Chapter 15 Buffer A solution that resists a

Chapter 15

Buffer: A solution that resists a change in its p. H when either H+ or OH- ions are added Very important example; blood (cells can only survive in very narrow p. H range, thus constant blood p. H is vital. ) Buffers can contain weak acids & salts or weak bases & salts (solutions can be buffered at almost any p. H) Strong acids / bases cannot be used…

Buffered Solutions Buffers: resist change in p. H when acid or base is added. Buffer Solutions: contain a common ion and are important in biochemical and physiological processes Organisms (and humans) have built-in buffers to protect them against changes in p. H. Blood: (p. H 7. 4) Death = 7. 0 <p. H > 7. 8 = Death Human blood is maintained by a combination of CO 3 -2, PO 4 -3 and protein buffers.
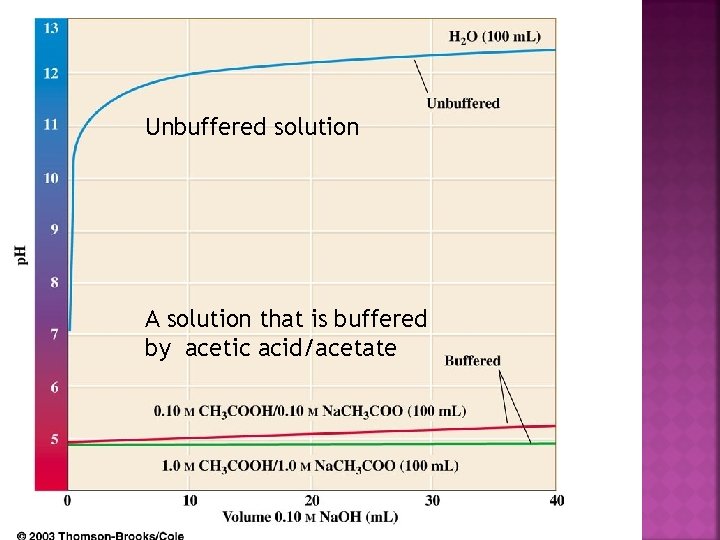
Unbuffered solution A solution that is buffered by acetic acid/acetate
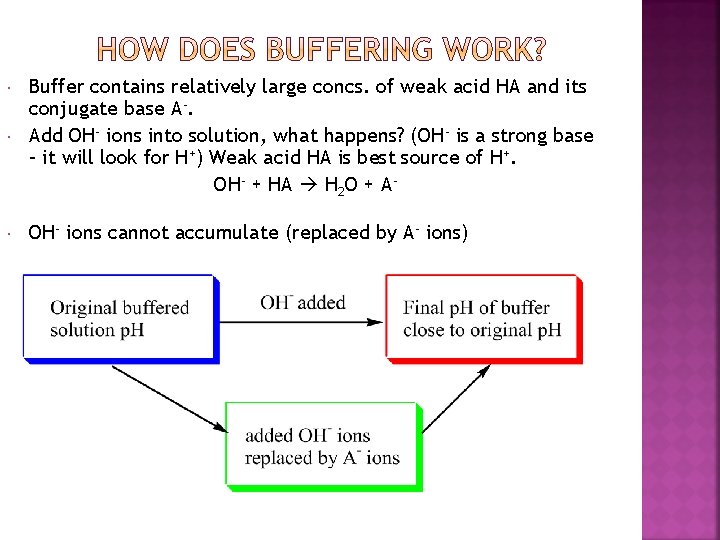
Buffer contains relatively large concs. of weak acid HA and its conjugate base A-. Add OH- ions into solution, what happens? (OH- is a strong base – it will look for H+) Weak acid HA is best source of H+. OH- + HA H 2 O + A- OH- ions cannot accumulate (replaced by A- ions)

If OH- ions converted to A- ions, how does the p. H stay so stable? Look at eq. expression for [H+]; dissociation equilibrium for HA Eq. [H+] (and thus p. H) determined by ratio [HA] / [A-]. If [HA] and [A-] are large relative to [OH-] added, change in [HA] / [A-] will be very small. Thus, [H+] and p. H changes will also be very small. The essence of buffering: [HA] and [A-] are very large relative to [OH-] added.

How Do Buffers Work? HA H+ + A – HA = generic acid • Ka = [H+][A-]/[HA] => [H+] = Ka[HA]/[A-] • If Ka is small (weak acid) then [H+] does not change much when [HA] and [A-] change. If [HA] and [A-] are large, and [HA]/[A-] ≈ 1, then small additions of acid ([H+]) or base ([OH-]) don’t change the ratio much.

Which of the following are buffer systems? (a) KF/HF (b) KBr/HBr (c) Na 2 CO 3/Na. HCO 3 17. 3


Same calculations as before using ICE charts. Calculate the p. H of a solution that is. 50 M HAc and. 25 M Na. Ac (Ka = 1. 8 x 10 -5)

Na+ is a spectator and the reaction we are worried about is HAc H+ + Ac. Initial Final l 0. 50 M 0 -x 0. 50 -x Choose x to be small We can fill in the table x x 0. 25 M x 0. 25+x

Initial Final HAc l 0 0. 50 M Ac 0. 25 M -x x x 0. 50 -x x 0. 25+x Do the math Ka = 1. 8 x 10 -5 = l H+ + x (0. 25+x) (0. 50 -x) Assumption is valid p. H = -log (3. 6 x 10 -5) = 4. 44 = x (0. 25) x = 3. 6 x 10 -5 (0. 50)

Do the stoichiometry first. Use moles not molar A strong base will grab protons from the weak acid reducing [HA]0 A strong acid will add its proton to the anion of the salt reducing [A-]0 Then do the equilibrium problem. What is the p. H of 1. 0 L of the previous solution when 0. 010 mol of solid Na. OH is added?

HAc l l l H+ + Ac- 0. 50 mol 0. 25 mol 0. 49 M 0. 26 M In the initial mixture M x L = mol 0. 50 M HAc x 1. 0 L = 0. 50 mol HAc 0. 25 M Ac- x 1. 0 L = 0. 25 mol Ac. Adding 0. 010 mol OH- will reduce the HAc and increase the Ac- by 0. 010 mole Because it is in 1. 0 L, we can convert it to molarity

HAc l H+ + Ac- 0. 50 mol 0. 25 mol 0. 49 M 0. 26 M Fill in the table Initial Final HAc 0. 49 M H+ + 0 Ac 0. 26 M -x x x 0. 49 -x x 0. 26+x

Initial Final HAc 0 0. 49 M Ac 0. 26 M -x x x 0. 49 -x x 0. 26+x Do the math Ka = 1. 8 x 10 -5 = l H+ + x (0. 26+x) = x (0. 26) x = 3. 4 x 10 -5 (0. 49 -x) (0. 49) p. H = -log (3. 4 x 10 -5) = 4. 47

If we had added 0. 010 mol of Na. OH to 1 L of water, the p. H would have been. 0. 010 M OHp. OH = 2 p. H = 12 But with a mixture of an acid and its conjugate base the p. H doesn’t change much Called a buffer.

A. Weak base problem: HONH 2 + Initial 0. 100 M Change -x Equil. 0. 100 - x Kb = 1. 1 × 10 -8 = H 2 O ⇌ HONH 3+ + OH → 0 +x x Kb = 1. 1 × 10 -8 ~0 +x x ≈ x = [OH ] = 3. 3 × 10 -5 M; p. OH = 4. 48; p. H = 9. 52

Weak acid problem (Cl has no acidic/basic properties); HONH 3+ ⇌ HONH 2 + Initial 0. 100 M 0 Change -x → +x Equil. 0. 100 – x x Ka = = 9. 1 × 10 -7 = ≈ x = [H+] = 3. 0 × 10 -4 M; p. H = 3. 52 H+ ~0 +x x
![Ka = [H+] [A-] [HA] so [H+] = Ka [HA] [A-] The [H+] Ka = [H+] [A-] [HA] so [H+] = Ka [HA] [A-] The [H+]](http://slidetodoc.com/presentation_image_h2/fa2703fe56067515f1be7ef91470c290/image-21.jpg)
Ka = [H+] [A-] [HA] so [H+] = Ka [HA] [A-] The [H+] depends on the ratio [HA]/[A-] taking the negative log of both sides p. H = -log(Ka [HA]/[A-]) p. H = -log(Ka)-log([HA]/[A-]) p. H = p. Ka + log([A-]/[HA])
![p. H = p. Ka + log([A-]/[HA]) p. H = p. Ka + p. H = p. Ka + log([A-]/[HA]) p. H = p. Ka +](http://slidetodoc.com/presentation_image_h2/fa2703fe56067515f1be7ef91470c290/image-22.jpg)
p. H = p. Ka + log([A-]/[HA]) p. H = p. Ka + log(base/acid) Works for an acid and its salt Like HNO 2 and Na. NO 2 Or a base and its salt Like NH 3 and NH 4 Cl But remember to change Kb to Ka

Calculate the p. H of the following 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) lp. H = 3. 38

Calculate the p. H of the following 0. 25 M NH 3 and 0. 40 M NH 4 Cl (Kb = 1. 8 x 10 -5) Ka = 1 x 10 -14 1. 8 x 10 -5 Ka = 5. 6 x 10 -10 remember its the ratio base over acid lp. H = 9. 05

d. Buffer solution where p. Ka = -log (9. 1 × 10 -7) = 6. 04. Using the Henderson-Hasselbalch equation: p. H = p. Ka + log = 6. 04

What would the p. H be if. 020 mol of HCl is added to 1. 0 L of both of the preceding solutions. What would the p. H be if 0. 050 mol of solid Na. OH is added to 1. 0 L of each of the proceeding. Remember adding acids increases the acid side, Adding base increases the base side.

What would the p. H be if. 020 mol of HCl is added to 1. 0 L of preceding solutions. 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) Initially After acid HC 3 H 5 O 3 H+ + C 3 H 5 O 3 - 0. 75 mol 0. 77 mol 0 Compared to 3. 38 before acid was added 0. 25 mol 0. 23 mol

What would the p. H be if 0. 050 mol of solid Na. OH is added to 1. 0 L of the solutions. 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) Initially After acid HC 3 H 5 O 3 H+ + C 3 H 5 O 3 - 0. 75 mol 0. 70 mol 0 Compared to 3. 38 before acid was added 0. 25 mol 0. 30 mol

A. Added H+ reacts completely with HONH 2 (the best base present) to form HONH 3+. HONH 2 + H+ → HONH 3+ Before 0. 100 M 0. 020 M 0 Change -0. 020 +0. 020 After 0. 080 0 0. 020 After this reaction, a buffer solution exists, i. e. , a weak acid (HONH 3+) and its conjugate base (HONH 2) are present at the same time. Using the Henderson. Hasselbalch equation to solve for the p. H where: p. Ka = -log (Kw /Kb) = 6. 04: p. H = 6. 04 + log = 6. 04 + 0. 60 = 6. 64

b. We have a weak acid and a strong acid present at the same time. The H+ contribution from the weak acid, HONH 3+, will be negligible. So, we have to consider only the H+ from HCl. [H+] = 0. 020 M; p. H = 1. 70 c. This is a strong acid in water. [H+] = 0. 020 M; p. H = 1. 70

D. H+ will react completely with HONH 2, the best base present. HONH 2 + H+ → HONH 3+ Before 0. 100 M 0. 020 M 0. 100 M Change -0. 020 +0. 020 After 0. 080 0 0. 120 A buffer solution results after reaction. Using the Henderson-Hasselbalch equation: p. H = 6. 04 + log = 6. 04 - 0. 18 = 5. 86

a. We have a weak base and a strong base present at the same time. The OH contribution from the weak base, HONH 2, will be negligible. Consider only the added strong base as the primary source of OH. [OH ] = 0. 020 M; p. OH = 1. 70; p. H = 12. 30

b. Added strong base will react to completion with the best acid present, HONH 3+. OH + HONH 3+ → HONH 2 + H 2 O Before 0. 020 M 0. 100 M 0 Change -0. 020 → +0. 020 After 0 0. 080 0. 020 The resulting solution is a buffer (a weak acid and its conjugate base). Using the Henderson. Hasselbalch equation: p. H = 6. 04 + log = 6. 04 - 0. 60 = 5. 44
![c. This is a strong base in water. [OH-] = 0. 020 M; p. c. This is a strong base in water. [OH-] = 0. 020 M; p.](http://slidetodoc.com/presentation_image_h2/fa2703fe56067515f1be7ef91470c290/image-34.jpg)
c. This is a strong base in water. [OH-] = 0. 020 M; p. OH = 1. 70; p. H = 12. 30 D. Again, the added strong base reacts completely with the best acid present, HONH 3+ + OH → HONH 2 + H 2 O Before 0. 100 M 0. 020 M 0. 100 M Change -0. 020 → +0. 020 After 0. 080 0 0. 120 A buffer solution results. Using the Henderson. Hasselbalch equation: p. H = 6. 04 + log = 6. 04 + 0. 18 = 6. 22

BUFFER CAPACITY Buffer capacity is the amount of protons or hydroxide ions that can be absorbed without a significant change in p. H is determined by the ratio of [A–]/[HA] and p. Ka Capacity is determined by the magnitudes of [HA] and [A–].

Amount of H+/OH- a buffer can absorb without a considerable change in p. H Consider two 1. 0 L ammonia/ammonium buffer solutions: Buffer 1: 1. 0 M NH 3, and 1. 0 M NH 4+ Ka = [NH 3][H+] / [NH 4+] [H+] = Ka. [NH 4+] / [NH 3] = 5. 6 x 10 -10 M; p. H = 9. 2 What happens to the p. H if 0. 1 moles of Na. OH is added? After the reaction with Na. OH, calculate the [H+] and thus the p. H…. p. H=9. 33

Buffer 2: 0. 01 M NH 3, and 0. 01 M NH 4+ Ka = [NH 3][H+] / [NH 4+] [H+] = Ka. [NH 4+] / [NH 3] = 5. 6 x 10 -10 M; p. H = 9. 2 What happens to the p. H if 0. 009 moles of Na. OH is added? After the reaction with Na. OH, calculate the [H+] and thus the p. H….

So - what general statement can be made about buffer capacity with respect to the initial concentration of the weak acid and its conjugate base in the buffer? High concentrations: resist change to p. H, good buffers Low concentrations: weak buffers, change easily
![Large changes in the ratio [A]/[HA] will produce large changes in p. H. Large changes in the ratio [A]/[HA] will produce large changes in p. H.](http://slidetodoc.com/presentation_image_h2/fa2703fe56067515f1be7ef91470c290/image-39.jpg)
Large changes in the ratio [A]/[HA] will produce large changes in p. H. Optimal Buffering occurs when [HA] is equal to [A]. Therefore we want the ratio of [HA]/[A] to equal 1 According to Henderson-Hasselbach p. H = p. Ka + log([A-]/[HA]) or p. H = p. Ka + log(1) So, the p. Ka of the weak acid to be used as the buffer should be as close as possible to the desired p. H for an optimal buffer.

Buffer Summary Buffer Design: Add known amount of HA (weak acid) and salt of HA (its conjugate base, A─) [H+] or p. H depends on Ka and the ratio of acid to salt or [A─]. Thus if both conc. HA and A- are large then small additions of acid or base don’t change the ratio much 40

How to Actually Make a Buffer (a buffered solution in a lab) HA H+ + A – • Assume you want to make a 1 L of of 1 M buffer at p. H 4. 74. Use acetic acid, because the p. Ka of acetic acid is 4. 74. • To make the Buffer: • The easy way (this is how we actually do it in the lab): Weigh out the appropriate amount of Na+Acetate- (70 g). Add it to a volumetric flask. Add water to around 0. 9 Liters. Add a stir bar and stir. Slowly add HCl, while monitoring the p. H with a p. H meter. When the p. H reaches the p. Ka (4. 74), stop. Take out the stir bar and add enough water to give 1 L. • The hard way. Start with two solutions. Solution A is 1 M acetic acid. Solution B is 1 M Na+Acetate-. Add solution A to solution B. Then check the p. H with a p. H meter. Adjust the p. H with HCl or Na. OH as required, to get the p. H to 4. 74 But this will change the concentration of A- and HA.


Millimole (mmol) = 1/1000 mol Molarity = mmol/m. L = mol/L Adding a solution of known concentration until the substance being tested is consumed. This is called the equivalence point. Graph of p. H vs. m. L is a titration curve. Completion is signaled by a change in some physical property, such as the color of the reacting mixture or the color of an indicator that has been added to it. [Na. OH] Indicator phenolphthalein “X” Zumdahl Chapter 8

Titrations and p. H Curves 44


Methyl Red Bromothymol blue Phenolphtalein HIn(aq) + H O(l) ↔ H O+(aq) + In-(aq) Zumdahl Chapter 8 10/30/2021 46

Weak acids that change color when they become bases. weak acid written HIn Weak base HIn H+ + Inclear red Equilibrium is controlled by p. H End point - when the indicator changes color. Try to match the equivalence point with the pka and p. H
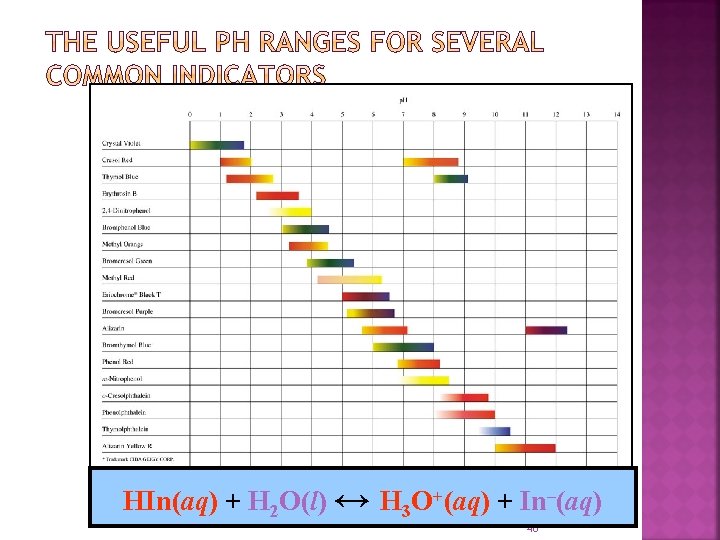
HIn(aq) + H 2 O(l) ↔ H 3 O+(aq) + In–(aq) 48

Equivalence point determined defined by the Stoichiometry, not by the p. Ka. nbase = nacid number of moles of acid = number of moles of base Zumdahl Chapter 8 10/30/2021 49

Indicator Selection • Want indicator color change and titration equivalence point to be as close as possible • Easier with a large p. H change at the equivalence point Strong acid Could use either indicator Weak acid Methyl red changes color to early 50

Strong acid with strong Base Equivalence at p. H 7 m. L of Base added

Weak acid with strong Base l Equivalence at p. H >7 l When the acid is neutralized it makes a weak base l >7 p. H 7 m. L of Base added

Strong base with strong acid l Equivalence at p. H 7 l p. H 7 m. L of acid added

Weak base with strong acid l Equivalence at p. H <7 l When the base is neutralized it makes a weak acid l 7 p. H <7 m. L of acid added

Do the stoichiometry. m. L x M = mmol There is no equilibrium. They both dissociate completely. The reaction is H+ + OH- HOH Use [H+] or [OH-] to figure p. H or p. OH Ex. The titration of 50. 0 m. L of 0. 200 M HNO 3 with 10 ml of 0. 100 M Na. OH H+=. 05 x. 2=. 01 mols OH=. 001 x. 1=. 0001 mol Since H + OH H 2 O all OH are consumed leaving. 009 mol H+/. 051 L=. 15 M p. H=. 82

There is an equilibrium. Ex. Titrate 50. 0 m. L of 0. 10 M HC 2 H 3 O 2 (Ka = 1. 8 x 10 -5) with 10 ml of 0. 10 M Na. OH Do stoichiometry. Use moles OH- + HC 2 H 3 O 2 +H 2 O . 05 L x. 1 M HF=. 005 mol. 01 L x. 1 M Na. OH=. 001 mol So OH- is limiting and is all consumed at equilibrium and. 001 mols of C 2 H 3 O 2 are made. OH- + HC 2 H 3 O 2 C 2 H 3 O 2 + 0 mol/. 06 L . 005 -. 001/. 06 L H 2 O

Then do equilibrium. HC 2 H 3 O 2 H+ + C 2 H 3 O 2[C 2 H 3 O 2][H+]/[HC 2 H 3 O 2]= Ka = 1. 8 x 10 -5 So, I C E HC 2 H 3 O 2 . 06 M -x. 06 -x C 2 H 3 O 2 . 016 M +x. 016+x 1. 8 x 10 -5 =. 016(x)/. 06 p. H= -log 7. 2 x 10 -5= 4. 14 H+ 0 x x x= 7. 2 x 10 -5

This is a strong base, Ba(OH)2, titrated by a strong acid, HCl. The added strong acid will neutralize the OH from the strong base. As is always the case when a strong acid and/or strong base reacts, the reaction is assumed to go to completion. A. Only a strong base is present, but it breaks up into two moles of OH ions for every mole of Ba(OH)2. [OH ] = 2 × 0. 100 M = 0. 200 M; p. OH = 0. 699; p. H = 13. 301 B. mmol OH present = 80. 0 m. L × = 16. 0 mmol OH mmol H+ added = 20. 0 m. L × = 8. 00 mmol H+ Before Change After [OH ]excess OH + H+ → H 2 O 16. 0 mmol 8. 00 mmol -8. 00 mmol 8. 0 mmol 0 = = 0. 080 M; p. OH = 1. 10; p. H =12. 90

c. mmol H+ added = 30. 0 m. L × 0. 400 M = 12. 0 mmol H+ OH + H+ → H 2 O Before 16. 0 mmol 12. 0 mmol After 4. 0 mmol 0 [OH ]excess = = 0. 036 M; p. OH = 1. 44; p. H = 12. 56

d. mmol H+ added = 40. 0 m. L × 0. 400 M = 16. 0 mmol H+; This is the equivalence point. Because the H+ will exactly neutralize the OH from the strong base, all we have in solution is Ba 2+, Cl and H 2 O. All are neutral species, so p. H = 7. 00. e. mmol H+ added = 80. 0 m. L × 0. 400 M = 32. 0 mmol H+ OH + → H 2 O Before 16. 0 mmol 32. 0 mmol After 0 16. 0 mmol [H+]excess = = 0. 100 M; p. H = 1. 000

This is a weak base (H 2 NNH 2) titrated by a strong acid (HNO 3). To calculate the p. H at the various points, let the strong acid react completely with the weak base present, then see what is in solution. A. Only a weak base is present. Solve the weak base equilibrium problem. H 2 NNH 2 + H 2 O ⇌ H 2 NNH 3+ + OH Initial 0. 100 M 0 ~0 Equil. 0. 100 – x x x Kb = 3. 0 × 10 -6 ≈ x = [OH ] = 5. 5 × 10 -4 M p. OH = 3. 26; p. H = 10. 74;

B. mmol H 2 NNH 2 = 100. 0 m. L × = 10. 0 mmol H 2 NNH 2 mmol H+ added = 20. 0 m. L × = 4. 00 mmol H+ H 2 NNH 2 + H+ → H 2 NNH 3+ Before 10. 0 mmol 4. 00 mmol 0 Change -4. 00 mmol → +4. 00 mmol After 6. 0 mmol 0 4. 00 mmol A buffered solution results after the titration reaction. Solving using the Henderson-Hasselbalch equation: p. H = p. Ka + log , Ka = = 3. 3 × 10 -9 p. H = -log (3. 3 × 10 -9 ) + log where VT = total volume, which cancels p. H = 8. 48 + log (1. 5) = 8. 48 + 0. 18 = 8. 66

mmol H+ = 25. 0 m. L × 0. 200 M = 5. 00 mmol H+ H 2 NNH 2 + H+ → H 2 NNH 3+ Before 10. 0 mmol 5. 00 mmol 0 After 5. 0 mmol 0 5. 00 mmol This is the halfway point to equivalence where [H 2 NNH 3+] = [H 2 NNH 2]. At this point, p. H = p. Ka (which is characteristic of the halfway point for any weak base/strong acid titration). p. H = -log (3. 3 × 10 -9 ) = 8. 48 c.

d. mmol H+ added = 40. 0 m. L × 0. 200 M = 8. 00 mmol H+ H 2 NNH 2 + H+ → H 2 NNH 3+ Before 10. 0 mmol 8. 00 mmol 0 After 2. 0 mmol 0 8. 00 mmol A buffer solution results. p. H = 8. 48 + log = 8. 48 + (-0. 60) = 7. 88

e. This is the equivalence point where just enough H+ has been added to react with all of the weak base present. mmol H+ added = 50. 0 m. L × 0. 200 M = 10. 0 mmol H+. H 2 NNH 2 + H+ Before 10. 0 mmol After 0 0 → H 2 NNH 3+ 0 10. 0 mmol As is always the case in a weak base/strong acid titration, the p. H at the equivalence point is acidic because only a weak acid (H 2 NNH 3+) is present (conjugate acids of all weak bases are weak acids themselves). Solving the weak acid equilibrium problem: H 2 NNH 3+ ⇌ H+ + H 2 NNH 2 Initial 10. 0 mmol/150. 0 m. L 0 0 Equil. 0. 0667 - x x x Ka = 3. 3 × 10 -9 ≈ x = [H+] = 1. 5 × 10 -5 M p. H = 4. 82

f. mmol H+ added = 100. 0 m. L × 0. 200 M = 20. 0 mmol H+ H 2 NNH 2 + H+ → Before 10. 0 mmol 20. 0 mmol After 0 10. 0 mmol H 2 NNH 3+ 0 10. 0 mmol Two acids are present past the equivalence point, but the excess H+ will determine the p. H of the solution since H 2 NNH 3+ is a weak acid. Whenever two acids are present, the stronger acid usually determines the p. H. [H+]excess = p. H = 1. 301 = 0. 0500 M;

Will it all dissolve, and if not, how much?

SOLUBILITY EQUILIBRIA Many natural processes depend on the precipitation or dissolving of a slightly soluble ionic substance. Applies to the equilibria of slightly soluble, or nearly insoluble, ionic compounds. Their equilibrium constants can be used to answer questions regarding solubility and precipitation.


Source: ©E. R. Degginger

In general, the solubility product constant is the equilibrium constant for the solubility equilibrium of a slightly soluble (or nearly insoluble) ionic compound. It equals the product of the equilibrium concentrations of the ions in the compound. Each concentration is raised to a power equal to the number of such ions in the formula of the compound.

SOLUBILITY EQUILIBRIA When an excess of a slightly soluble ionic compound is mixed with water, an equilibrium is established between the solid and the ions in the saturated solution. For the salt calcium oxalate, Ca. C 2 O 4, you have the following equilibrium. Ca. C 2 O 4(s) Ca 2+(aq) + C 2 O 42 -(aq) The equilibrium constant for the process is termed the solubility product constant Ksp = [Ca 2+][C 2 O 42 -]

SOLUBILITY EQUILIBRIA Many ionic solids form saturated solutions with very small amounts of solid added to water (a few micrograms) The extent of solubility in water is defined by a solubility product constant (Ksp) For an ionic solid with the general formula Mm. Xx(s) and ionic charges n and y, respectively: Mm. Xx(s) m. Mn+(aq) + x. Xy-(aq) Ksp = [Mn+]m[Xy-]x For the ionic solid Ag. Cl(s) Ag+(aq) + Cl-(aq) Ksp = [Ag+][Cl-] = 1. 8 x 10 -10 (at 25 o. C)

SOLUBILITY EQUILIBRIA Ksp expression is exactly the format used for all other equilibria previously discussed; K is now applied to a specific type of process: solubility of ionic substances The value of Ksp is measured by experiment from the concentrations of ions in solution. For example, for the solid Pb. Br 2 at 25 o. C, the equilibrium concentrations of lead (2+) and bromide (-1) ions formed from a saturated solution of lead bromide Pb. Br 2(s) Pb 2+(aq) + 2 Br-(aq) are found to be: [Pb 2+] = 0. 0120 M & [Br-] = 0. 0240 M Ksp = [Pb 2+][Br-]2 = (0. 0120)(0. 0240)2 = 6. 9 x 10 -6

SOLUBILITY EQUILIBRIA The solubility of ionic substances can be determined from Ksp values Q. Find the solubility of silver sulfate given a Ksp for silver sulfate of 1. 2 x 10 -5 at 25 o. C. Write out equilibrium & set up equilibrium table Ag 2 SO 4(s) 2 Ag+(aq) + SO 42 -(aq) x 2 x x let x = solubility of silver sulfate Ksp = [Ag+]2[SO 42 -] = (2 x)2 x = 4 x 3

Write the solubility product expressions for the following ionic solids: (a) Ba. Cr. O 4; (b) Ni. S; (c) Pb 3(As. O 4)2 a. Ksp = [Ba 2+][Cr. O 42 -] b. Ksp = [Ni 2+][S 2 -] c. Ksp = [Pb 2+]3[As. O 43 -]2

SOLUBILITY RELATIONS General relations between solubility and Ksp are given below (M = metal; X = anion or polyatomic ion): General formulas given as Mm. Xx General Formula MX (e. g. , Ag. Cl) MX 2, M 2 X (e. g. , Pb. I 2) MX 3, M 3 X (e. g. , Ag 3 PO 4) M 2 X 3, M 3 X 2 (e. g. , Fe. Cl 3) Solubility, s s = (Ksp)1/2 s = [Ksp/4]1/3 s = [Ksp/27]1/4 s = [Ksp/108]1/5 The exponent of each s expression equals 1/(m + x)

A 1. 0 -L sample of a saturated calcium oxalate solution, Ca. C 2 O 4, contains 0. 0061 g of the salt at 25 o. C. Calculate the Ksp for this salt at 25 o. C. We must first convert the solubility of calcium oxalate from 0. 0061 g/liter to moles per liter.

A 1. 0 -L sample of a saturated calcium oxalate solution, Ca. C 2 O 4, contains 0. 0061 g of the salt at 25 o. C. Calculate the Ksp for this salt at 25 o. C. When 4. 8 x 10 -5 mol of solid dissolve it forms 4. 8 x 10 -5 mol of each ion. H 2 O Starting 0 0 Change +4. 8 x 10 -5 Equilibrium 4. 8 x 10 -5

A 1. 0 -L sample of a saturated calcium oxalate solution, Ca. C 2 O 4, contains 0. 0061 g of the salt at 25 o. C. Calculate the Ksp for this salt at 25 o. C. You can now substitute into the equilibrium-constant expression.

A. Pb 3(PO 4)2(s) ⇌ 3 Pb 2+(aq) + 2 PO 43 (aq) Initial 0 0 Change -x → +3 x +2 x Equil. 3 x 2 x Ksp = [Pb 2+]3 [PO 43 ]2 = (3 x)3(2 x)2 = 108 x 5, Ksp = 108(6. 2 × 10 -12)5 = 9. 9 × 10 -55 B. Li 2 CO 3(s) ⇌ 2 Li+(aq) Initial 0 Equil. 2 s Ksp = [Li+]2 [CO 32 ] = (2 s)2(s) = 4 s 3, Ksp = 4(7. 4 × 10 -2)3 = 1. 6 × 10 -3 + CO 32 -(aq) 0 s

Ksp will only allow us to compare the solubility of solids that fall apart into the same number of ions. The bigger the Ksp of those the more soluble. If they fall apart into different number of pieces you have to do the math.

Which of the following salts is the most soluble (mol/L)? Least soluble? Next to most soluble? Salt a. Ba. CO 3 b. Ca. CO 3 c. Cu. Cl d. Ba. SO 3 e. Ca. Cr. O 4 Ksp 8. 1 e-9 3. 8 e-9 1. 9 e-7 8. 0 e-7 7. 1 e-4


Consider solution containing HF (weak acid) and salt Na. F Species in solution: HF, H 2 O, Na+, FWhat effect does presence of Na. F have on dissociation equilibrium of HF? HF(aq) H+(aq) + F-(aq) Use Le Chatelier’s principle; extra F- (from Na. F) shifts equilibrium to the left Because of this, the [H+] will decrease What happens to the p. H? This is the Common Ion Effect

34. 6 g of NH 4 Cl is added to 3. 98 L of a 0. 0145 M solution of NH 3. Kb(NH 3) = 1. 8 x 10 -5. What is the p. H of the original solution before the addition of NH 4 Cl? p. H=10. 7 NH 3(aq) + H 2 O(l) NH 4+(aq) + OH-(aq) So – what’s the p. H of the solution after the addition of the NH 4 Cl (assume that the volume stays constant)? p. H=8. 2

What is the general statement that we can make about what happens to the acidity of a weak acid upon addition of a common ion? Should go up!! Do to Lechatliers Principle.

How does the presence of other ion effect the solubility calculations? The importance of the Ksp becomes apparent when you consider the solubility of one salt in the solution of another having the same cations. Each salt contributes the same cation (Ca 2+) The effect is to make calcium oxalate less soluble than it would be in pure water. For example, suppose you wish to know the solubility of calcium oxalate in a solution of calcium chloride.

What is the molar solubility of calcium oxalate in 0. 15 M calcium chloride? The Ksp for calcium oxalate is 2. 3 x 10 -9. Note that before the calcium oxalate dissolves, there is already 0. 15 M Ca 2+ in the solution. H 2 O Starting 0. 15 0 Change +x +x Equilibrium 0. 15+x x

What is the molar solubility of calcium oxalate in 0. 15 M calcium chloride? The Ksp for calcium oxalate is 2. 3 x 10 -9. You substitute into the equilibriumconstant equation

What is the molar solubility of calcium oxalate in 0. 15 M calcium chloride? The Ksp for calcium oxalate is 2. 3 x 10 -9. Now rearrange this equation to give We expect x to be negligible compared to 0. 15.

What is the molar solubility of calcium oxalate in 0. 15 M calcium chloride? The Ksp for calcium oxalate is 2. 3 x 10 -9. Now rearrange this equation to give

What is the molar solubility of calcium oxalate in 0. 15 M calcium chloride? The Ksp for calcium oxalate is 2. 3 x 10 -9. Therefore, the molar solubility of calcium oxalate in 0. 15 M Ca. Cl 2 is 1. 5 x 10 -8 M. In pure water, the molarity was 4. 8 x 10 -5 M, which is over 3000 times greater.


FACTORS WHICH AFFECT SOLUBILITY Important solution factors which may affect the solubility of ionic substances in water Temperature - solubility is temperature dependent Common ions - the presence of common ions in solution lowers solubility p. H - solution p. H can affect solubility if the anion of the ionic solid (i. e. , X) is the conjugate base of a weak acid Complex ions - ammonia reacts with many metal ions in solution through complexation reactions

Precipitation is merely another way of looking at solubility equilibrium. Rather than considering how much of a substance will dissolve, we ask: Will precipitation occur for a given starting ion concentration?

To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. To predict the direction of reaction, you compare Qc with Kc (Chapter 13). The reaction quotient has the same form as the Ksp expression, but the concentrations of products are starting values.

To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. Consider the following equilibrium. H 2 O

To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. The Qc expression is where initial concentration is denoted by i.

To determine whether an equilibrium system will go in the forward or reverse direction requires that we evaluate the reaction quotient, Qc. If Qc exceeds the Ksp, precipitation occurs. If Qc is less than Ksp, more solute can dissolve. If Qc equals the Ksp, the solution is saturated.

The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate to precipitate? Ksp for calcium oxalate is 2. 3 x 10 -9. The ion product quotient, Qc, is:

The concentration of calcium ion in blood plasma is 0. 0025 M. If the concentration of oxalate ion is 1. 0 x 10 -7 M, do you expect calcium oxalate to precipitate? Ksp for calcium oxalate is 2. 3 x 10 -9. This value is smaller than the Ksp, so you do not expect precipitation to occur.

When solutions containing soluble ionic compounds are mixed, when do we predict a precipitate will form? Determine which compound may precipitate after addition (apply solubility rules) Calculate concentrations in solution assuming NO precipitation has occurred Determine Qsp, and compare with Ksp Question: 50. 00 m. L of 0. 00320 M Pb(NO 3)2 is mixed with 50. 00 m. L of 0. 111 M Na. Cl. Will a precipitate form? 1. potential ppt would be Pb. Br 2 (Na. NO 3 is soluble)

2. Look up Ksp of Pb. Cl 2 and write out solubility eq for Pb. Cl 2 in water Pb. Cl 2(s) Pb 2+(aq) + 2 Cl-(aq) Ksp = 1. 6 e-5 Qsp = [Pb 2+][Cl-]2 = (0. 0016)(0. 0555)2 = 4. 93 e-6 Qsp/Ksp <1 thus, no ppt formed

The formation of Mg(OH)2(s) is the only possible precipitate. Mg(OH)2(s) will form if Q > Ksp. Mg(OH)2(s) ⇌ Mg 2+(aq) + 2 OH (aq) Ksp = [Mg 2+][OH ]2 = 8. 9 × 10 -12 (Page A 24) [Mg 2+]o = = 2. 0 × 10 -4 M [OH ]o = = 1. 0 × 10 -4 M Q= Since Q < Ksp, then Mg(OH)2(s) will not precipitate, so no precipitate forms. [Mg 2+]o[OH ]o = (2. 0 × 10 -4 M)(1. 0 × 10 -4 )2 = 2. 0 × 10 -12

Sometimes it is necessary to account for other reactions aqueous ions might undergo. For example, if the anion is the conjugate base of a weak acid, it will react with H 3 O+. You should expect the solubility to be affected by p. H.

Consider the following equilibrium. H 2 O Because the oxalate ion is conjugate to a weak acid (HC 2 O 4 -), it will react with H 3 O+. H 2 O

According to Le Chatelier’s principle, as C 2 O 42 - ion is removed by the reaction with H 3 O+, more calcium oxalate dissolves. Therefore, you expect calcium oxalate to be more soluble in acidic solution (low p. H) than in pure water.

The formation of soluble metal complexes increases the solubility of sparingly soluble ionic solids An example is the reaction of silver ion with ammonia: Ag. Cl(s) Ag+(aq) + Cl-(aq) Ksp = 1. 8 x 10 -10 Ag+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq) Kf = 2. 0 x 107 Kc = Ksp. X Kf = 3. 6 x 10 -3 Ag(NH 3)2+ = complex ion Ag+ = Lewis acid (electron pair acceptor) NH 3 = Lewis base (electron pair donor) The chemical processes are coupled, such that Ag. Cl(s) + 2 NH 3(aq) Ag(NH 3)2+(aq) + Cl-(aq)

A charged ion surrounded by ligands. Ligands are Lewis bases using their lone pair to stabilize the charged metal ions. Common ligands are NH 3, H 2 O, Cl-, CNCoordination number is the number of attached ligands. Cu(NH 3)42+ has a coordination # of 4

Usually the ligand is in large excess. And the individual K’s will be large so we can treat them as if they go to completion. The complex ion will be the biggest ion in solution.
- Slides: 111