Chapter 15 Chemical Equilibrium The Concept of Equilibrium

Chapter 15 Chemical Equilibrium

The Concept of Equilibrium Chemical equilibrium occurs when a reaction and its reverse reaction proceed at the same rate.

The Equilibrium Constant • Consider the generalized reaction a. A + b. B c. C + d. D • The equilibrium expression for this reaction would be [C]c[D]d Kc = [A]a[B]b

Sample Exercise 15. 1 Writing Equilibrium-Constant Expressions Write the equilibrium expression for Kc for the following reactions: (a) (b) (c) Solution: (a) (b) (c)
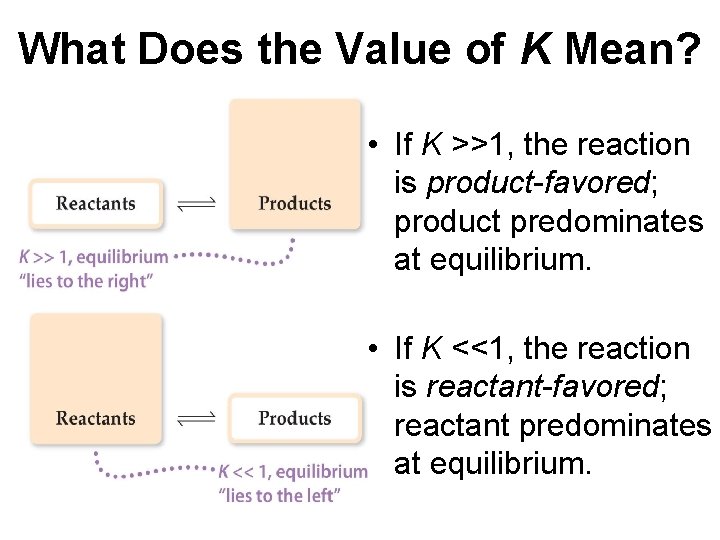
What Does the Value of K Mean? • If K >>1, the reaction is product-favored; product predominates at equilibrium. • If K <<1, the reaction is reactant-favored; reactant predominates at equilibrium.

Manipulating Equilibrium Constants The equilibrium constant of a reaction in the reverse reaction is the reciprocal of the equilibrium constant of the forward reaction: N 2 O 4(g) 2 NO 2(g) N 2 O 4(g) Kc = [NO 2]2 = 0. 212 at 100 C [N 2 O 4] = 4. 72 at 100 C 2 [NO 2]

Sample Exercise 15. 4 Evaluating an Equilibrium Constant When an Equation is Reversed For the reaction that is run at 25 C, Kc = 1 10 30. Use this information to write the equilibriumconstant expression and calculate the equilibrium constant for the reaction Solution:

Heterogeneous Equilibrium The concentrations of solids and liquids are essentially constant, and equals its density (g/L) divided by its molar mass. Therefore, they do not appear in the equilibrium expression.

Sample Exercise 15. 6 Writing Equilibrium-Constant Expressions for Heterogeneous Reactions Write the equilibrium-constant expression Kc for Solution: (a) (b)

Le Châtelier’s Principle “If a system at equilibrium is disturbed by a change in temperature, pressure, or the concentration of one of the components, the system will shift its equilibrium position so as to counteract the effect of the disturbance. ”

Catalysts increase the rate of both the forward and reverse reactions.
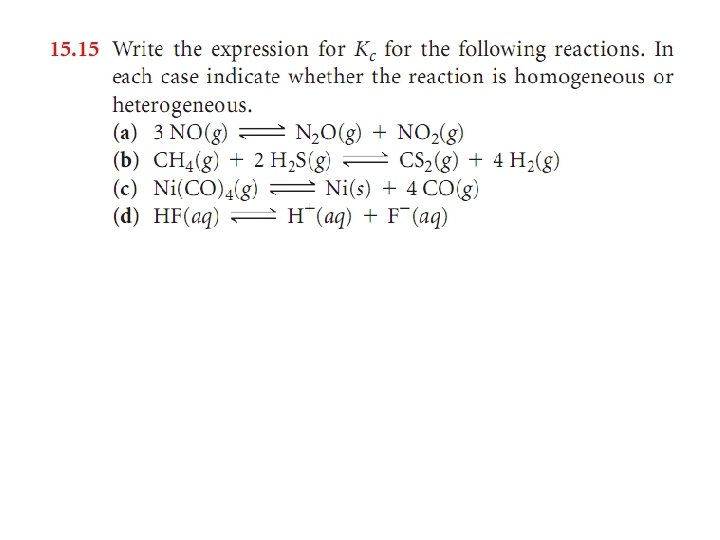

- Slides: 13