Chapter 15 Applying equilibrium The Common Ion Effect

Chapter 15 Applying equilibrium

The Common Ion Effect When the salt with the anion of a weak acid is added to that acid, l Lowers the percent dissociation of the acid. l Ex: Na. F and HF mixed together – F- is the common ion HF ↔ H+ + FAdding F- shifts the equilibrium to the left and therefore fewer H+ ions present l

The same principle applies to salts with the cation of a weak base. l The calculations are the same as last chapter. l NH 4 Cl and NH 3 (NH 4+ is the common ion) l NH 3 + H+ NH 4+ l

Buffered solutions A solution that resists a change in p. H. l Consists of either a weak acid and its salt or a weak base and its salt. l We can make a buffer of any p. H by varying the concentrations of these solutions. l Same calculations as before. l Calculate the p. H of a solution that is. 50 M HAc and. 25 M Na. Ac (Ka = 1. 8 x 10 -5) l

Na+ is a spectator and the reaction we are worried about is HAc H+ + Ac. Initial 0. 50 M Change -x Final 0. 50 -x 0 x x Choose x to be small l We can fill in the table l 0. 25 M x 0. 25+x

HAc Initial 0. 50 M Change -x Final H+ + 0 x Ac- 0. 25 M x 0. 50 -x x 0. 25+x l Do the math l Ka = 1. 8 x 10 -5 x (0. 25) x (0. 25+x) = 1. 8 x 10 -5 = (0. 50) (0. 50 -x) l Assume x is small x = 3. 6 x 10 -5 l Assumption is valid l p. H = -log (3. 6 x 10 -5) = 4. 44

Adding a strong acid or base Do the stoichiometry first. – Use moles not molar l A strong base will grab protons from the weak acid reducing [HA]0 l A strong acid will add its proton to the anion of the salt reducing [A-]0 l Then do the equilibrium problem. l What is the p. H of 1. 0 L of the previous solution when 0. 010 mol of solid Na. OH is added? l

HAc H+ + Ac- 0. 25 mol 0. 50 mol 0. 26 mol 0. 49 mol l In the initial mixture M x L = mol l 0. 50 M HAc x 1. 0 L = 0. 50 mol HAc l 0. 25 M Ac- x 1. 0 L = 0. 25 mol Acl Adding 0. 010 mol OH- will reduce the HAc and increase the Ac- by 0. 010 mole l Because it is in 1. 0 L, we can convert it to molarity

HAc H+ + Ac- 0. 25 mol 0. 50 mol 0. 26 M 0. 49 M l In the initial mixture M x L = mol l 0. 50 M HAc x 1. 0 L = 0. 50 mol HAc l 0. 25 M Ac- x 1. 0 L = 0. 25 mol Acl Adding 0. 010 mol OH- will reduce the HAc and increase the Ac- by 0. 010 mole l Because it is in 1. 0 L, we can convert it to molarity

HAc H+ + Ac 0. 25 mol 0. 26 M 0. 50 mol 0. 49 M l Fill in the table HAc Initial 0. 49 M -x Final 0. 49 -x H+ + 0 x x Ac- 0. 26 M x 0. 26+x

HAc Initial 0. 49 M -x Final H+ + 0 x Ac- 0. 26 M x 0. 49 -x x 0. 26+x l Do the math l Ka = 1. 8 x 10 -5 x (0. 26) x (0. 26+x) = 1. 8 x 10 -5 = (0. 49) (0. 49 -x) l Assume x is small x = 3. 4 x 10 -5 l Assumption is valid l p. H = -log (3. 4 x 10 -5) = 4. 47

Notice If we had added 0. 010 mol of Na. OH to 1 L of water, the p. H would have been. l 0. 010 M OHl p. OH = 2 l p. H = 12 l But with a mixture of an acid and its conjugate base the p. H doesn’t change much l Called a buffer. l
![General equation Ka = [H+] [A-] [HA] l so [H+] = Ka [HA] [A-] General equation Ka = [H+] [A-] [HA] l so [H+] = Ka [HA] [A-]](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-13.jpg)
General equation Ka = [H+] [A-] [HA] l so [H+] = Ka [HA] [A-] l The [H+] depends on the ratio [HA]/[A-] l taking the negative log of both sides l p. H = -log(Ka [HA]/[A-]) l p. H = -log(Ka)-log([HA]/[A-]) l p. H = p. Ka + log([A-]/[HA]) l
![This is called the Henderson. Hasselbach equation p. H = p. Ka + log([A-]/[HA]) This is called the Henderson. Hasselbach equation p. H = p. Ka + log([A-]/[HA])](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-14.jpg)
This is called the Henderson. Hasselbach equation p. H = p. Ka + log([A-]/[HA]) l p. H = p. Ka + log(base/acid) l Works for an acid and its salt l Like HNO 2 and Na. NO 2 l Or a base and its salt l Like NH 3 and NH 4 Cl l But remember to change Kb to Ka l

Calculate the p. H of the following l 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) l lp. H = 3. 38

Calculate the p. H of the following l 0. 25 M NH 3 and 0. 40 M NH 4 Cl (Kb = 1. 8 x 10 -5) l Ka = 1 x 10 -14 1. 8 x 10 -5 l Ka = 5. 6 x 10 -10 l remember its the ratio base over acid l lp. H = 9. 05

Prove they’re buffers What would the p. H be if. 020 mol of HCl is added to 1. 0 L of both of the preceding solutions. l What would the p. H be if 0. 050 mol of solid Na. OH is added to 1. 0 L of each of the proceeding. l Remember adding acids increases the acid side, l Adding base increases the base side. l

Prove they’re buffers What would the p. H be if. 020 mol of HCl is added to 1. 0 L of preceding solutions. l 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) HC 3 H 5 O 3 H + + C 3 H 5 O 3 l Initially After acid 0. 75 mol 0. 77 mol 0 0. 25 mol 0. 23 mol Compared to 3. 38 before acid was added

Prove they’re buffers What would the p. H be if 0. 050 mol of solid Na. OH is added to 1. 0 L of the solutions. l 0. 75 M lactic acid (HC 3 H 5 O 3) and 0. 25 M sodium lactate (Ka = 1. 4 x 10 -4) HC 3 H 5 O 3 H + + C 3 H 5 O 3 l Initially After acid 0. 75 mol 0. 70 mol 0 0. 25 mol 0. 30 mol Compared to 3. 38 before acid was added

Prove they’re buffers What would the p. H be if. 020 mol of HCl is added to 1. 0 L of preceding solutions. l 0. 25 M NH 3 and 0. 40 M NH 4 Cl Kb = 1. 8 x 10 -5 so Ka = 5. 6 x 10 -10 NH 4+ H+ + NH 3 Initially 0. 40 mol 0 0. 25 mol After acid 0. 42 mol 0. 23 mol l Compared to 9. 05 before acid was added

Prove they’re buffers What would the p. H be if 0. 050 mol of solid Na. OH is added to 1. 0 L each solutions. l 0. 25 M NH 3 and 0. 40 M NH 4 Cl Kb = 1. 8 x 10 -5 so Ka = 5. 6 x 10 -10 NH 4+ H+ + NH 3 Initially 0. 40 mol 0 0. 25 mol After acid 0. 35 mol 0. 30 mol l Compared to 9. 05 before acid was added

Buffer capacity The p. H of a buffered solution is determined by the ratio [A-]/[HA]. l As long as this doesn’t change much the p. H won’t change much. l The more concentrated these two are the more H+ and OH- the solution will be able to absorb. l Larger concentrations = bigger buffer capacity. l

Buffer Capacity Calculate the change in p. H that occurs when 0. 040 mol of HCl(g) is added to 1. 0 L of each of the following: l 5. 00 M HAc and 5. 00 M Na. Ac l 0. 050 M HAc and 0. 050 M Na. Ac l Ka= 1. 8 x 10 -5 l l p. H = p. Ka

Buffer Capacity Calculate the change in p. H that occurs when 0. 040 mol of HCl(g) is added to 1. 0 L of 5. 00 M HAc and 5. 00 M Na. Ac l Ka= 1. 8 x 10 -5 HAc H+ + Ac. Initially 5. 00 mol 0 5. 00 mol After acid 5. 04 mol 4. 96 mol l Compared to 4. 74 before acid was added

Buffer Capacity Calculate the change in p. H that occurs when 0. 040 mol of HCl(g) is added to 1. 0 L of 0. 050 M HAc and 0. 050 M Na. Ac l Ka= 1. 8 x 10 -5 HAc H+ + Ac. Initially 0. 050 mol 0 0. 050 mol After acid 0. 090 mol 0 0. 010 mol l Compared to 4. 74 before acid was added
![Buffer capacity The best buffers have a ratio [A-]/[HA] = 1 l This is Buffer capacity The best buffers have a ratio [A-]/[HA] = 1 l This is](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-26.jpg)
Buffer capacity The best buffers have a ratio [A-]/[HA] = 1 l This is most resistant to change l True when [A-] = [HA] l Makes p. H = p. Ka (since log 1 = 0) l

Titrations

Titrations Millimole (mmol) = 1/1000 mol l Molarity = mmol/m. L = mol/L l Makes calculations easier because we will rarely add liters of solution. l Adding a solution of known concentration until the substance being tested is consumed. l This is called the equivalence point. l Where moles of acid = moles of base l
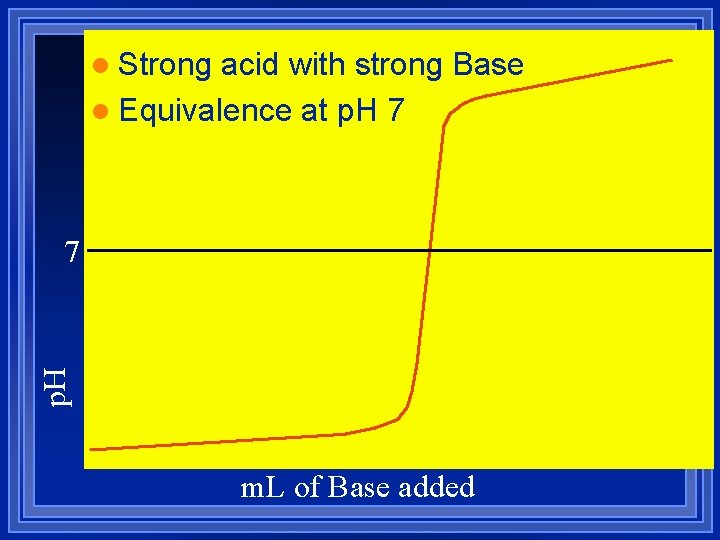
l Strong acid with strong Base l Equivalence at p. H 7 m. L of Base added
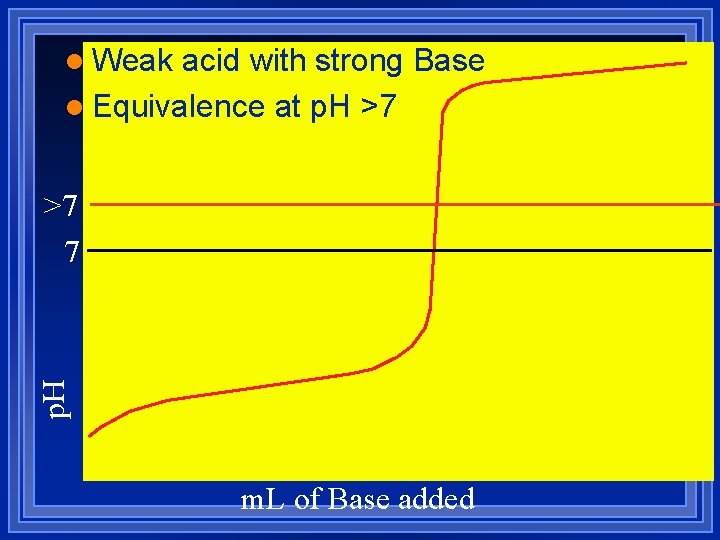
l Weak acid with strong Base l Equivalence at p. H >7 7 m. L of Base added
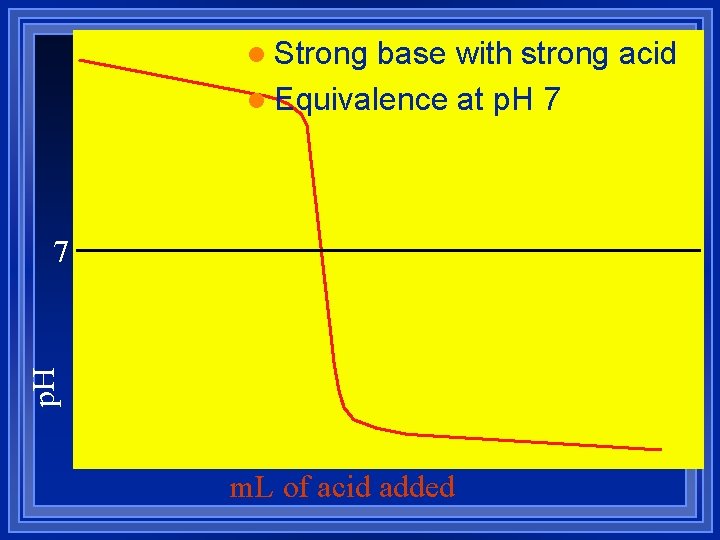
l Strong base with strong acid l Equivalence at p. H 7 m. L of acid added
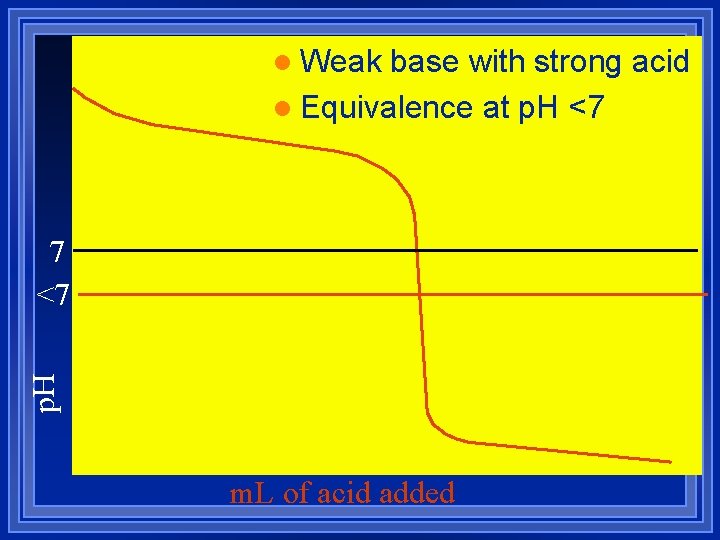
l Weak base with strong acid l Equivalence at p. H <7 p. H 7 <7 m. L of acid added

Strong acid with Strong Base Do the stoichiometry. l m. L x M = mmol l There is no equilibrium. l They both dissociate completely. l The reaction is H+ + OH- HOH l Use [H+] or [OH-] to figure p. H or p. OH l The titration of 20. 0 m. L of 0. 10 M HNO 3 with 0. 10 M Na. OH l

Weak acid with Strong base There is an equilibrium. l Do stoichiometry. – Use moles l Determine major species l Then do equilibrium. Calculate the p. H after 0. 0 m. L, 15. 0 m. L, 25. 0 m. L, and 30. 0 m. L of 0. 10 M Na. OH are added to 25. 0 m. L of 0. 10 M of Nicotinic acid Ka= 1. 4 x 10 -5 l

Summary Strong acid and base just stoichiometry. l Weak acid with 0 ml of base - Ka l Weak acid before equivalence point –Stoichiometry first –Then Henderson-Hasselbach l Weak acid at equivalence point- Kb -Calculate concentration l Weak acid after equivalence - leftover strong base. -Calculate concentration l

Example l A 25. 0 m. L sample of benzoic acid (0. 120 M) is titrated w/ Na. OH (. 210 M) Ka= 6. 3 x 10 -5 – Calculate # moles of Benzoic acid – Calculate volume of Na. OH needed to reach equiv. pt – Calc. the p. H before any base is added – Calc. p. H after 10 ml base is added – Find the p. H at equivalence pt – Calculate the p. H after 15. 3 m. L of OH- is added

Summary Weak base before equivalence point. –Stoichiometry first –Then Henderson-Hasselbach l Weak base at equivalence point Ka. -Calculate concentration l Weak base after equivalence – left over strong acid. -Calculate concentration l

l Titrate 50. 0 m. L of 0. 10 M HF (Ka = 7. 2 x 10 -4) with 0. 10 M Na. OH

Indicators Weak acids that change color when they become bases. l weak acid written HIn l Weak base l HIn H+ + Inclear red l Equilibrium is controlled by p. H l End point - when the indicator changes color. l Try to match the equivalence point l

Indicators Since it is an equilibrium the color change is gradual. l It is noticeable when the ratio of [In-]/[HI] is 1/10 or [HI]/[In-] is 10/1 l Since the Indicator is a weak acid, it has a Ka. l p. H the indicator changes is l p. H=p. Ka +log([In-]/[HI]) = p. Ka +log(1/10) l p. H=p. Ka - 1 l
![Indicators p. H=p. Ka + log([HI]/[In-]) = p. Ka + log(10) l p. H=p. Indicators p. H=p. Ka + log([HI]/[In-]) = p. Ka + log(10) l p. H=p.](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-41.jpg)
Indicators p. H=p. Ka + log([HI]/[In-]) = p. Ka + log(10) l p. H=p. Ka+1 l Choose the indicator with a p. Ka 1 more than the p. H at equivalence point if you are titrating with base. l Choose the indicator with a p. Ka 1 less than the p. H at equivalence point if you are titrating with acid. l

Sample Problem l Two drops of indicator HIn (Ka= 1. 0 x 10 -9), hwere HIn is yellow and In- is blue are placed in 100. 0 m. L of 0. 10 M HCl. – What color is the solution initially? – The solution is titrated with 0. 10 M Na. OH. At what p. H will the color change (greenish yellow) occur? – What color will the solution be after 200. 0 m. L of Na. OH has been added?

l What color will the solution be after 200. 0 m. L of Na. OH has been added?

Solubility Equilibria Will it all dissolve, and if not, how much?

All dissolving is an equilibrium. l If there is not much solid it will all dissolve. l As more solid is added the solution will become saturated. l Solid dissolved l The solid will precipitate as fast as it dissolves. l Equilibrium l

General equation M+ stands for the cation (usually metal). l Nm- stands for the anion (a nonmetal). l Ma. Nmb(s) a. M+(aq) + b. Nm- (aq) l Remember the concentration of a solid doesn’t change. So, what’s the K expression? l Ksp = [M+]a[Nm-]b l Called the solubility product for each compound. l

Watch out Solubility is not the same as solubility product. l Solubility product is an equilibrium constant. l it doesn’t change except with temperature. l Solubility is an equilibrium positionusually expressed as a Molarity for how much can dissolve. l

Calculating Solubility Ex: Calc. Solubility of Ca. C 2 O 4 if the value of Ksp= 4. 8 x 10 -5 mol/L Ca. C 2 O 4 (s) ↔ Ca+2(aq) + C 2 O 4 -2 (aq) l Ksp = [Ca+2] [C 2 O 4 -2] Ksp= [s]2 = 4. 8 x 10 -5 = s 2 s= 6. 9 x 10 -3 M

Calculating Ksp The solubility of copper(I) bromide is 2. 0 x 10 -4 mol/L. Calc. Ksp value. Cu. Br(s) ↔ Cu+1(aq) + Br-1 (aq) I. 0002 M 0 M 0 M C -. 0002 M +. 0002 M E 0. 0002 M l Ksp = [. 0002] = 4. 0 x 10 -8 (units usually omitted)

l The solubility of Calcium Carbonate is 8. 7 x 10 -9 mol/L. Calc. Ksp.

Calculating Ksp The solubility of iron(II) oxalate Fe. C 2 O 4 is 65. 9 mg/L l The solubility of Li 2 CO 3 is 5. 48 g/L l

Common Ion Effect If we try to dissolve the solid in a solution with either the cation or anion already present less will dissolve. l Calculate the solubility of Sr. SO 4, with a Ksp of 3. 2 x 10 -7 in M in a solution of 0. 010 M Na 2 SO 4. l Calculate the solubility of Ca. F 2 Ksp= 4. 0 x 10 -11 in a 0. 025 M Na. F solution. l

Ag. Cl and Na. Cl in solution l l l Cl- is the common ion When [Ag+] [Cl-] < Ksp, no precipitate will be formed. When [Ag+] [Cl-] > Ksp, a precipitate will be formed.

p. H and solubility OH- can be a common ion. l More soluble in acid. l For other anions if they come from a weak acid they are more soluble in a acidic solution than in water. l Ca. C 2 O 4 Ca+2 + C 2 O 4 -2 l H+ + C 2 O 4 -2 HC 2 O 4 l Reduces [C 2 O 4 -2] in acidic solution. l

Sample problem l What is the p. H in a saturated solution of Ca(OH)2? Ksp = 5. 5 x 10 -6 for Ca(OH)2.
![Q and Precipitation not necessarily at Equil. l Q= Ion product l Q =[M+]a[Nm-]b Q and Precipitation not necessarily at Equil. l Q= Ion product l Q =[M+]a[Nm-]b](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-56.jpg)
Q and Precipitation not necessarily at Equil. l Q= Ion product l Q =[M+]a[Nm-]b l If Q>Ksp a precipitate forms. (supersaturated) l If Q<Ksp No precipitate. (unsaturated) l If Q = Ksp equilibrium. (saturated) l
![Example 1 l Chemical analysis gave [Pb 2+] = 0. 012 M, and [Br-] Example 1 l Chemical analysis gave [Pb 2+] = 0. 012 M, and [Br-]](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-57.jpg)
Example 1 l Chemical analysis gave [Pb 2+] = 0. 012 M, and [Br-] = 0. 024 M in a solution. From a table, you find Ksp for Pb. Br 2 has a value of 4 x 10 -5. Is the solution saturated, oversaturated or unsaturated?

Example 2 l A solution of 750. 0 m. L of 4. 00 x 10 -3 M Ce(NO 3)3 is added to 300. 0 m. L of 2. 00 x 10 -2 M KIO 3. Will Ce(IO 3)3 (Ksp= 1. 9 x 10 -10 M) precipitate and if so, what is the concentration of the ions?

Selective Precipitations Used to separate mixtures of metal ions in solutions. l Add anions that will only precipitate certain metals at a time. l Used to purify mixtures. l l Often use H 2 S because in acidic solution Hg+2, Cd+2, Bi+3, Cu+2, Sn+4 will precipitate.
![Selective Precipitation Then add OH-solution [S-2] will increase so more soluble sulfides will precipitate. Selective Precipitation Then add OH-solution [S-2] will increase so more soluble sulfides will precipitate.](http://slidetodoc.com/presentation_image_h/ec67230a578bcc47fa9f1d1761f4d14b/image-60.jpg)
Selective Precipitation Then add OH-solution [S-2] will increase so more soluble sulfides will precipitate. l Co+2, Zn+2, Mn+2, Ni+2, Fe+2, Cr(OH)3, Al(OH)3 l

Selective precipitation Follow the steps l First with insoluble chlorides (Ag, Pb, Ba) l Then sulfides in Acid. l Then sulfides in base. l Then insoluble carbonate (Ca, Ba, Mg) l Alkali metals and NH 4+ remain in solution. l

Complex ion Equilibria A charged ion surrounded by ligands. l Ligands are Lewis bases using their lone pair to stabilize the charged metal ions. l Common ligands are NH 3, H 2 O, Cl-, CNl Coordination number is the number of attached ligands. l Cu(NH 3)42+ has a coordination # of 4 l

The addition of each ligand has its own equilibrium Usually the ligand is in large excess. l And the individual K’s will be large so we can treat them as if they go to completion. l The complex ion will be the biggest ion in solution. l

Calculate the concentrations of Ag+, Ag(S 2 O 3)-2, and Ag(S 2 O 3)2 -3 in a solution made by mixing 150. 0 m. L of 0. 010 M Ag. NO 3 with 200. 0 m. L of 5. 00 M Na 2 S 2 O 3 l Ag+ + S 2 O 3 -2 Ag(S 2 O 3)K 1=7. 4 x 108 l Ag(S 2 O 3)- + 2 S 2 O 3 -2 Ag(S 2 O 3)2 -3 K 2=3. 9 x 104 (Hint: set up ice problem, determine L. R. ) This problem is found on pg. 768 -769 in your book. Check your work! l

Complex Ions and Solubility The dissolving of solid Ag. Cl in excess NH 3 is represented as… Ag. Cl(s) + 2 NH 3(aq) ↔ Ag(NH 3)2+ (aq) + Cl- (aq) However, this is a result of several steps Ag. Cl(s) ↔ Ag+ + Cl. Ksp= 1. 6 x 10 -10 Ag+ + NH 3 ↔ Ag(NH 3)+ K 1 = 2. 1 x 103 Ag(NH 3)+ + NH 3 ↔ Ag(NH 3)2+ K 2= 8. 2 x 103
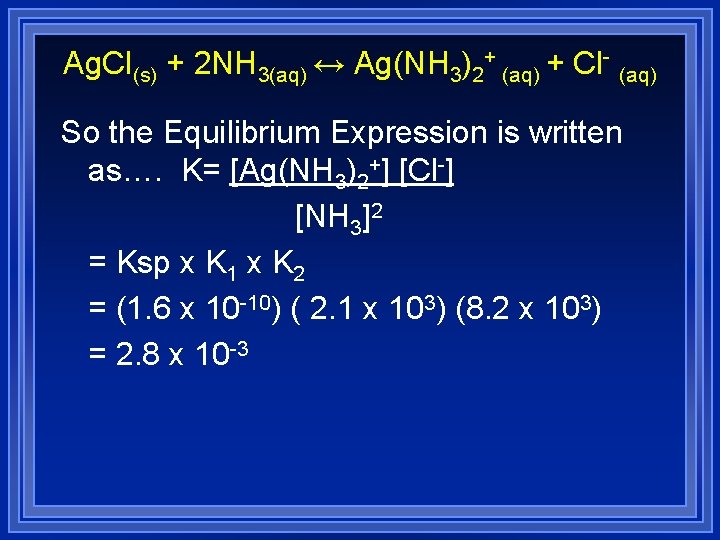
Ag. Cl(s) + 2 NH 3(aq) ↔ Ag(NH 3)2+ (aq) + Cl- (aq) So the Equilibrium Expression is written as…. K= [Ag(NH 3)2+] [Cl-] [NH 3]2 = Ksp x K 1 x K 2 = (1. 6 x 10 -10) ( 2. 1 x 103) (8. 2 x 103) = 2. 8 x 10 -3

Summary 3 Factors that affect solubility: - Common Ions - p. H - Complexing Agents/Ions Mixtures of Ions can be separated by selective precipitation (you know this as Quantitative Analysis) - Steps listed on pg. 765 or in your lab
- Slides: 67