Chapter 15 Chemical Equilibrium Prentice Hall 2003 Chapter

Chapter 15 Chemical Equilibrium Prentice Hall © 2003 Chapter 15

The Equilibrium Constant • For a general reaction the equilibrium constant expression for everything in solution is where Keq is the equilibrium constant. Prentice Hall © 2003 Chapter 15

The Equilibrium Constant Gas Phase Reaction • For a general reaction in the gas phase the equilibrium constant expression is where Keq is the equilibrium constant. Prentice Hall © 2003 Chapter 15

The Equilibrium Constant General Notes • Keq is based on the molarities (or partial pressures) of reactants and products at equilibrium. (Note the upper case K!) • We generally omit the units of the equilibrium constant. • The equilibrium constant expression boils down to “products over reactants”. • The same equilibrium is established no matter how the reaction is begun. Prentice Hall © 2003 Chapter 15
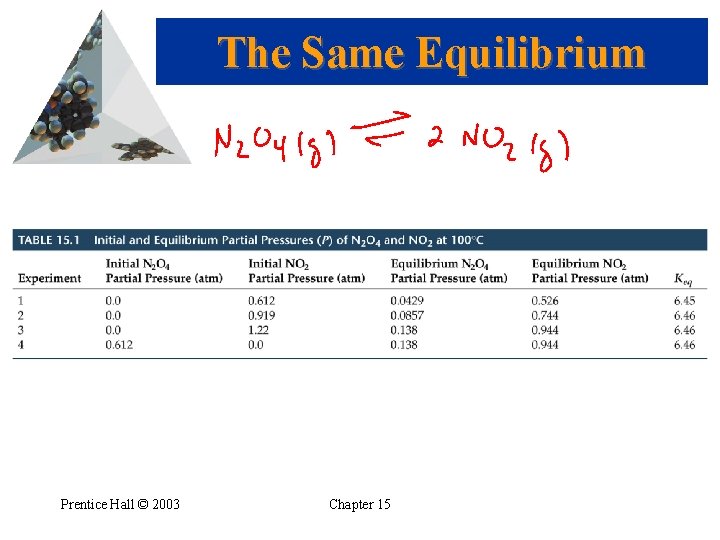
The Same Equilibrium Prentice Hall © 2003 Chapter 15

Significance of K • K expresses the relative amounts of products to reactants. • A larger K means more products are present at equilibrium (and vice versa!) • If K >> 1, then products dominate at equilibrium and equilibrium lies to the right. • If K << 1, then reactants dominate at equilibrium and the equilibrium lies to the left. Prentice Hall © 2003 Chapter 15
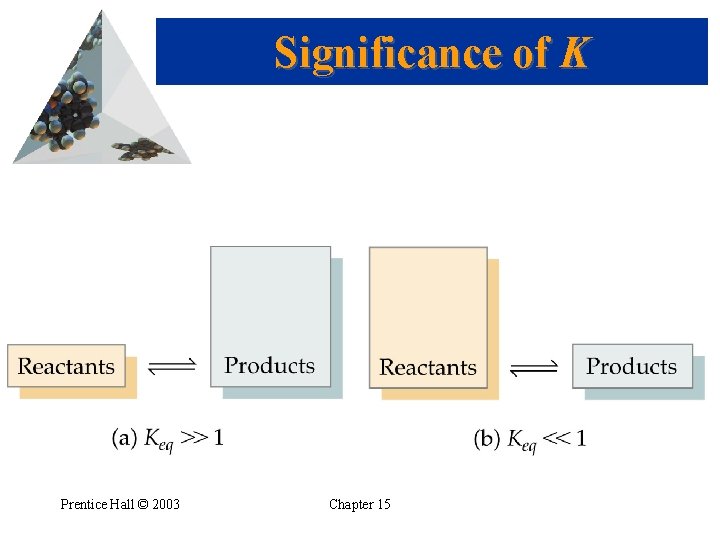
Significance of K Prentice Hall © 2003 Chapter 15

The Equilibrium Constant The Direction of the Chemical Equation • An equilibrium can be approached from any direction. • Example: Prentice Hall © 2003 Chapter 15

The Equilibrium Constant Other Ways to Manipulate Chemical Equations and Keq Values • The equilibrium constant for a reaction which is the sum of other reactions is the product of the equilibrium constants for the individual reactions. 2 NOBr(g) = 2 NO(g) + Br 2(g) Keq = 0. 42 Br 2(g) + Cl 2(g) = 2 Br. Cl Keq = 7. 2 2 NOBr(g) + Cl 2(g) = 2 NO(g) + 2 Br. Cl(g) Keq = Prentice Hall © 2003 Chapter 15

Heterogeneous Equilibria • Experimentally, the amount of CO 2 does not depend on the • amounts of Ca. O and Ca. CO 3. Why? • The concentration of a solid or pure liquid is its density (g/m. L) divided by its molar mass (g/mol). Both the density and the molar mass are constants. • Thus, the concentrations of solids and pure liquids are constant. • We ignore the concentrations of pure liquids and pure solids in equilibrium constant expressions. Prentice Hall © 2003 Chapter 15
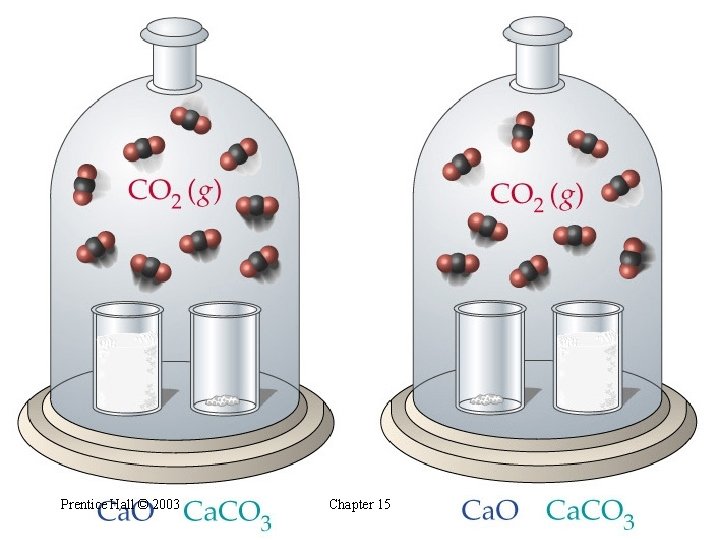
Prentice Hall © 2003 Chapter 15

Calculating Equilibrium Concentrations • Proceed as follows: – Tabulate initial and equilibrium concentrations (or partial pressures). – If an initial and equilibrium concentration is given for a species, calculate the change in concentration. – Use stoichiometry on the change in concentration line only to calculate the changes in concentration of all species. – Deduce the equilibrium concentrations of all species. • Usually, the initial concentration of products is zero. Prentice Hall © 2003 Chapter 15

Predicting the Direction of Reaction • We define Q, the reaction quotient, as follows: • Q = K only at equilibrium. • If Q > K then the reaction must proceed in reverse to reach equilibrium. • If Q < K then the forward reaction must occur to reach equilibrium. Prentice Hall © 2003 Chapter 15
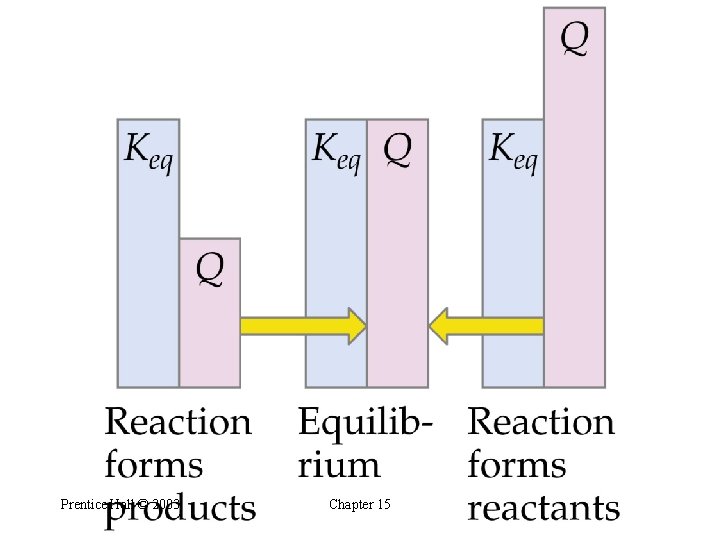
Prentice Hall © 2003 Chapter 15
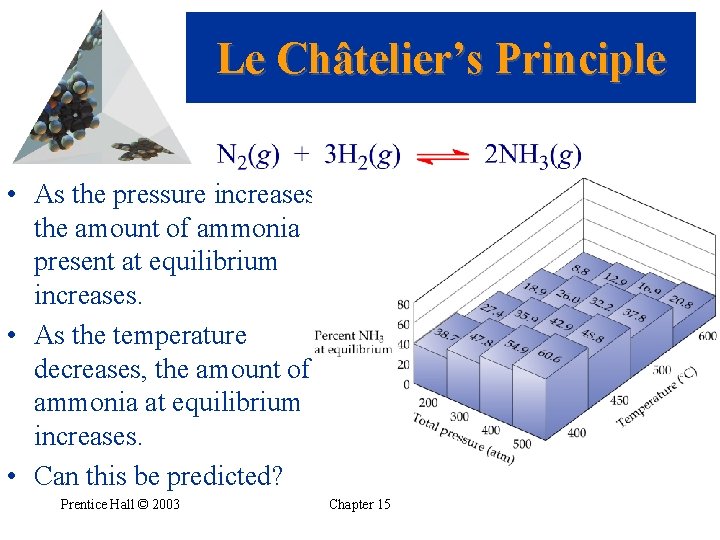
Le Châtelier’s Principle • As the pressure increases, the amount of ammonia present at equilibrium increases. • As the temperature decreases, the amount of ammonia at equilibrium increases. • Can this be predicted? Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle • Le Châtelier’s Principle: • If a system at equilibrium is disturbed, the system will move in such a way as to counteract the disturbance. • An equilibrium reaction adjusts the “position of equilibrium” to relieve any stress that is applied. Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle Reactant and Product Concentrations • If H 2 is added while the system is at equilibrium, the system must respond to counteract the added H 2 • The system reacts to consume the H 2 and produce NH 3 until equilibrium is once again established. • So, [H 2] and [N 2] will decrease and [NH 3] increases. Prentice Hall © 2003 Chapter 15
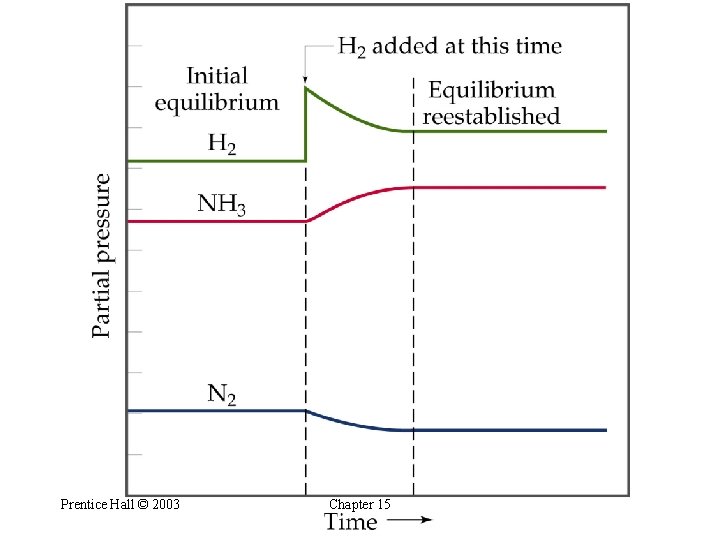
Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle Reactant and Product Concentrations • Adding a reactant or product shifts the equilibrium away from the increase. • Removing a reactant or product shifts the equilibrium towards the decrease. • Usually we desire to maximize the amount of the desired product of a chemical reaction. Two strategies that are often used are (1) use excess of one of the reactants and (2) remove one of the products as it forms. Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle • • Volume and Pressure Changes As volume is decreased pressure increases. If pressure is increased the system will shift to counteract the increase. That is, the equilibrium shifts to remove gases and decrease pressure. An increase in pressure favors the direction that has fewer moles of gas. In a reaction with the same number of product and reactant moles of gas, pressure has no effect. Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle Volume and Pressure Changes • An increase in pressure (by decreasing the volume) causes the system to not be at equilibrium because the concentration of both gases is increased. • The system shifts to reduce the number of moles of gas. • Thus, it shifts to the right (the forward reaction is favored). Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle Effect of Temperature Changes • The equilibrium constant is temperature dependent. • For an endothermic reaction, H > 0 and heat can be considered as a reactant. • For an exothermic reaction, H < 0 and heat can be considered as a product. Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle Effect of Temperature Changes • Adding heat (i. e. heating the vessel) favors away from the increase: – if H > 0, adding heat favors the forward reaction, – if H < 0, adding heat favors the reverse reaction. • Removing heat (i. e. cooling the vessel), favors towards the decrease: – if H > 0, cooling favors the reverse reaction, – if H < 0, cooling favors the forward reaction. Prentice Hall © 2003 Chapter 15

Le Châtelier’s Principle The Effect of Catalysis • A catalyst lowers the activation energy barrier for both the forward and the reverse reaction. • Therefore, a catalyst will decrease the time taken to reach equilibrium. • A catalyst does not effect the composition of the equilibrium mixture. Prentice Hall © 2003 Chapter 15
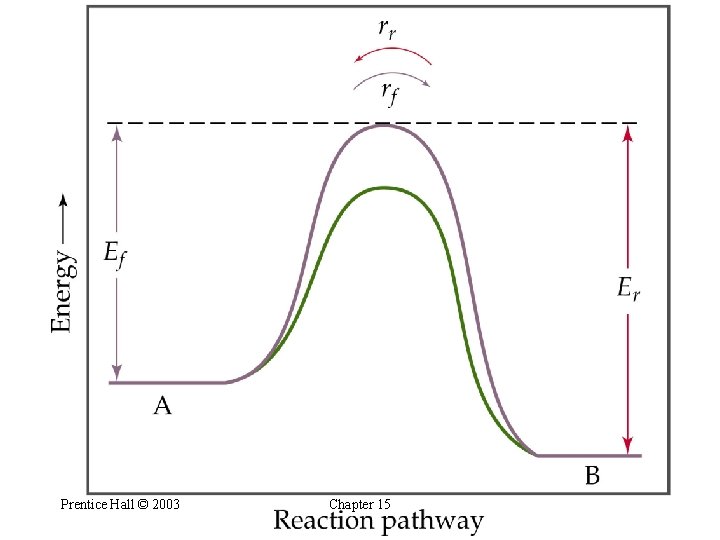
Prentice Hall © 2003 Chapter 15
- Slides: 25