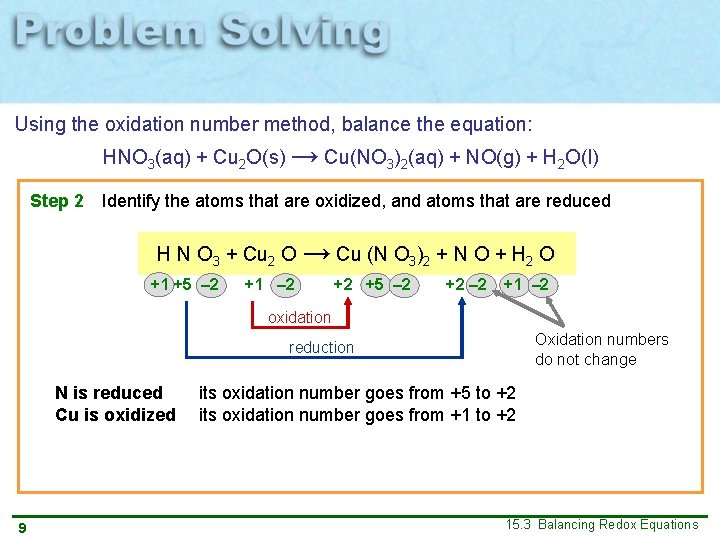
Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 2 Identify the atoms that are oxidized, and atoms that are reduced H N O 3 + Cu 2 O → Cu (N O 3)2 + N O + H 2 O +1 +5 – 2 +1 – 2 +2 +5 – 2 +2 – 2 +1 – 2 oxidation Oxidation numbers do not change reduction N is reduced Cu is oxidized 9 its oxidation number goes from +5 to +2 its oxidation number goes from +1 to +2 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 3 Balance all atoms whose oxidation numbers have changed H N O 3 + Cu 2 O → 2 Cu (N O 3)2 + N O + H 2 O +1 +5 – 2 +1 – 2 +2 +5 – 2 +2 – 2 +1 – 2 oxidation reduction N is reduced Cu is oxidized its oxidation number goes from +5 to +2 its oxidation number goes from +1 to +2 N is already balanced: 1 atom on each side Cu needs to be adjusted with a coefficient of 2 10 15. 3 Balancing Redox Equations
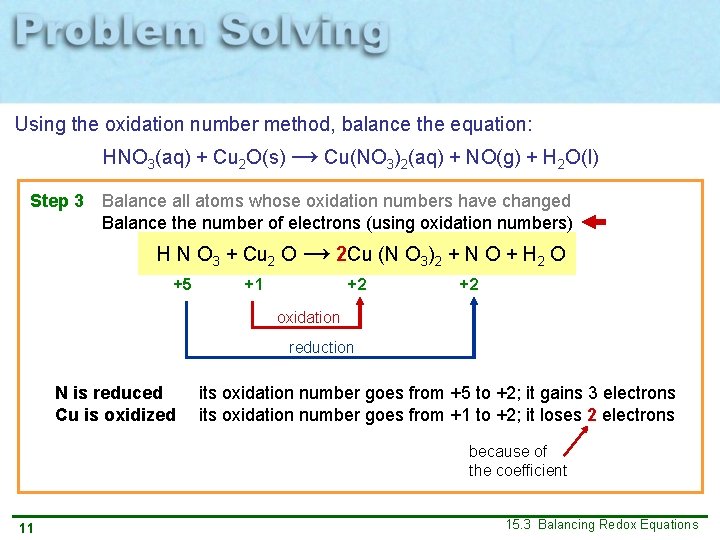
Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 3 Balance all atoms whose oxidation numbers have changed Balance the number of electrons (using oxidation numbers) H N O 3 + Cu 2 O → 2 Cu (N O 3)2 + N O + H 2 O +1 +5 – 2 +1 – 2 +2 +5 – 2 +2 – 2 +1 – 2 oxidation reduction N is reduced Cu is oxidized its oxidation number goes from +5 to +2; it gains 3 electrons its oxidation number goes from +1 to +2; it loses 2 electrons because of the coefficient 11 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 3 Balance all atoms whose oxidation numbers have changed Balance the number of electrons (using oxidation numbers) H N O 3 + Cu 2 O → 2 Cu (N O 3)2 + N O + H 2 O +1 +5 – 2 +1 – 2 +2 +5 – 2 +2 – 2 +1 – 2 e– + 3 e– N is reduced Cu is oxidized its oxidation number goes from +5 to +2; it gains 3 electrons its oxidation number goes from +1 to +2; it loses 2 electrons The number of electrons transferred must be the same 12 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 3 Balance all atoms whose oxidation numbers have changed Balance the number of electrons (using oxidation numbers) 2 H N O 3 + 3 Cu 2 O → 6 Cu (N O 3)2 + 2 N O + H 2 O +1 +5 – 2 +1 – 2 +2 +5 – 2 +2 (– 2 e–) x 3 = – 6 e– (+ 3 e–) x 2 = +6 e– N is reduced Cu is oxidized its oxidation number goes from +5 to +2; it gains 3 electrons its oxidation number goes from +1 to +2; it loses 2 electrons The number of electrons transferred must be the same 13 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 4 Check the overall mass balance 12 HNO 3 + 3 Cu 2 O → 6 Cu(NO 3)2 + 2 NO + 7 H 2 O H N Cu O 2 2 6 7 H 2 N 14 Cu 6 O 39 Masses are not balanced yet Start balancing N 14 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 4 Check the overall mass balance 14 HNO 3 + 3 Cu 2 O → 6 Cu(NO 3)2 + 2 NO + 7 H 2 O H 14 N 14 Cu 6 O 45 H 2 N 14 Cu 6 O 39 Masses are not balanced yet Now balance O and H 15 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Step 4 Check the overall mass balance 14 HNO 3 + 3 Cu 2 O → 6 Cu(NO 3)2 + 2 NO + 7 H 2 O H 14 N 14 Cu 6 O 45 Masses are now balanced 16 15. 3 Balancing Redox Equations

Using the oxidation number method, balance the equation: HNO 3(aq) + Cu 2 O(s) → Cu(NO 3)2(aq) + NO(g) + H 2 O(l) Answer: 14 HNO 3 + 3 Cu 2 O → 6 Cu(NO 3)2 + 2 NO + 7 H 2 O 17 15. 3 Balancing Redox Equations

Two methods At the end, both mass and charge have to be balanced There are two methods: 1) The oxidation number method 2) The half-reaction method 18 The redox reaction is split into two half-reactions: the oxidation reaction, and the reduction reaction. 15. 3 Balancing Redox Equations

The half-reaction method Step 1 19 Write the unbalanced equation showing explicitly all ions 15. 3 Balancing Redox Equations

The half-reaction method 20 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) 15. 3 Balancing Redox Equations

The half-reaction method 21 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions 15. 3 Balancing Redox Equations

The half-reaction method 22 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ 15. 3 Balancing Redox Equations

The half-reaction method 23 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ Step 5 Balance the charge for both half-reactions by adding electrons 15. 3 Balancing Redox Equations

The half-reaction method 24 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ Step 5 Balance the charge for both half-reactions by adding electrons Step 6 Adjust coefficients to balance the number of electrons transferred 15. 3 Balancing Redox Equations

The half-reaction method 25 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ Step 5 Balance the charge for both half-reactions by adding electrons Step 6 Adjust coefficients to balance the number of electrons transferred Step 7 Combine the two half-reactions, and add the spectator ions 15. 3 Balancing Redox Equations

The half-reaction method 26 Step 1 Write the unbalanced equation showing explicitly all ions Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Step 3 Write down the two unbalanced half-reactions Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ Step 5 Balance the charge for both half-reactions by adding electrons Step 6 Adjust coefficients to balance the number of electrons transferred Step 7 Combine the two half-reactions, and add the spectator ions Step 8 Simplify and check that both mass and charge are balanced 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 1 Write the unbalanced equation showing explicitly all ions Zn(s) + Cu 2+(aq) + SO 4– 2(aq) → Zn 2+(aq) + SO 4– 2(aq) + Cu(s) 27 15. 3 Balancing Redox Equations
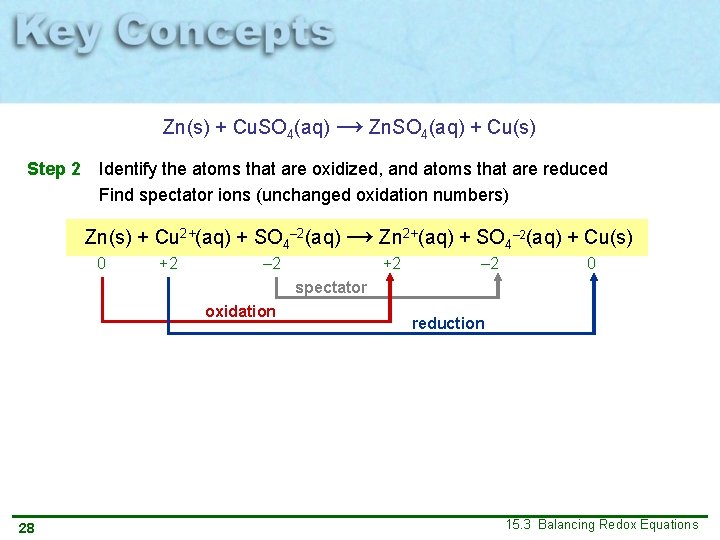
Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 2 Identify the atoms that are oxidized, and atoms that are reduced Find spectator ions (unchanged oxidation numbers) Zn(s) + Cu 2+(aq) + SO 4– 2(aq) → Zn 2+(aq) + SO 4– 2(aq) + Cu(s) 0 +2 – 2 0 spectator oxidation 28 reduction 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 3 Write down the unbalanced half-reactions Zn(s) + Cu 2+(aq) + SO 4– 2(aq) → Zn 2+(aq) + SO 4– 2(aq) + Cu(s) 0 +2 – 2 oxidation 29 +2 – 2 0 reduction Oxidation: Reduction: Zn(s) → Zn 2+(aq) Cu 2+(aq) → Cu(s) 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 4 Balance mass with elements other than oxygen and hydrogen Balance oxygen by adding H 2 O, then balance hydrogen with H+ Oxidation: Reduction: Zn(s) → Zn 2+(aq) Cu 2+(aq) → Cu(s) mass already balanced 30 mass already balanced 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 5 31 Balance the charge for both half-reactions by adding electrons Oxidation: Reduction: Zn(s) → Zn 2+(aq) + 2 e– Cu 2+(aq) + 2 e– → Cu(s) 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 6 Adjust coefficients to balance the number of electrons transferred Oxidation: Reduction: Zn(s) → Zn 2+(aq) + 2 e– Cu 2+(aq) + 2 e– → Cu(s) already the same in both half-reactions 32 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 7 Combine the two half-reactions, and add the spectator ions Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) 33 Oxidation: Reduction: Zn(s) → Zn 2+(aq) + 2 e– Cu 2+(aq) + 2 e– → Cu(s) 15. 3 Balancing Redox Equations

Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Step 8 Simplify and check that both mass and charge are balanced Zn(s) + Cu. SO 4(aq) → Zn. SO 4(aq) + Cu(s) Using the half-reaction method, we were able to determine that 2 electrons were transferred The full equation could not provide us with that information 34 15. 3 Balancing Redox Equations

At the end, both mass and charge have to be balanced There are two methods: 1) The oxidation number method 2) The half-reaction method 35 15. 3 Balancing Redox Equations
- Slides: 35