Chapter 15 Chemical Equilibrium Tro Chapter 15 Equilibrium

Chapter 15 Chemical Equilibrium Tro - Chapter 15

Equilibrium vs. Disequilibrium • when systems are at equilibrium with their surroundings, their conditions are the same as the surroundings and they stay that way • when systems are in disequilibrium with their surroundings, their conditions are not the same as the surroundings • systems that are in disequilibrium tend to change until they reach equilibrium with their surroundings • living things are in controlled disequilibrium with their environment – they are not at the same conditions as the environment and do not tend to change toward those conditions Tro - Chapter 15 2

Reaction Rates • some chemical reactions proceed rapidly ü like the precipitation reactions studied in Chp 7, where the products form practically the instant the two solutions are mixed • other reactions proceed slowly ü like the decomposition of dye molecules of a sofa placed in front of a window • the rate of a reaction is measured in the amount of reactant that changes into product in a given period of time ü generally moles of reactant used per second Ø like miles per hour • chemists study ways of controlling reaction rates Tro - Chapter 15 3

Reaction Rates After 4530 seconds, secondsthe theleft right left reaction complete, stillcomplete, not complete Initially, only reactants are present. After 15 seconds, reaction isis 60% whereas thereaction right reaction onlycomplete 40% done but the right is only is 20% Tro - Chapter 15 4

2 N 2 O 5 (g) → 4 NO 2(g) + O 2(g) Over time, the concentrations of reactants decrease as products increase. Tro - Chapter 15 5

2 N 2 O 5 (g) → 4 NO 2(g) + O 2(g) Rate vs. Time Because reactant concentrations decrease, the rates of reactions slow down over time. Tro - Chapter 15 6

Collision Theory • in order for a reaction to take place, the reacting molecules must collide with each other. • once molecules collide they may react together or they may not, depending on two factors 1. whether the collision has enough energy to “start to break the bonds holding reactant molecules together"; 2. whether the reacting molecules collide in the proper orientation for new bonds to form. Tro - Chapter 15 7

Effective Collisions • collisions in which these two conditions are met (and therefore the reaction occurs) are called effective collisions. • the higher the frequency of effective collisions the faster the reaction rate. • there is a minimum energy needed for a collision to be effective – we call this the activation energy üthe lower the activation energy, the faster the reaction will be Tro - Chapter 15 8

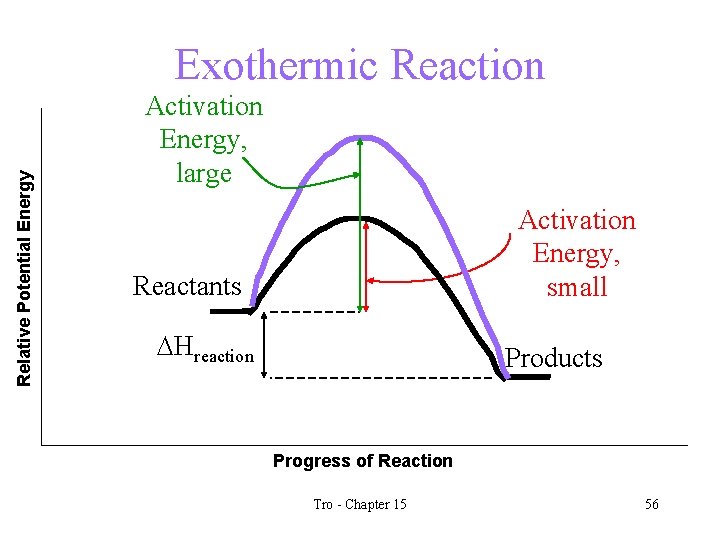
Activation Energy • the energy barrier that prevents any collision between molecules from being an effective collision is called the activation energy • the larger the activation energy of a reaction, the slower it will be üat a given temperature Tro - Chapter 15 9
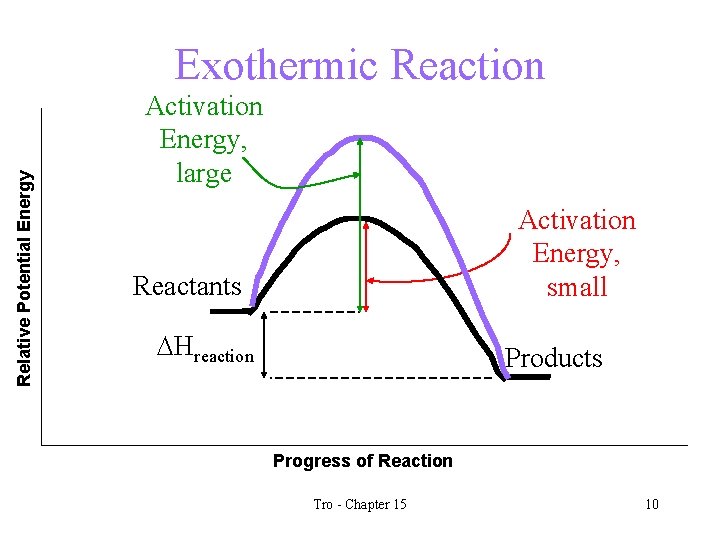
Relative Potential Energy Exothermic Reaction Activation Energy, large Activation Energy, small Reactants DHreaction Products Progress of Reaction Tro - Chapter 15 10
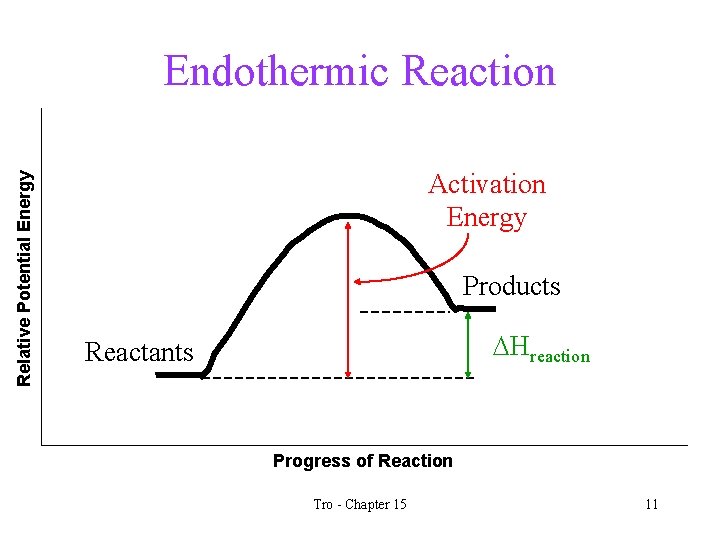
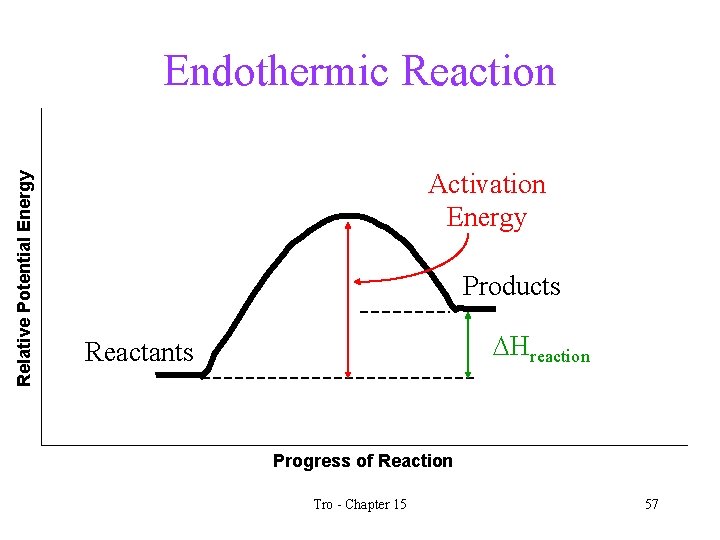
Relative Potential Energy Endothermic Reaction Activation Energy Products DHreaction Reactants Progress of Reaction Tro - Chapter 15 11

Factors Effecting Reaction Rate Reactant Concentration • the higher the concentration of reactant molecules, the faster the reaction will generally go. üincreases the frequency of reactant molecule collisions • since reactants are consumed as the reaction proceeds, the speed of a reaction generally slows over time Tro - Chapter 15 12
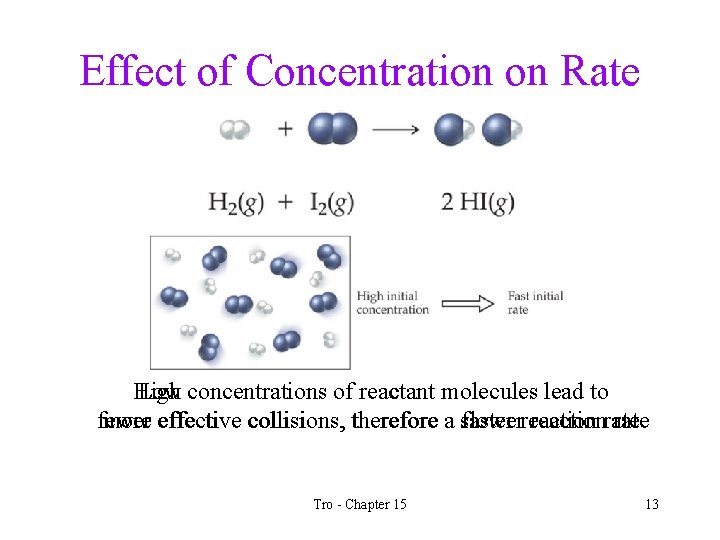
Effect of Concentration on Rate High Low concentrations of reactant molecules lead to fewer more effective collisions, therefore a slower faster reactionrate Tro - Chapter 15 13

Factors Effecting Reaction Rate Temperature • increasing temperature increases the reaction rate ü for each 10°C rise in temperature, the speed of the reaction generally doubles • increasing the temperature increases the number of molecules in the sample with enough energy so that their collisions can overcome the activation energy • increasing the temperature also increases the frequency of collisions – so the rate increases because the frequency of effective collisions increases Tro - Chapter 15 14
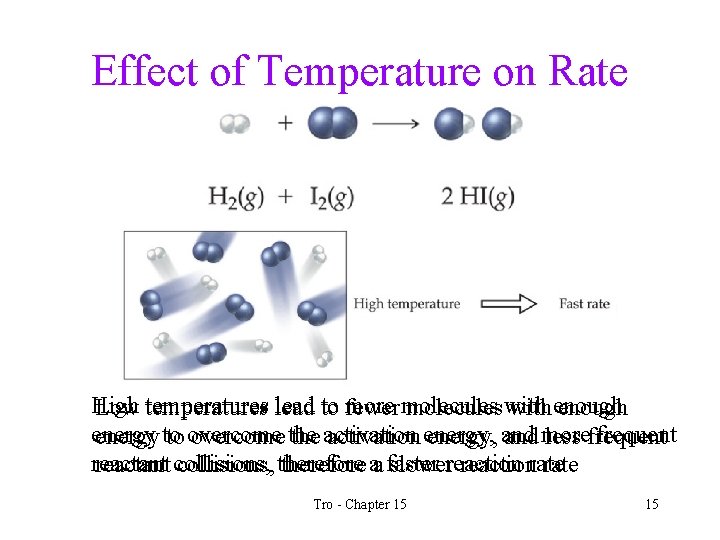
Effect of Temperature on Rate High more molecules Low temperatures lead to fewer molecules with enough energy frequent energy to to overcome the activation energy, and more less frequent reactant collisions, therefore aa faster slowerreactionrate Tro - Chapter 15 15

Reaction Dynamics • If the products of a reaction are removed from the system as they are made, then a chemical reaction will proceed until the limiting reactants are used up. • However, if the products are allowed to accumulate; they will start reacting together to form the original reactants. This is called the reverse reaction. Tro - Chapter 15 16

Reaction Dynamics • The forward reaction slows down as the amounts of reactants decreases. • At the same time the reverse reaction speeds up as the concentration of the products increases. • Eventually the forward reaction is using reactants and making products as fast as the reverse reaction is using products and making reactants. This is called chemical equilibrium. ü dynamic equilibrium is reached when the rates of two opposite processes are the same Tro - Chapter 15 17

Chemical Equilibrium • When a reaction reaches equilibrium, the amounts of reactants and products in the system stay constant • the forward and reverse reactions still continue • because they go at the same rate, the amounts of materials don't change. Tro - Chapter 15 18
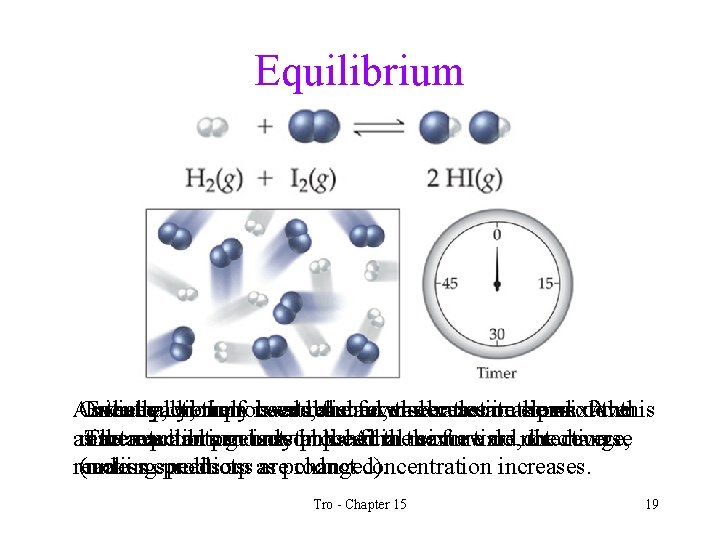
Equilibrium As Once Initially, theequilibrium reaction wethe only proceeds, is have established, reactant the forward molecules the concentrations reaction in the slows mixture. of down thethis Eventually, forward and reverse rates are equal. At as reactants The thereaction reactants and products can getonly proceed inup. the. At final in thethe mixture same forward time, do not direction, thechange, reverse time equilibrium isused established. reaction (unless makingconditions speeds products. up as areproduct changed). concentration increases. Tro - Chapter 15 19
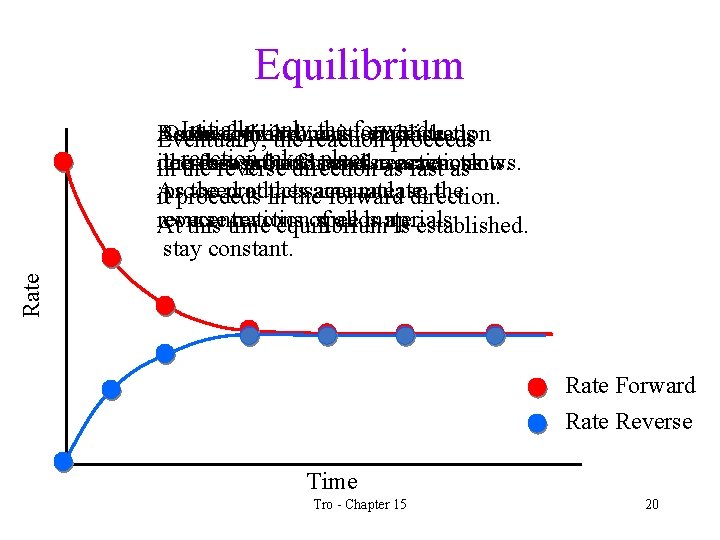
Equilibrium Rate Initially, theisforward Because As Once theequilibrium forward theonly reactant reaction established, concentration proceeds Eventually, the reaction proceeds reaction takes place. it decreases, the makes forward products the and forward reverse and uses reaction reactants. in the reverse direction asreactions fast asslows. As the products at in thethe same accumulate, rate, so the itproceeds forward direction. reverse concentrations reaction of speeds all materials up. At this time equilibrium is established. stay constant. Rate Forward Rate Reverse Time Tro - Chapter 15 20

Equilibrium Equal • the rates of the forward and reverse reactions are equal at equilibrium • but that does not mean the concentrations of reactants and products are equal • some reactions reach equilibrium only after almost all the reactant molecules are consumed – we say the position of equilibrium favors the products • other reactions reach equilibrium when only a small percentage of the reactant molecules are consumed – we say the position of equilibrium favors the reactants Tro - Chapter 15 21

An Analogy: Population Changes When However, Narnians as time feelpasses, overcrowded, emigration some will occur emigrate in to both Middle directions Earth. at the same rate, leading to populations in Narnia and Middle Earth that are constant, though not necessarily equal Tro - Chapter 15 22

Equilibrium Constant • even though the concentrations of reactants and products are not equal at equilibrium, there is a relationship between them • for the general equation a. A + b. B c. C + d. D, the relationship given below is called the equilibrium constant, Keq ü the lower case letters represent the coefficients of the balanced chemical equation ü always products over reactants Tro - Chapter 15 23

Writing Equilibrium Constant Expressions • for a. A + b. B c. C + d. D the equilibrium constant expression is • so for the reaction 2 N 2 O 5 4 NO 2 + O 2 the equilibrium constant expression is: Tro - Chapter 15 24

What Does the Value of Keq Imply? • when the value of Keq >> 1, we know that when the reaction reaches equilibrium there will be many more product molecules present than reactant molecules ü the position of equilibrium favors products • when the value of Keq << 1, we know that when the reaction reaches equilibrium there will be many more reactant molecules present than product molecules ü the position of equilibrium favors reactants Tro - Chapter 15 25
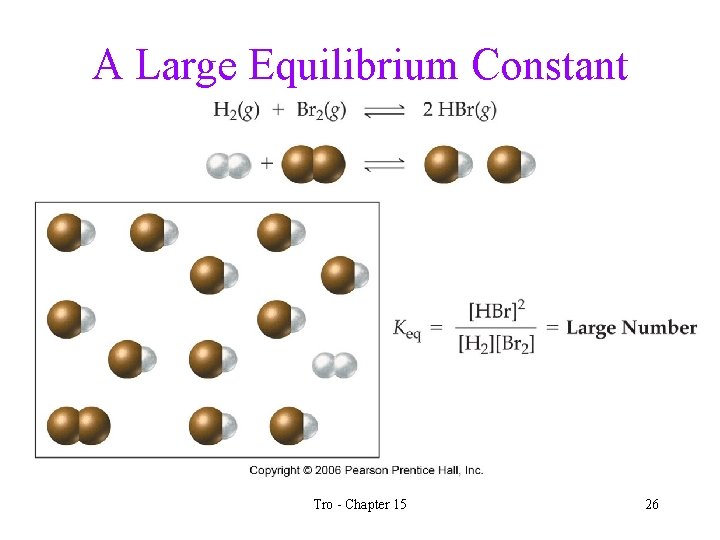
A Large Equilibrium Constant Tro - Chapter 15 26
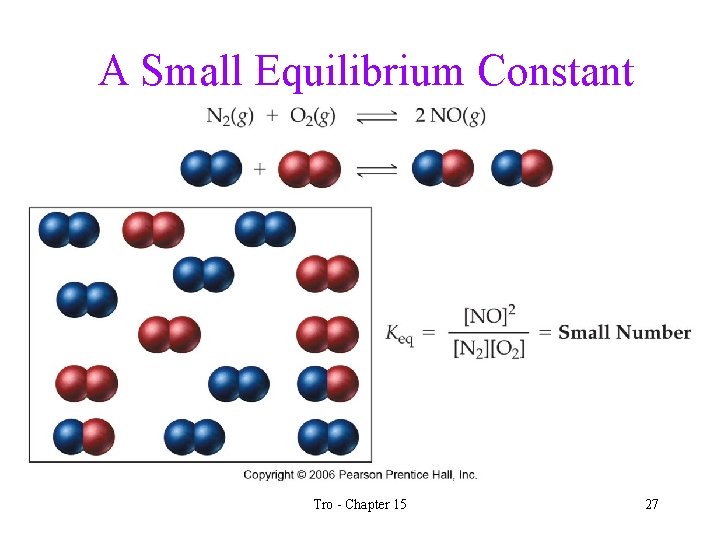
A Small Equilibrium Constant Tro - Chapter 15 27

Equilibrium Constants for Heterogeneous Equilibria • pure substances in the solid and liquid state have constant concentrations ü adding or removing some does not change the concentration because they do not expand to fill the container or spread throughout a solution • therefore these substances are not included in the equilibrium constant expression for the reaction Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + CO 2(g) + H 2 O(l) Tro - Chapter 15 28

Calculating Keq • the value of the equilibrium constant may be determined by measuring the concentrations of all the reactants and products in the mixture after the reaction reaches equilibrium, then substituting in the expression for Keq • even though you may have different amounts of reactants and products in the equilibrium mixture, the value of Keq will always be the same ü the value of Keq depends only on the temperature ü the value of Keq does not depend on the amounts of reactants or products you start with Tro - Chapter 15 29
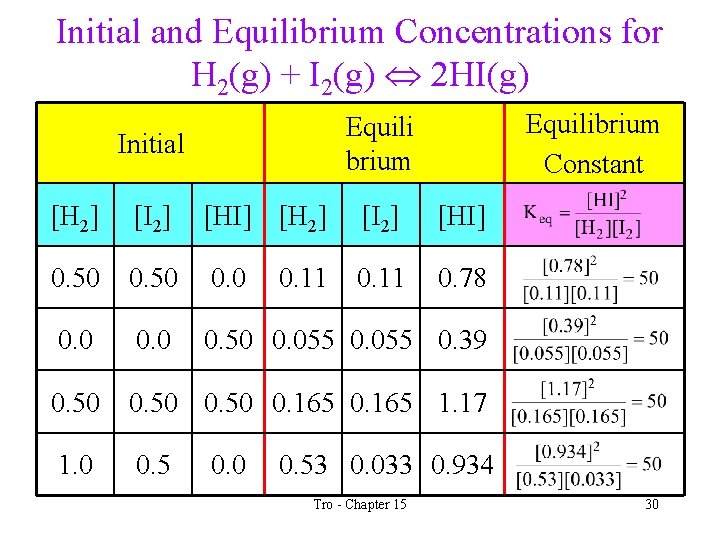
Initial and Equilibrium Concentrations for H 2(g) + I 2(g) 2 HI(g) Equilibrium Constant Equili brium Initial [H 2] [I 2] [HI] 0. 50 0. 11 0. 78 0. 0 0. 50 0. 055 0. 39 0. 50 0. 165 1. 17 1. 0 0. 5 0. 0 0. 11 0. 53 0. 033 0. 934 Tro - Chapter 15 30

Example: • A mixture of CH 4, C 2 H 2 and H 2 is allowed to come to equilibrium at 1700°C. The measured equilibrium concentrations are [CH 4] = 0. 0203 M, [C 2 H 2] = 0. 0451 M, and [H 2] = 0. 112 M. What is the value of the equilibrium constant at this temperature? 2 CH 4(g) C 2 H 2(g) + 3 H 2(g) Tro - Chapter 15 31

Example: • In an equilibrium mixture the concentrations of H 2 and I 2 are both 0. 020 M. What is the value of the equilibrium concentration of HI? H 2(g) + I 2(g) 2 HI(g) Keq = 69 at 340°C Tro - Chapter 15 32

Disturbing and Re-establishing Equilibrium • once a reaction is at equilibrium, the concentrations of all the reactants and products remain the same • however if the conditions are changed, the concentrations of all the chemicals will change until equilibrium is re-established • the new concentrations will be different, but the equilibrium constant will be the same üunless you change the temperature Tro - Chapter 15 33

Le Châtelier’s Principle • Le Châtelier’s Principle guides us in predicting the effect on the position of equilibrium when conditions change • “When a chemical system at equilibrium is disturbed, the system shifts in a direction that will minimize the disturbance” Tro - Chapter 15 34

An Analogy: Population Changes The result will be people moving from Middle Earth into Narnia faster than people moving from Narnia When the an influx populations population ofwill Narnia enters and Middle Earth into Middle Earth. of This continue until a new are from in equilibrium, somewhere outside Narnia, rates it isdisturbs between thethe equilibrium betweenthe theemigration populations established, equilibrium two states established equal between so the populations Narnia andstay Middle constant. Earth. however theare new populations will have different numbers of people than the old ones. Tro - Chapter 15 35

The Effect of Concentration Changes on Equilibrium • Adding a reactant will decrease the amounts of the other reactants and increase the amount of the products until a new position of equilibrium is found üthat has the same Keq • Removing a product will increase the amounts of the other products and decrease the amounts of the reactants. üyou can use to this to drive a reaction to completion! • Remember, adding more of a solid or liquid does not change its concentration – and therefore has no effect on the equilibrium Tro - Chapter 15 36

The Effect of Concentration Changes on Equilibrium When NO 2 is added, some of it combines to make more N 2 O 4 Tro - Chapter 15 37
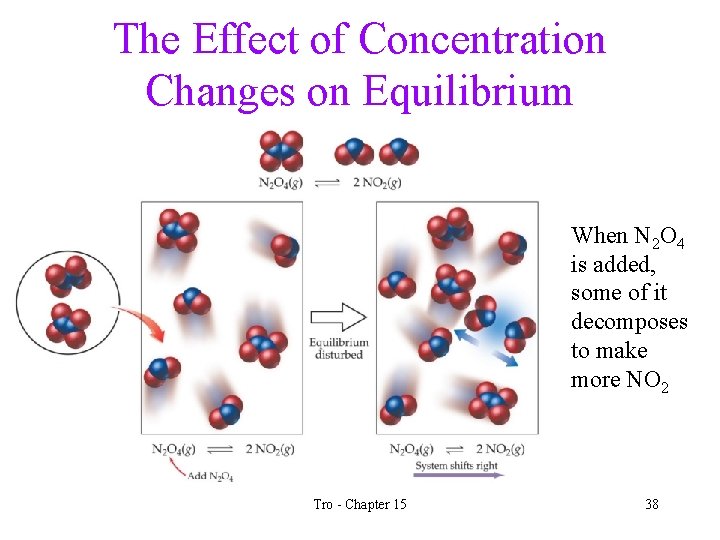
The Effect of Concentration Changes on Equilibrium When N 2 O 4 is added, some of it decomposes to make more NO 2 Tro - Chapter 15 38

Practice - Predict the Effect on the Equilibrium when the Underlined Substance is Added to the Following Systems • 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) Tro - Chapter 15 39

Practice - Predict the Effect on the Equilibrium when the Underlined Substance is Added to the Following Systems • 2 CO 2(g) 2 CO(g) + O 2(g) shift right, removing some of the added CO 2 and increasing the concentrations of CO and O 2 • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) shift left, removing some of the added Ba 2+ and reducing the concentration of SO 42 - • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) shift right, removing some of the added CO 2 and decreasing the O 2, - Chapter 15 40 while increasing. Tro the concentration of CO 2

Effect of Volume Change on Equilibrium • for solids, liquids or solutions, changing the size of the container has no effect on the concentration • changing the volume of a container does change the concentration of a gas üsame number of moles, but different number of liters, resulting in a different molarity Tro - Chapter 15 41

Effect of Volume Change on Equilibrium • decreasing the size of the container increases the concentration of all the gases in the container ü increases their partial pressures • if their partial pressures increase, then the total pressure in the container will increase • according to Le Châtelier’s Principle, the equilibrium should shift to remove that pressure • the way to reduce the pressure is to reduce the number of molecules in the container • when the volume decreases, the equilibrium shifts to the side with fewer molecules Tro - Chapter 15 42

The Effect of Volume Changes on Equilibrium When Since thethere pressure are more is decreased gas by molecules increasing on the volume, reactantsthe side position of theofreaction, equilibrium whenshifts the toward pressure the side is increased with thethe greater position numberofofequilibrium molecules shifts – the toward reactant the products. side. Tro - Chapter 15 43

Practice - Predict the Effect on the Equilibrium when the Volume is Reduced • 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) Tro - Chapter 15 44

Practice - Predict the Effect on the Equilibrium when the Volume is Reduced • 2 CO 2(g) 2 CO(g) + O 2(g) shift left, because there are fewer gas molecules on the reactant side than on the product side • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) no effect since none of the substances are gases • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) shift right, because there are fewer gas molecules Tro - Chapter 15 on the product side than on the reactant side 45

The Effect of Temperature Changes on Equilibrium • exothermic reactions release energy and endothermic reactions absorb energy • if we write Heat as a product in an exothermic reaction or as a reactant in an endothermic reaction, it will help us use Le Châtelier’s Principle to predict the effect of temperature changes üeven though heat is not matter and not written in a proper equation Tro - Chapter 15 46

The Effect of Temperature Changes on Equilibrium for Exothermic Reactions • for an exothermic reaction, heat is a product • increasing the temperature is like adding heat • according to Le Châtelier’s Principle, the equilibrium will shift away from the added heat • the concentrations of C and D will decrease and the concentrations of A and B will increase • the value of Keq will decrease • how will decreasing the temperature effect the system? a. A + b. B c. C + d. D + Heat Tro - Chapter 15 47

The Effect of Temperature Changes on Equilibrium for Endothermic Reactions • for an endothermic reaction, heat is a reactant • increasing the temperature is like adding heat • according to Le Châtelier’s Principle, the equilibrium will shift away from the added heat • the concentrations of C and D will increase and the concentrations of A and B will decrease • the value of Keq will increase • how will decreasing the temperature effect the system? Heat + a. A + b. B c. C + d. D Tro - Chapter 15 48

The Effect of Temperature Changes on Equilibrium Tro - Chapter 15 49

Practice - Predict the Effect on the Equilibrium when the Temperature is Reduced • Heat + 2 CO 2(g) 2 CO(g) + O 2(g) • Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) endothermic • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) Tro - Chapter 15 exothermic 50

Practice - Predict the Effect on the Equilibrium when the Temperature is Reduced • Heat + 2 CO 2(g) 2 CO(g) + O 2(g) shift left, reducing the value of Keq • Heat + Ba. SO 4(s) Ba 2+(aq) + SO 42 -(aq) shift left, reducing the value of Keq • CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(l) + Heat shift right, increasing the value of Keq Tro - Chapter 15 51

Solubility & Solubility Product • even “insoluble” salts dissolve somewhat in water ü insoluble = less than 0. 1 g per 100 g H 2 O • the solubility of insoluble salts is described in terms of equilibrium between undissolved solid and aqueous ions produced An. Ym(s) Û n A+(aq) + m Y-(aq) • equilibrium constant for this process is called the solubility product constant, Ksp = [A+]n[Y-]m • if there is undissolved solid in equilibrium with the solution, the solution is saturated • larger Ksp = more soluble ü for salts that produce the same number of ions Tro - Chapter 15 52

Example - Determine the Ksp of Pb. Br 2 if its solubility is 1. 44 x 10 -2 M Pb. Br 2(s) Û Pb 2+(aq) + 2 Br–(aq) init -0 0 equil -0. 0144 0. 0288 Ksp = [Pb 2+][Br–]2 = (0. 0144)(0. 0288)2 = 1. 19 x 10 -5 Tro - Chapter 15 53

Example: • Calculate the molar solubility of Ba. SO 4. Ksp = 1. 07 x 10 -10 at 25°C Tro - Chapter 15 54

Activation Energy • the energy barrier that prevents any collision between molecules from being an effective collision is called the activation energy • the larger the activation energy of a reaction, the slower it will be üat a given temperature Tro - Chapter 15 55
Relative Potential Energy Exothermic Reaction Activation Energy, large Activation Energy, small Reactants DHreaction Products Progress of Reaction Tro - Chapter 15 56
Relative Potential Energy Endothermic Reaction Activation Energy Products DHreaction Reactants Progress of Reaction Tro - Chapter 15 57

Catalysts • a catalyst is a substance that increases the rate of a reaction, but is not consumed in the reaction • catalysts lower the activation energy of a reaction • catalysts work by providing a easier pathway for the reaction • Link to catalyzed reaction of H 2 S and SO 2 above Tro - Chapter 15 58
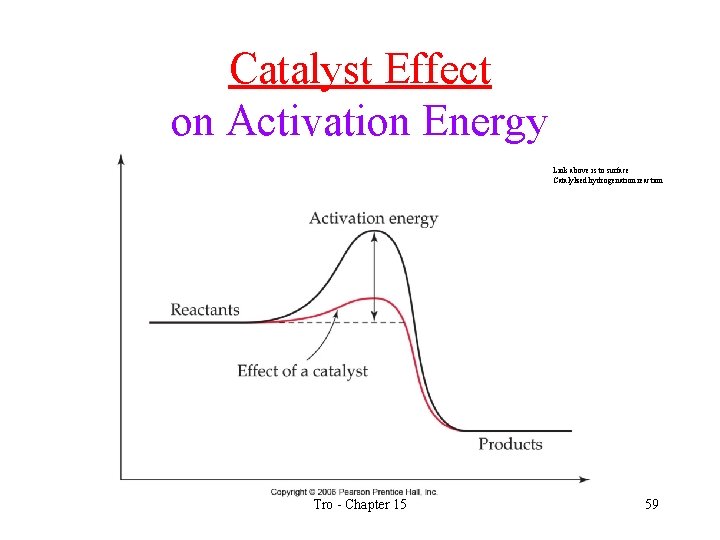
Catalyst Effect on Activation Energy Link above is to surface Catalylzed hydrogenation reaction Tro - Chapter 15 59

Catalyst Effect on Activation Energy Tro - Chapter 15 60

Enzymes • enzymes are protein molecules produced by living organisms that catalyze chemical reactions • the enzyme molecules have an active site that organic molecules bind to • when the organic molecule is bound to the active site, certain bonds are weakened • this allows a particular chemical change to occur easier and quicker ü i. e. the activation energy is lowered Tro - Chapter 15 61

Sucrase Tro - Chapter 15 62
- Slides: 62