Chapter 15 Applications of Aqueous Equilibria Titrations GENERAL

Chapter 15: Applications of Aqueous Equilibria - Titrations GENERAL CHEMISTRY: ATOMS FIRST John E. Mc. Murray – Robert C. Fay Prentice Hall

Titration • in an acid-base titration, a solution of known concentration (titrant) is slowly added from a burette to a solution of unknown concentration in a flask until the reaction is complete ü when the reaction is complete we have reached the endpoint of the titration • an indicator may be added to determine the endpoint ü an indicator is a chemical that changes color when the p. H changes • when the moles of H 3 O+ = moles of OH−, the titration has reached its equivalence point

Titration

The Titration Curve • is a plot of p. H vs. amount of added titrant • the inflection point of the curve is the equivalence point • • of the titration prior to the equivalence point, the unknown solution in the flask is in excess, so the p. H is closest to its p. H the p. H of the equivalence point depends on the p. H of the salt solution ü equivalence point of neutral salt, p. H = 7 ü equivalence point of acidic salt, p. H < 7 ü equivalence point of basic salt, p. H > 7 • beyond the equivalence point, the known solution in the flask (from the burette) is in excess, so the p. H approaches its p. H
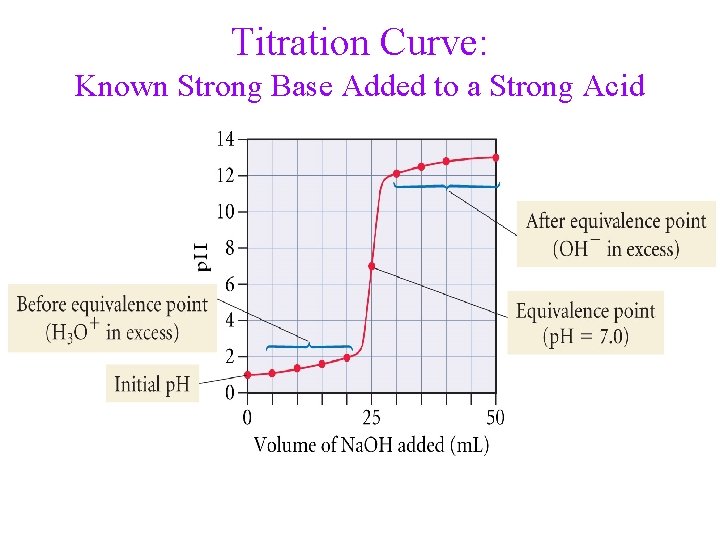
Titration Curve: Known Strong Base Added to a Strong Acid

Strong Acid/Strong Base Titration of 25 m. L of 0. 100 M HCl with 0. 100 M Na. OH HCl(aq) + Na. OH(aq) Na. Cl(aq) + H 2 O(aq) • initial p. H = -log(0. 100) = 1. 00 • initial moles HCl = (0. 0250 L)(0. 100 mol/L) = 2. 50 x 10 -3 mol HCl • now add 5. 0 m. L (0. 0050 L) Na. OH

Titration of 25 m. L of 0. 100 M HCl with 0. 100 M Na. OH

Titration of 25 m. L of 0. 100 M HCl with 0. 100 M Na. OH • when 25 m. L Na. OH added you reach the equivalence point • no HCl and no Na. OH present in flask, p. H = 7. 00 • after 30 m. L (0. 0300 L) Na. OH added, after the equivalence point:

Titration of 25 m. L of 0. 100 M HCl with 0. 100 M Na. OH [OH-] = 9. 09 x 10 -3
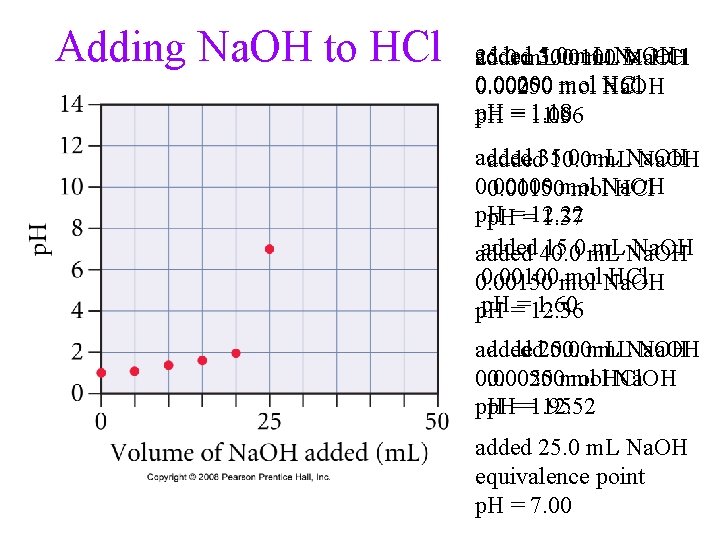
Adding Na. OH to HCl added 5. 0 m. L 25. 0 m. L 30. 0 0. 100 m. LNa. OH M Na. OH HCl 0. 00200 0. 00250 mol HCl 0. 00050 Na. OH 1. 18 p. H = 11. 96 1. 00 added 35. 0 10. 0 m. LNa. OH 0. 00100 0. 00150 mol mol. Na. OH HCl p. H==12. 22 1. 37 added 40. 0 15. 0 m. LNa. OH added 0. 00100 mol mol. Na. OH HCl 0. 00150 p. H==12. 36 1. 60 p. H added 20. 0 50. 0 m. LNa. OH 0. 00050 0. 00250 mol mol. HCl Na. OH p. H==1. 95 12. 52 added 25. 0 m. L Na. OH equivalence point p. H = 7. 00

Titration of 25 m. L of 0. 100 M HCl with 0. 100 M Na. OH • HCHO 2(aq) + Na. OH(aq) Na. CHO 2 (aq) + H 2 O(l) • after equivalence point

Titrating Weak Acid with a Strong Base • the initial p. H is that of the weak acid solution ücalculate like a weak acid equilibrium problem Øe. g. , 15. 5 and 15. 6 • before the equivalence point, the solution becomes a buffer ücalculate mol HAinit and mol A−init using reaction stoichiometry ücalculate p. H with Henderson-Hasselbalch using mol HAinit and mol A−init • half-neutralization p. H = p. Ka

Titrating Weak Acid with a Strong Base • at the equivalence point, the mole HA = mol Base, so the resulting solution has only the conjugate base anion in it before equilibrium is established ümol A− = original mole HA Øcalculate the volume of added base like Ex 4. 8 ü[A−]init = mol A−/total liters ücalculate like a weak base equilibrium problem Øe. g. , 15. 14 • beyond equivalence point, the OH is in excess ü[OH−] = mol MOH xs/total liters ü[H 3 O+][OH−]=1 x 10 -14
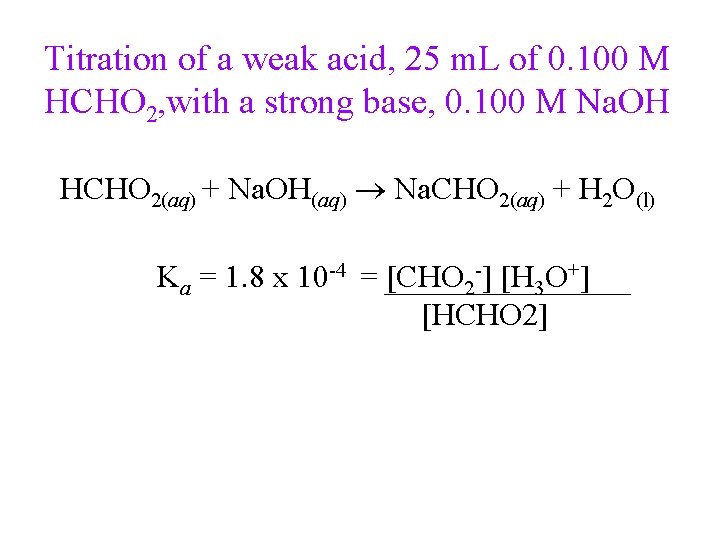
Titration of a weak acid, 25 m. L of 0. 100 M HCHO 2, with a strong base, 0. 100 M Na. OH HCHO 2(aq) + Na. OH(aq) Na. CHO 2(aq) + H 2 O(l) Ka = 1. 8 x 10 -4 = [CHO 2 -] [H 3 O+] [HCHO 2]

Titration of a weak acid, 25 m. L of 0. 100 M HCHO 2, with a strong base, 0. 100 M Na. OH HCHO 2(aq) + H 2 O(l) CHO 2 -(aq) + H 3 O+(aq) [HCHO 2] [CHO 2 -] [H 3 O+] initial 0. 100 0. 000 ≈0 change -x +x +x x x equilibrium 0. 100 - x Ka = 1. 8 x 10 -4

Titration of a weak acid, 25 m. L of 0. 100 M HCHO 2, with a strong base, 0. 100 M Na. OH HCHO 2(aq) + Na. OH(aq) Na. CHO 2 (aq) + H 2 O(l) • initial mol of HCHO 2 = 0. 0250 L x 0. 100 mol/L = 2. 50 x 10 -3 mols start mols added mols after HA A- OH− 2. 50 E-3 0 0 - - 5. 0 E-4 2. 00 E-3 5. 0 E-4 given Ka = 1. 8 x 10 -4 ≈0 add 5. 0 m. L Na. OH

Titration of a weak acid, 25 m. L of 0. 100 M HCHO 2, with a strong base, 0. 100 M Na. OH • HCHO 2(aq) + Na. OH(aq) Na. CHO 2 (aq) + H 2 O(aq) • initial mol of HCHO 2 = 0. 0250 L x 0. 100 mol/L = 2. 50 x 10 -3 • at equivalence CHO 2−(aq) + H 2 O(l) HCHO 2(aq) + OH−(aq) HA A- OH− mols Before 2. 50 E-3 0 0 mols added - - 2. 50 E-3 mols After 0 2. 50 E-3 ≈0 added 25. 0 m. L Na. OH

Titration of a weak acid, 25 m. L of 0. 100 M HCHO 2, with a strong base, 0. 100 M Na. OH** • HCHO 2(aq) + Na. OH(aq) Na. CHO 2 (aq) + H 2 O(aq) • initial mol of HCHO 2 = 0. 0250 L x 0. 100 mol/L = 2. 50 x 10 -3 • at equivalence CHO 2−(aq) + H 2 O(l) HCHO 2(aq) + OH−(aq) [HCHO 2] [CHO 2 -] [OH−] initial 0 0. 0500 ≈0 change +x -x +x equilibrium x 5. 00 E-2 -x x [OH-] = 1. 7 x 10 -6 M Kb = 5. 6 x 10 -11
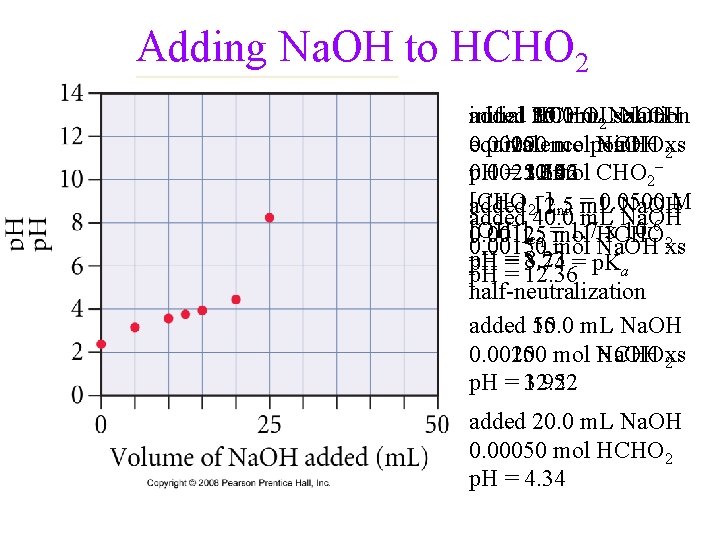
Adding Na. OH to HCHO 2 added initial HCHO 30. 0 35. 0 25. 0 m. L 10. 0 m. L solution Na. OH 2 Na. OH 0. 00050 0. 00100 equivalence 0. 00250 molpoint 0. 00200 0. 00150 Na. OH HCHO 2 xs p. H = 12. 22 0. 00250 11. 96 2. 37 3. 14 3. 56 mol CHO 2− −] [CHO 0. 0500 M added m. L Na. OH 212. 5 init = added m. L Na. OH −] 40. 0 [OH = 1. 7 x 10 -6 2 0. 00125 eq mol HCHO 0. 00150 mol Na. OH xs p. H = 8. 23 p. H = 3. 74 12. 36= p. Ka half-neutralization added 50. 0 15. 0 m. L Na. OH 0. 00100 mol Na. OH 0. 00250 HCHO 2 xs p. H = 12. 52 3. 92 added 20. 0 m. L Na. OH 0. 00050 mol HCHO 2 p. H = 4. 34

A 40. 0 m. L sample of 0. 100 M HNO 2 is titrated with 0. 200 M KOH. Calculate the volume of KOH at the equivalence point Write an equation for the reaction for B with HA. Use Stoichiometry to determine the volume of added B HNO 2 + KOH NO 2 + H 2 O

A 40. 0 m. L sample of 0. 100 M HNO 2 is titrated with 0. 200 M KOH. Calculate the p. H after adding 5. 00 m. L KOH Write an equation for the reaction for B with HA. HNO 2 + KOH NO 2 + H 2 O Determine the moles of HAbefore & moles of added B Make a stoichiometry table and determine the moles of HA in excess and moles A made HNO 2 - OH− 0 ≈0 mols Before 0. 00400 0. 00100 mols added mols After 0. 00300 0. 00100 ≈ 0

A 40. 0 m. L sample of 0. 100 M HNO 2 is titrated with 0. 200 M KOH. Calculate the p. H after adding 5. 00 m. L KOH. Write an equation for the reaction of HA with H 2 O HNO 2 + H 2 O NO 2 + H 3 O+ Table 15. 5 Ka = 4. 6 x 10 -4 Determine Ka and p. Ka for HA Use the Henderson. Hasselbalch Equation to determine the p. H HNO 2 - OH− 0 ≈0 mols Before 0. 00400 0. 00100 mols added mols After 0. 00300 0. 00100 ≈ 0

A 40. 0 m. L sample of 0. 100 M HNO 2 is titrated with 0. 200 M KOH. Calculate the p. H at the half-equivalence point Write an equation for the reaction for B with HA. Determine the moles of HAbefore & moles of added B Make a stoichiometry table and determine the moles of HA in excess and moles A made HNO 2 + KOH NO 2 + H 2 O at half-equivalence, moles KOH = ½ mole HNO 2 - OH− 0 ≈0 mols Before 0. 00400 0. 00200 mols added mols After 0. 00200 ≈ 0

A 40. 0 m. L sample of 0. 100 M HNO 2 is titrated with 0. 200 M KOH. Calculate the p. H at the half-equivalence point. Write an equation for the reaction of HA with H 2 O HNO 2 + H 2 O NO 2 + H 3 O+ Table 15. 5 Ka = 4. 6 x 10 -4 Determine Ka and p. Ka for HA Use the Henderson. Hasselbalch Equation to determine the p. H HNO 2 - OH− 0 ≈0 mols Before 0. 00400 0. 00200 mols added mols After 0. 00200 ≈ 0
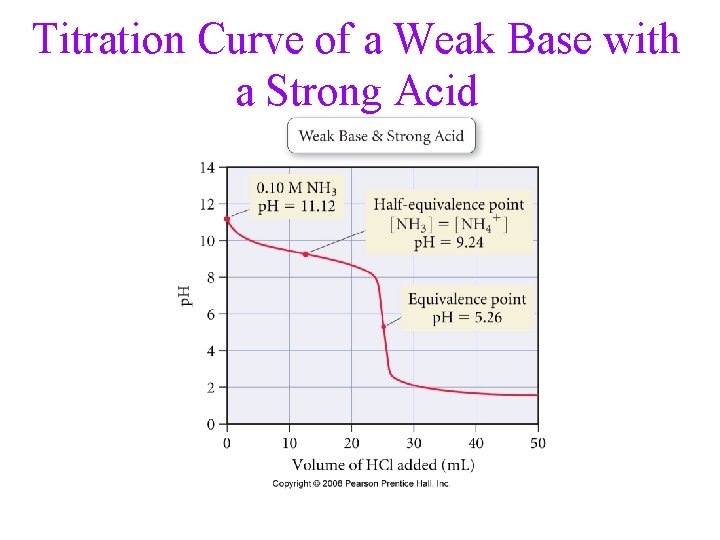
Titration Curve of a Weak Base with a Strong Acid

Titration of a Polyprotic Acid • if Ka 1 >> Ka 2, there will be two equivalence points in the titration üthe closer the Ka’s are to each other, the less distinguishable the equivalence points are titration of 25. 0 m. L of 0. 100 M H 2 SO 3 with 0. 100 M Na. OH

Monitoring p. H During a Titration • the general method for monitoring the p. H during the course of a titration is to measure the conductivity of the solution due to the [H 3 O+] ü using a probe that specifically measures just H 3 O+ • the endpoint of the titration is reached at the • equivalence point in the titration – at the inflection point of the titration curve if you just need to know the amount of titrant added to reach the endpoint, we often monitor the titration with an indicator

Monitoring p. H During a Titration

Monitoring a Titration with an Indicator • for most titrations, the titration curve shows a • very large change in p. H for very small additions of base near the equivalence point an indicator can therefore be used to determine the endpoint of the titration if it changes color within the same range as the rapid change in p. H üp. Ka of H-Indicator ≈ p. H at equivalence point

Indicators • many dyes change color depending on the p. H of the solution • these dyes are weak acids, establishing an equilibrium with the H 2 O and H 3 O+ in the solution H-Ind(aq) + H 2 O(l) Ind (aq) + H 3 O+(aq) • the color of the solution depends on the relative concentrations of Ind : HInd ü when Ind : H-Ind ≈ 1, the color will be mix of the colors of Ind and HInd ü when Ind : H-Ind > 10, the color will be mix of the colors of Ind ü when Ind : H-Ind < 0. 1, the color will be mix of the colors of H -Ind
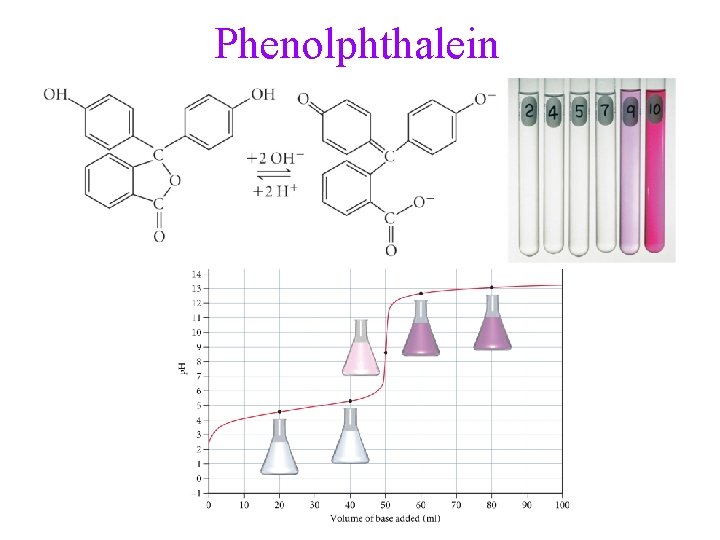
Phenolphthalein
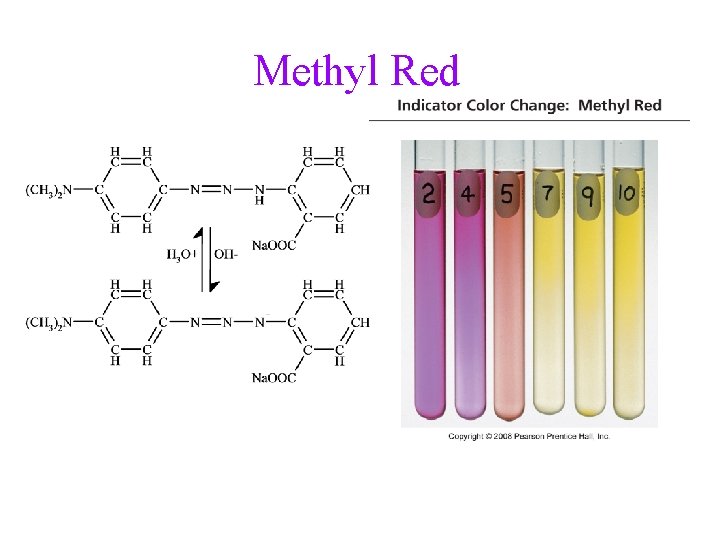
Methyl Red
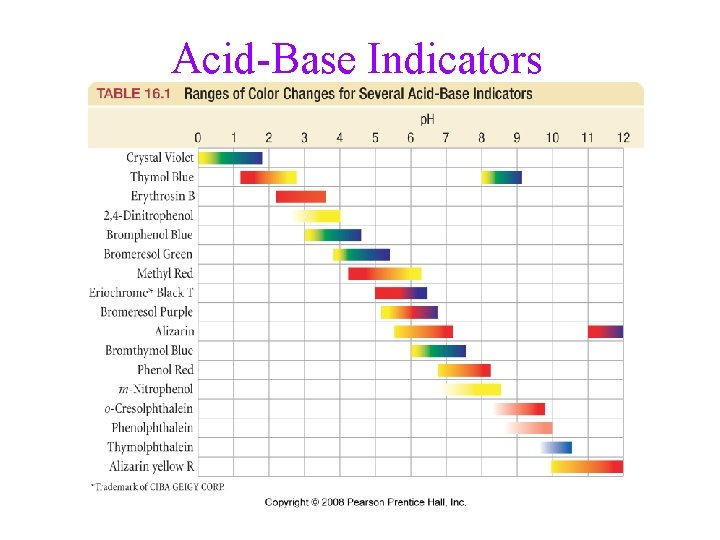
Acid-Base Indicators

Chapter 15: Applications of Aqueous Equilibria-Buffers GENERAL CHEMISTRY: ATOMS FIRST John E. Mc. Murray – Robert C. Fay Prentice Hall

Buffers • buffers are solutions that resist changes in p. H • • • when an acid or base is added they act by neutralizing the added acid or base there is a limit to their neutralizing ability, eventually the p. H changes Solution made by mixing a weak acid with a soluble salt containing its conjugate base anion
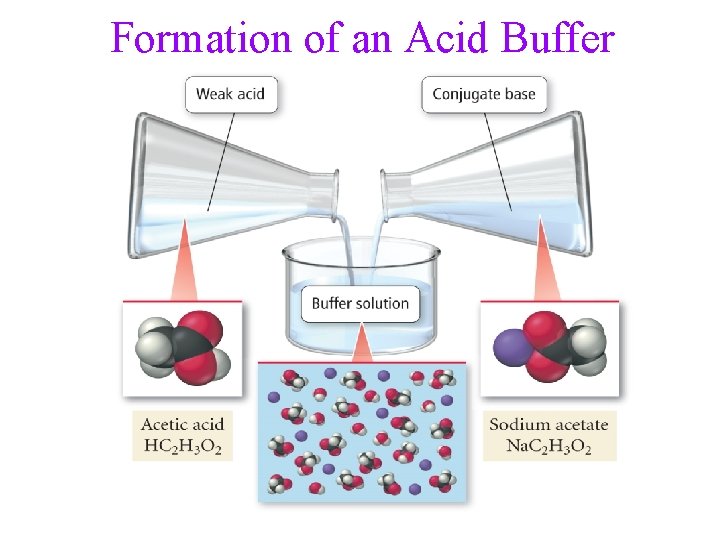
Formation of an Acid Buffer

How Acid Buffers Work HA(aq) + H 2 O(l) A−(aq) + H 3 O+(aq) • buffers follow Le Châtelier’s Principle • buffers contain significant amount of weak acid, HA ü The HA molecules react with added base to neutralize it ü the H 3 O+ combines with OH− to make H 2 O ü H 3 O+ is then replaced by the shifting equilibrium • buffer solutions also contain significant amount of conjugate base anion, A− ü The A− molecules react with added acid to make more HA and keep H 3 O+ constant
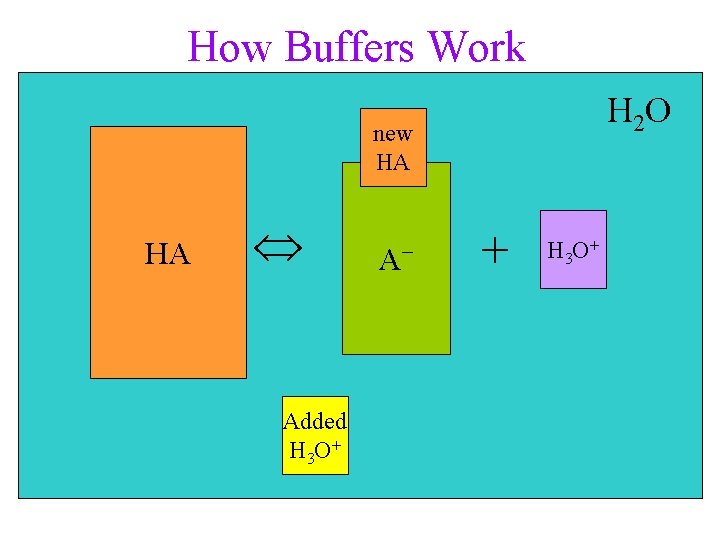
How Buffers Work H 2 O new HA HA HA Added H 3 O+ A−− + H 3 O+
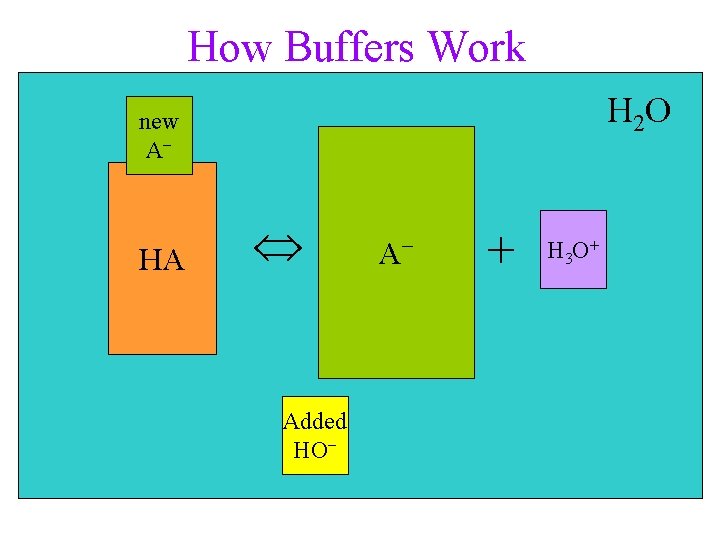
How Buffers Work H 2 O new A− HA HA Added HO− A−− + H 3 O+

Common Ion Effect HA(aq) + H 2 O(l) A−(aq) + H 3 O+(aq) Le Châtelier’s Principle • adding a salt, Na. A, containing the acid anion, • shifts the position of equilibrium to the left (A− is the conjugate base of the acid) this lowers the H 3 O+ ion concentration and causes the p. H to be higher

Common Ion Effect

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Write the reaction for the acid with water Construct an ICE table for the reaction Enter the initial concentrations – assuming the [H 3 O+] from water is ≈ 0 Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 H 2 O + HC 2 H 3 O 2 + H 3 O+ [HA] initial change equilibrium 0. 100 [A-] [H 3 O+] 0. 100 ≈0

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 represent the change in the concentrations in terms of x sum the columns to find the equilibrium concentrations in terms of x substitute into the equilibrium constant expression H 2 O + HC 2 H 3 O 2 + H 3 O+ initial change [HA] [A-] 0. 100 x +x equilibrium 0. 100 x 0. 100 + x [H 3 O+] 0 +x x

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 determine the value of Ka [HA] [A-] [H 3 O+] since Ka is very small, 0. 100 initial ≈0 approximate the change -x +x +x [HA]eq = [HA]init and [A−]eq = [A−]init solve for x equilibrium 0. 100 x 0. 100+x x

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 check if the approximation is valid by seeing if x < 5% of [HC 2 H 3 O 2]init [HA] initial change equilibrium 0. 100 -x 0. 100 x = 1. 8 x 10 -5 the approximation is valid [A-] [H 3 O+] 0. 100 ≈0 +x +x 0. 100 x

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 substitute x into the equilibrium concentration definitions and solve [HA] [A-] [H 3 O+] ≈0 +x 0. 100 initial change -x +x 0. 100 + x 1. 8 E-5 x equilibrium 0. 100 x = 1. 8 x 10 -5

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 substitute [H 3 O+] into the formula for p. H and solve [HA] [A-] +x [H 3 O+] ≈0 +x 0. 100 initial change -x equilibrium 0. 100 1. 8 E-5

What is the p. H of a buffer that is 0. 100 M HC 2 H 3 O 2 and 0. 100 M Na. C 2 H 3 O 2? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 check by substituting the equilibrium initial concentrations back into the equilibrium constant change expression and equilibrium comparing the calculated Ka to the given Ka the values match [HA] [A-] 0. 100 -x +x [H 3 O+] ≈0 +x 0. 100 1. 8 E-5

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Write the reaction for the acid with water Construct an ICE table for the reaction Enter the initial concentrations – assuming the [H 3 O+] from water is ≈ 0 Ka for HF = 7. 0 x 10 -4 H 2 O + HF F + H 3 O+ [HA] initial change equilibrium 0. 14 [A-] [H 3 O+] 0. 071 ≈0

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? represent the change in the concentrations in terms of x sum the columns to find the equilibrium concentrations in terms of x substitute into the equilibrium constant expression Ka for HF = 7. 0 x 10 -4 H 2 O + HF F + H 3 O+ initial change [HA] [A-] 0. 14 0. 071 x +x equilibrium 0. 14 x 0. 071 + x [H 3 O+] 0 +x x

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Ka for HF = 7. 0 x 10 -4 determine the value of Ka [HA] [A-] [H 3 O+] since Ka is very small, 0. 14 0. 071 initial ≈0 approximate the change -x +x +x [HA]eq = [HA]init and [A−]eq = [A−]init solve for x equilibrium 0. 14 0. 100+x x 0. 071 0. 012 x

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Ka for HF = 7. 0 x 10 -4 check if the approximation is valid by seeing if x < 5% of [HC 2 H 3 O 2]init [HA] initial change equilibrium 0. 14 -x 0. 14 x = 1. 4 x 10 -3 the approximation is valid [A 2 -] [H 3 O+] 0. 071 ≈0 +x +x 0. 071 x

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Ka for HF = 7. 0 x 10 -4 substitute x into the equilibrium concentration definitions and solve [HA] [A 2 -] [H 3 O+] ≈0 +x 0. 14 0. 071 initial change -x +x 0. 14 x 0. 071 0. 072+ x 1. 4 E-3 x equilibrium 0. 14 x = 1. 4 x 10 -3

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Ka for HF = 7. 0 x 10 -4 substitute [H 3 O+] into the formula for p. H and solve initial change equilibrium [HA] [A-] 0. 14 0. 071 -x +x [H 3 O+] ≈0 +x 0. 14 0. 072 1. 4 E-3

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? Ka for HF = 7. 0 x 10 -4 check by substituting the equilibrium initial concentrations back into the equilibrium constant change expression and equilibrium comparing the calculated Ka to the given Ka the values are close enough [HA] [A-] 0. 14 0. 071 -x +x [H 3 O+] ≈0 +x 0. 14 0. 072 1. 4 E-3

Henderson-Hasselbalch Equation • calculating the p. H of a buffer solution can be • simplified by using an equation derived from the Ka expression called the Henderson. Hasselbalch Equation the equation calculates the p. H of a buffer from the Ka and initial concentrations of the weak acid and salt of the conjugate base üas long as the “x is small” approximation is valid

Deriving the Henderson-Hasselbalch Equation Take the log of both sides and multiply by -1 Remember: Therefore

What is the p. H of a buffer that is 0. 050 M HC 7 H 5 O 2 and 0. 150 M Na. C 7 H 5 O 2? Assume the [HA] and [A-] equilibrium concentrations are the same as the initial Substitute into the Henderson-Hasselbalch Equation Check the “x is small” approximation HC 7 H 5 O 2 + H 2 O C 7 H 5 O 2 + H 3 O+ Ka for HC 7 H 5 O 2 = 6. 5 x 10 -5

What is the p. H of a buffer that is 0. 14 M HF (p. Ka = 3. 15) and 0. 071 M KF? find the p. Ka from the given Ka Assume the [HA] and [A-] equilibrium concentrations are the same as the initial Substitute into the Henderson-Hasselbalch Equation Check the “x is small” approximation HF + H 2 O F + H 3 O+

Do I Use the Full Equilibrium Analysis or the Henderson-Hasselbalch Equation? • • a) b) • the Henderson-Hasselbalch equation is generally good enough when the “x is small” approximation is applicable generally, the “x is small” approximation will work when both of the following are true: the initial concentrations of acid and salt are not very dilute the Ka is fairly small for most problems, this means that the initial acid and salt concentrations should be over 1000 x larger than the value of Ka

How Much Does the p. H of a Buffer Change When an Acid or Base Is Added? • • 1. although buffers do resist change in p. H when acid or base are added to them, their p. H does change calculating the new p. H after adding acid or base requires breaking the problem into 2 parts a stoichiometry calculation for the reaction of the added chemical with one of the ingredients of the buffer to reduce its initial concentration and increase the concentration of the other ü added acid reacts with the A− to make more HA ü added base reacts with the HA to make more A− 2. an equilibrium calculation of [H 3 O+] using the new initial values of [HA] and [A−]

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? If the added chemical is a base, write a reaction for OH− with HA. If the added chemical is an acid, write a reaction for it with A−. Construct a stoichiometry table for the reaction HC 2 H 3 O 2 + OH− C 2 H 3 O 2 + H 2 O HA A- mols Before 0. 100 mols added mols After OH− 0 0. 010

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? HC 2 H 3 O 2 + OH− C 2 H 3 O 2 + H 2 O Fill in the table – tracking the changes in the number of moles for each component mols Before mols added mols After HA A- OH− 0. 100 ≈0 - - 0. 010 0. 090 0. 110 ≈0

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? Write the reaction for the acid with water Construct an ICE table for the reaction Enter the initial concentrations – assuming the [H 3 O+] from water is ≈ 0, and using the new molarities of the [HA] and [A−] HC 2 H 3 O 2 + H 2 O C 2 H 3 O 2 + H 3 O+ [HA] initial change equilibrium 0. 090 [A-] [H 3 O+] 0. 110 ≈0

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? represent the change in the concentrations in terms of x sum the columns to find the equilibrium concentrations in terms of x substitute into the equilibrium constant expression HC 2 H 3 O 2 + H 2 O C 2 H 3 O 2 + H 3 O+ initial change [HA] [A-] 0. 090 0. 110 x +x equilibrium 0. 090 x 0. 110 + x [H 3 O+] 0 +x x

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 determine the value of Ka [HA] [A-] [H 3 O+] since Ka is very small, 0. 100 initial ≈0 approximate the change -x +x +x [HA]eq = [HA]init and [A−]eq = [A−]init solve for x equilibrium 0. 090 x 0. 110+x x

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 check if the approximation is valid by seeing if x < 5% of [HC 2 H 3 O 2]initial change equilibrium [HA] [A-] [H 3 O+] 0. 090 0. 110 ≈0 -x +x 0. 090 0. 110 +x x x = 1. 47 x 10 -5 the approximation is valid

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? substitute x into the equilibrium concentration definitions and solve [HA] [A-] [H 3 O+] ≈0 +x 0. 090 0. 110 initial change -x +x 0. 090 x 0. 110 x equilibrium 0. 090 + x 1. 5 E-5 x = 1. 47 x 10 -5

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? substitute [H 3 O+] into the formula for p. H and solve [HA] [A-] +x [H 3 O+] ≈0 +x 0. 090 initial change -x equilibrium 0. 090 0. 110 1. 5 E-5

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 check by substituting the equilibrium concentrations back into initial the equilibrium constant change expression and equilibrium comparing the calculated Ka to the given Ka the values match [HA] [A-] 0. 090 0. 110 -x +x [H 3 O+] ≈0 +x 0. 090 0. 110 1. 5 E-5

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? find the p. Ka from the given Ka HC 2 H 3 O 2 + H 2 O C 2 H 3 O 2 + H 3 O+ Assume the [HA] and [A-] equilibrium concentrations are the same as the initial Ka for HC 2 H 3 O 2 = 1. 8 x 10 -5 [HA] [A-] [H 3 O+] initial 0. 090 0. 110 ≈0 change -x +x +x 0. 090 0. 110 x equilibrium

What is the p. H of a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L that has 0. 010 mol Na. OH added to it? Substitute into the Henderson-Hasselbalch Equation Check the “x is small” approximation HC 2 H 3 O 2 + H 2 O C 2 H 3 O 2 + H 3 O+ p. Ka for HC 2 H 3 O 2 = 4. 745

Compare the effect on p. H of adding 0. 010 mol Na. OH to a buffer that has 0. 100 mol HC 2 H 3 O 2 and 0. 100 mol Na. C 2 H 3 O 2 in 1. 00 L to adding 0. 010 mol Na. OH to 1. 00 L of pure water? HC 2 H 3 O 2 + H 2 O C 2 H 3 O 2 + H 3 O+ p. Ka for HC 2 H 3 O 2 = 4. 745

What is the p. H of a buffer that is 0. 50 M NH 3 (p. Kb = 4. 75) and 0. 20 M NH 4 Cl? find the p. Ka of the conjugate acid (NH 4+) from the given Kb Assume the [B] and [HB+] equilibrium concentrations are the same as the initial Substitute into the Henderson-Hasselbalch Equation Check the “x is small” approximation NH 3 + H 2 O NH 4+ + OH−

Buffering Effectiveness • a good buffer should be able to neutralize moderate • • amounts of added acid or base however, there is a limit to how much can be added before the p. H changes significantly the buffering capacity is the amount of acid or base a buffer can neutralize the buffering range is the p. H range the buffer can be effective the effectiveness of a buffer depends on two factors (1) the relative amounts of acid and base, and (2) the absolute concentrations of acid and base

Effect of Relative Amounts of Acid & Conjugate Base Buffers are most effective when [acid] = [base] Buffer 1 0. 100 mol HA & 0. 100 mol AInitial p. H = 5. 00 HA + OH− A + H 2 O HA A- OH− mols Before 0. 100 0 mols added - - 0. 010 0. 090 0. 110 ≈0 mols After p. Ka (HA) = 5. 00

Effect of Relative Amounts of Acid & Conjugate Base Buffers are most effective when [acid] = [base] Buffer 12 0. 18 mol HA & 0. 020 mol AInitial p. H = 4. 05 HA mols Before mols added mols After A- 0. 18 0. 020 - - 0. 17 0. 030 OH− 0 0. 01 0 ≈0 HA + OH− A + H 2 O p. Ka (HA) = 5. 00

Effect of Relative Amounts of Acid and Conjugate Base Buffers are most effective when [acid] = [base] p. Ka (HA) = 5. 00 Buffer 1 0. 100 mol HA & 0. 100 mol AInitial p. H = 5. 00 HA + OH− A + H 2 O after adding 0. 010 mol Na. OH p. H = 5. 09 HA A- OH− mols Before 0. 100 0 mols added - - 0. 010 mols After 0. 090 0. 110 ≈0

Effect of Relative Amounts of Acid and Conjugate Base Buffers are most effective when [acid] = [base] Buffer 12 0. 18 mol HA & 0. 020 mol AInitial p. H = 4. 05 p. Ka (HA) = 5. 00 after adding 0. 010 mol Na. OH p. H = 4. 25 HA + OH− A + H 2 O HA A- OH− mols Before 0. 18 0. 020 0 mols added mols After - - 0. 010 0. 17 0. 030 ≈0

Start: Effect of Absolute Concentrations of Acid and Conjugate Base a buffer is most effective when the concentrations of acid and base are largest (0. 1 M vs. 0. 5 M) p. Ka (HA) = 5. 00 Buffer 1 0. 50 mol HA & 0. 50 mol AInitial p. H = 5. 00 HA + OH− A + H 2 O after adding 0. 010 mol Na. OH p. H = 5. 02 HA A- OH− mols Before 0. 500 0 mols added - - 0. 010 mols After 0. 49 0. 51 ≈0 vs. 5. 09 for 0. 1 M vs. 1. 8% for 0. 1 M

Effect of Absolute Concentrations of Acid and Conjugate Base a buffer is most effective when the concentrations of acid and base are largest Buffer 12 0. 050 mol HA & 0. 050 mol AInitial p. H = 5. 00 p. Ka (HA) = 5. 00 HA + OH− A + H 2 O after adding 0. 010 mol Na. OH p. H = 5. 18 HA A- OH− mols Before 0. 050 0 mols added mols After - - 0. 010 0. 040 0. 060 ≈0 Add 0. 01 mol Na. OH
![Effectiveness of Buffers • a buffer will be most effective when the [base]: [acid] Effectiveness of Buffers • a buffer will be most effective when the [base]: [acid]](http://slidetodoc.com/presentation_image_h2/72506c50e388c4e017e0747aed8cbd11/image-82.jpg)
Effectiveness of Buffers • a buffer will be most effective when the [base]: [acid] = 1 üequal concentrations of acid and base • effective when 0. 1 < [base]: [acid] < 10 • a buffer will be most effective when the [acid] and the [base] are large

Buffering Range • we have said that a buffer will be effective when • 0. 1 < [base]: [acid] < 10 substituting into the Henderson-Hasselbalch we can calculate the maximum and minimum p. H at which the buffer will be effective Lowest p. H Highest p. H therefore, the effective p. H range of a buffer is p. Ka ± 1 when choosing an acid to make a buffer, choose one whose is p. Ka is closest to the p. H of the buffer

Which of the following acids would be the best choice to combine with its sodium salt to make a buffer with p. H 4. 25? Chlorous Acid, HCl. O 2 Nitrous Acid, HNO 2 Formic Acid, HCHO 2 Hypochlorous Acid, HCl. O p. Ka = 1. 95 p. Ka = 3. 34 p. Ka = 3. 74 p. Ka = 7. 54

Which of the following acids would be the best choice to combine with its sodium salt to make a buffer with p. H 4. 25? Chlorous Acid, HCl. O 2 Nitrous Acid, HNO 2 Formic Acid, HCHO 2 Hypochlorous Acid, HCl. O p. Ka = 1. 95 p. Ka = 3. 34 p. Ka = 3. 74 p. Ka = 7. 54 The p. Ka of HCHO 2 is closest to the desired p. H of the buffer, so it would give the most effective buffering range.

What ratio of Na. CHO 2 : HCHO 2 would be required to make a buffer with p. H 4. 25? Formic Acid, HCHO 2, p. Ka = 3. 74 to make the buffer with p. H 4. 25, you would use 3. 24 times as much Na. CHO 2 as HCHO 2

Buffering Capacity • buffering capacity is the amount of acid or base that • • can be added to a buffer without destroying its effectiveness the buffering capacity increases with increasing absolute concentration of the buffer components as the [base]: [acid] ratio approaches 1, the ability of the buffer to neutralize both added acid and base improves buffers that need to work mainly with added acid generally have [base] > [acid] buffers that need to work mainly with added base generally have [acid] > [base]

Chapter 15: Applications of Aqueous Equilibria - Solubility Equilibria GENERAL CHEMISTRY: ATOMS FIRST John E. Mc. Murray – Robert C. Fay Prentice Hall

Solubility Equilibria • all ionic compounds dissolve in water to some degree ühowever, many compounds have such low solubility in water that we classify them as insoluble • we can apply the concepts of equilibrium to salts dissolving, and use the equilibrium constant for the process to measure relative solubilities in water

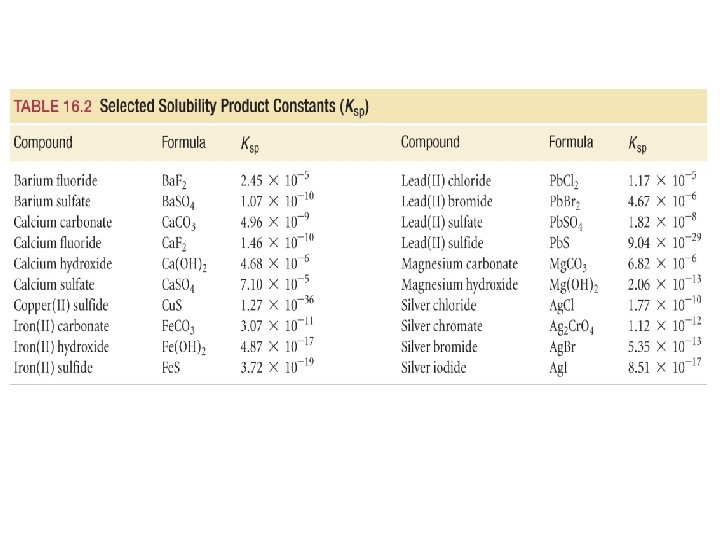
Solubility Product • the equilibrium constant for the dissociation of a solid • • salt into its aqueous ions is called the solubility product, Ksp for an ionic solid Mn. Xm, the dissociation reaction is: Mn. Xm(s) n. Mm+(aq) + m. Xn−(aq) the solubility product would be Ksp = [Mm+]n[Xn−]m for example, the dissociation reaction for Pb. Cl 2 is Pb. Cl 2(s) Pb 2+(aq) + 2 Cl−(aq) and its equilibrium constant is Ksp = [Pb 2+][Cl−]2

Molar Solubility • solubility is the amount of solute that will dissolve in a given amount of solution ü at a particular temperature • the molar solubility is the number of moles of solute that will dissolve in a liter of solution ü the molarity of the dissolved solute in a saturated solution • for the general reaction Mn. Xm(s) n. Mm+(aq) + m. Xn−(aq)
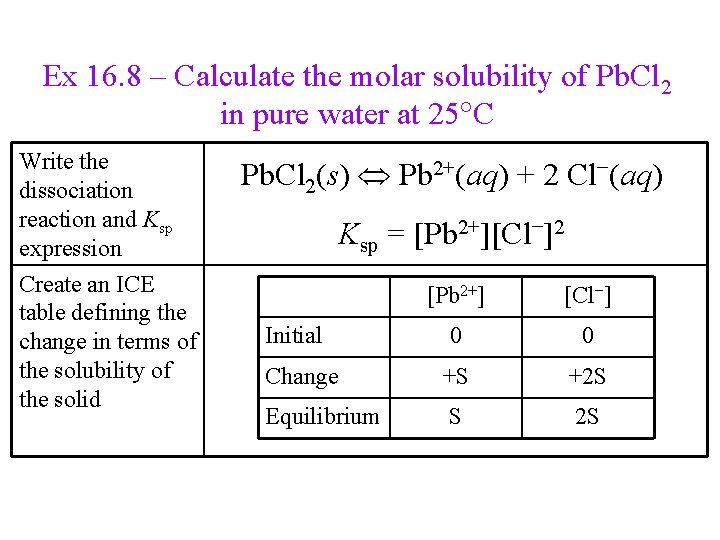
Ex 16. 8 – Calculate the molar solubility of Pb. Cl 2 in pure water at 25 C Write the dissociation reaction and Ksp expression Create an ICE table defining the change in terms of the solubility of the solid Pb. Cl 2(s) Pb 2+(aq) + 2 Cl−(aq) Ksp = [Pb 2+][Cl−]2 [Pb 2+] [Cl−] 0 0 Change +S +2 S Equilibrium S 2 S Initial

Ex 16. 8 – Calculate the molar solubility of Pb. Cl 2 in pure water at 25 C Substitute into the Ksp expression Find the value of Ksp from Table 16. 2, plug into the equation and solve for S Ksp = [Pb 2+][Cl−]2 Ksp = (S)(2 S)2 [Pb 2+] [Cl−] 0 0 Change +S +2 S Equilibrium S 2 S Initial

Practice – Determine the Ksp of Pb. Br 2 if its molar solubility in water at 25 C is 1. 05 x 10 -2 M

Practice – Determine the Ksp of Pb. Br 2 if its molar solubility in water at 25 C is 1. 05 x 10 -2 M Write the dissociation reaction and Ksp expression Create an ICE table defining the change in terms of the solubility of the solid Pb. Br 2(s) Pb 2+(aq) + 2 Br−(aq) Ksp = [Pb 2+][Br−]2 Initial [Pb 2+] [Br−] 0 0 Change +(1. 05 x 10 -2) +2(1. 05 x 10 -2) Equilibrium (1. 05 x 10 -2) (2. 10 x 10 -2)

Practice – Determine the Ksp of Pb. Br 2 if its molar solubility in water at 25 C is 1. 05 x 10 -2 M Substitute into the Ksp expression plug into the equation and solve Ksp = [Pb 2+][Br−]2 Ksp = (1. 05 x 10 -2)(2. 10 x 10 -2)2 Initial [Pb 2+] [Br−] 0 0 Change +(1. 05 x 10 -2) +2(1. 05 x 10 -2) Equilibrium (1. 05 x 10 -2) (2. 10 x 10 -2)

Ksp and Relative Solubility • molar solubility is related to Ksp • but you cannot always compare solubilities of • compounds by comparing their Ksp values in order to compare Ksp values, the compounds must have the same dissociation stoichiometry

The Effect of Common Ion on Solubility • addition of a soluble salt that contains one of the • ions of the “insoluble” salt, decreases the solubility of the “insoluble” salt for example, addition of Na. Cl to the solubility equilibrium of solid Pb. Cl 2 decreases the solubility of Pb. Cl 2(s) Pb 2+(aq) + 2 Cl−(aq) addition of Cl− shifts the equilibrium to the left

Ex 16. 10 – Calculate the molar solubility of Ca. F 2 in 0. 100 M Na. F at 25 C Write the dissociation reaction and Ksp expression Create an ICE table defining the change in terms of the solubility of the solid Ca. F 2(s) Ca 2+(aq) + 2 F−(aq) Ksp = [Ca 2+][F−]2 [Ca 2+] [F−] 0 0. 100 Change +S +2 S Equilibrium S 0. 100 + 2 S Initial

Ex 16. 10 – Calculate the molar solubility of Ca. F 2 in 0. 100 M Na. F at 25 C Substitute into the Ksp expression assume S is small Find the value of Ksp from Table 16. 2, plug into the equation and solve for S Ksp = [Ca 2+][F−]2 Ksp = (S)(0. 100 + 2 S)2 Ksp = (S)(0. 100)2 [Ca 2+] [F−] 0 0. 100 Change +S +2 S Equilibrium S 0. 100 + 2 S Initial

The Effect of p. H on Solubility • for insoluble ionic hydroxides, the higher the p. H, the lower the solubility of the ionic hydroxide ü and the lower the p. H, the higher the solubility ü higher p. H = increased [OH−] • M(OH)n(s) Mn+(aq) + n. OH−(aq) for insoluble ionic compounds that contain anions of weak acids, the lower the p. H, the higher the solubility M 2(CO 3)n(s) 2 Mn+(aq) + n. CO 32−(aq) H 3 O+(aq) + CO 32− (aq) HCO 3− (aq) + H 2 O(l)

Precipitation • precipitation will occur when the concentrations of the • ions exceed the solubility of the ionic compound if we compare the reaction quotient, Q, for the current solution concentrations to the value of Ksp, we can determine if precipitation will occur ü Q = Ksp, the solution is saturated, no precipitation ü Q < Ksp, the solution is unsaturated, no precipitation ü Q > Ksp, the solution would be above saturation, the salt above saturation will precipitate • some solutions with Q > Ksp will not precipitate unless disturbed – these are called supersaturated solutions

precipitation occurs if Q > Ksp a supersaturated solution will precipitate if a seed crystal is added

Selective Precipitation • a solution containing several different cations • can often be separated by addition of a reagent that will form an insoluble salt with one of the ions, but not the others a successful reagent can precipitate with more than one of the cations, as long as their Ksp values are significantly different
![Ex 16. 13 What is the minimum [OH−] necessary to just begin to precipitate Ex 16. 13 What is the minimum [OH−] necessary to just begin to precipitate](http://slidetodoc.com/presentation_image_h2/72506c50e388c4e017e0747aed8cbd11/image-106.jpg)
Ex 16. 13 What is the minimum [OH−] necessary to just begin to precipitate Mg 2+ (with [0. 059]) from seawater? precipitating may just occur when Q = Ksp From the Ksp table: Ksp = 2. 06 x 10 -13
![Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011]) Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011])](http://slidetodoc.com/presentation_image_h2/72506c50e388c4e017e0747aed8cbd11/image-107.jpg)
Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011]) just begins to precipitate from seawater? precipitating Mg 2+ begins when [OH−] = 1. 9 x 10 -6 M
![Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011]) Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011])](http://slidetodoc.com/presentation_image_h2/72506c50e388c4e017e0747aed8cbd11/image-108.jpg)
Ex 16. 14 What is the [Mg 2+] when Ca 2+ (with [0. 011]) just begins to precipitate from seawater? precipitating Mg 2+ begins when [OH−] = 1. 9 x 10 -6 M precipitating Ca 2+ begins when [OH−] = 2. 06 x 10 -2 M when Ca 2+ just begins to precipitate out, the [Mg 2+] has dropped from 0. 059 M to 4. 8 x 10 -10 M

Qualitative Analysis • an analytical scheme that utilizes selective precipitation to identify the ions present in a solution is called a qualitative analysis scheme üwet chemistry • a sample containing several ions is subjected to • the addition of several precipitating agents addition of each reagent causes one of the ions present to precipitate out
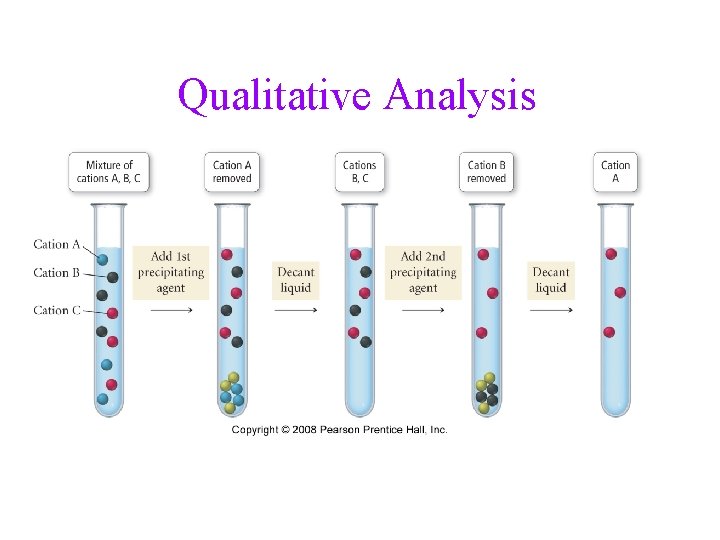
Qualitative Analysis
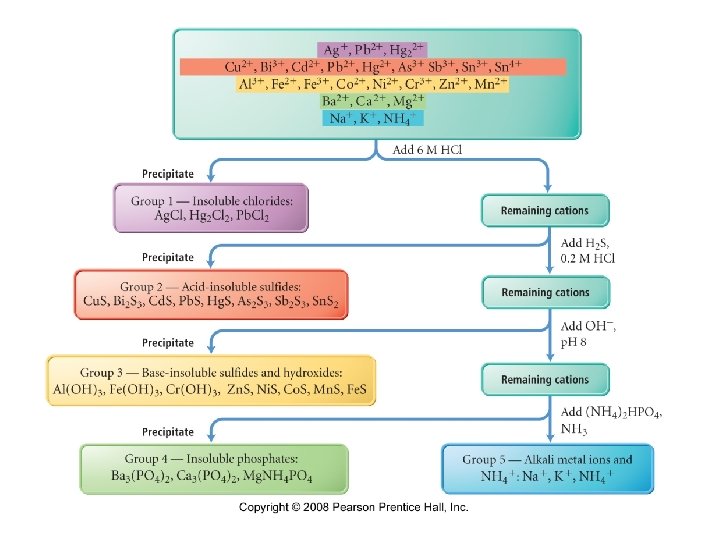

Group 1 Insoluble Chlorides • group one cations are Ag+, Pb 2+, and Hg 22+ • they form water insoluble compounds with Cl− ü Ag. Cl, Pb. Cl 2, Hg 2 Cl 2 üas long as the concentration is large enough üPb. Cl 2 may be borderline Ømolar solubility of Pb. Cl 2 = 1. 43 x 10 -2 M • precipitated by the addition of HCl, which acts to decrease the solubility due to Le. Chatelier’s principle.

Group 2 Acid Insoluble Sulfides • group two cations are Cd 2+, Cu 2+, Bi 3+, Sn 4+, As 3+, Pb 2+, Sb 3+, and Hg 2+ • Hg. S, Cd. S, Cu. S, Sn. S 2 , etc. • they form water insoluble compounds with HS− and S 2− at low p. H • They are precipitated by the addition of H 2 S in HCl

Group 3 Base Insoluble Sulfides & Hydroxides • group three cations are Fe 2+, Co 2+, Zn 2+, Mn 2+, Ni 2+ precipitated as sulfides ü Fe. S, Co. S, Zn. S, Mn. S, Ni. S • Cr 3+, Fe 3+, and Al 3+ precipitated as hydroxides ü Al(OH 3), Fe(OH 3), Cr(OH 3 ) • all these cations form compounds with S 2− that • are insoluble in water at high p. H precipitated by the addition of H 2 S in Na. OH

Group 4 Insoluble Phosphates • group four cations are Mg 2+, Ca 2+, Ba 2+ • all these cations form compounds with PO 43− • that are insoluble in water at high p. H precipitated by the addition of (NH 4)2 HPO 4

Group 5 • group five cations are Na+, K+, NH 4+ • all these cations form compounds that are • soluble in water – they do not precipitate They are identified by the color of their flame

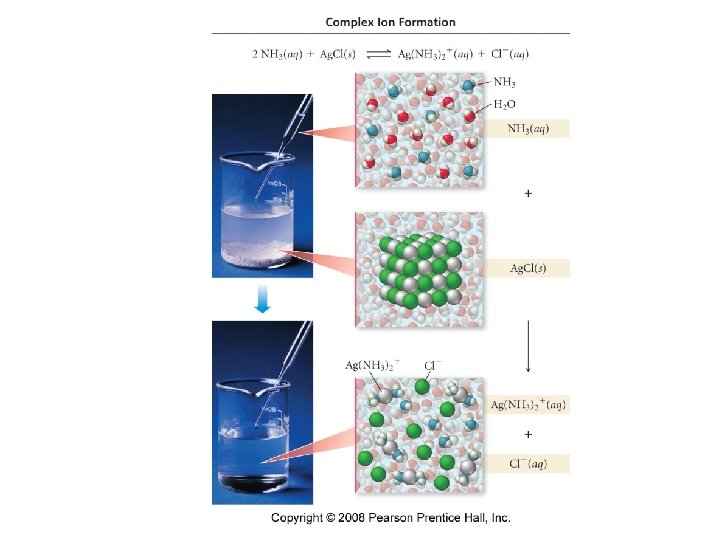
Complex Ion Formation • transition metals tend to be good Lewis acids • they often bond to one or more H 2 O molecules to form a hydrated ion ü H 2 O is the Lewis base, donating electron pairs to form coordinate covalent bonds • Ag+(aq) + 2 H 2 O(l) Ag(H 2 O)2+(aq) ions that form by combining a cation with several anions or neutral molecules are called complex ions ü e. g. , Ag(H 2 O)2+ • the attached ions or molecules are called ligands ü e. g. , H 2 O

Complex Ion Equilibria • if a ligand is added to a solution and it forms a stronger bond than the original ligand, it will replace the original ligand Ag(H 2 O)2+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq) + 2 H 2 O(l) ügenerally H 2 O is not included, since its complex ion is always present in aqueous solution Ag+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq)

Formation Constant • the reaction between an ion and ligands to form • a complex ion is called a complex ion formation reaction Ag+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq) the equilibrium constant for the formation reaction is called the formation constant, Kf

Formation Constants

Ex 16. 15 – 200. 0 m. L of 1. 5 x 10 -3 M Cu(NO 3)2 is mixed with 250. 0 m. L of 0. 20 M NH 3. What is the [Cu 2+] at equilibrium? Write the 2+(aq) + 4 NH (aq) Cu(NH ) 2+(aq) Cu 3 3 2 formation reaction and Kf expression. Look up Kf value Determine the concentration of ions in the diluted solutions

Ex 16. 15 – 200. 0 m. L of 1. 5 x 10 -3 M Cu(NO 3)2 is mixed with 250. 0 m. L of 0. 20 M NH 3. What is the [Cu 2+] at equilibrium? Cu 2+(aq) + 4 NH 3(aq) Cu(NH 3)22+(aq) Create an ICE [Cu 2+] [NH 3] [Cu(NH 3)22+] table. Since Kf is large, assume all Initial 6. 7 E-4 0. 11 0 2+ the Cu is Change -≈6. 7 E-4 -4(6. 7 E-4) + 6. 7 E-4 converted into complex ion, then Equilibrium x 0. 11 6. 7 E-4 the system returns X= the small amt of Cu 2+ left in solution to equilibrium

Ex 16. 15 – 200. 0 m. L of 1. 5 x 10 -3 M Cu(NO 3)2 is mixed with 250. 0 m. L of 0. 20 M NH 3. What is the [Cu 2+] at equilibrium? Substitute in and solve for x Cu 2+(aq) + 4 NH 3(aq) Cu(NH 3)42+(aq) confirm the “x is small” approximation Initial Change [Cu 2+] [NH 3] [Cu(NH 3)22+] 6. 7 E-4 0. 11 0 -≈6. 7 E-4 -4(6. 7 E-4) + 6. 7 E-4 Equilibrium x -4 0. 11 6. 7 E-4 -13 since 2. 7 x 10 << 6. 7 x 10 , the approximation is valid

The Effect of Complex Ion Formation on Solubility • the solubility of an ionic compound containing a metal cation that forms a complex ion increases in the presence of aqueous ligands Ag. Cl(s) Ag+(aq) + Cl−(aq) Ksp = 1. 77 x 10 -10 The silver chloride solubility increases due to consumption of the Ag+ ion by NH 3 according to Le. Chatelier’s principle as seen by the large formation constant: Ag+(aq) + 2 NH 3(aq) Ag(NH 3)2+(aq) Kf = 1. 7 x 107

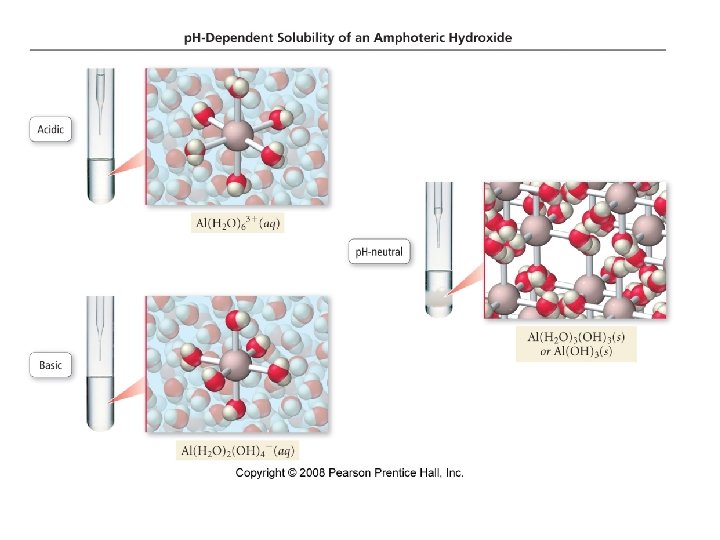
Solubility of Amphoteric Metal Hydroxides • many metal hydroxides are insoluble • all metal hydroxides become more soluble in acidic solution ü shifting the equilibrium to the right by removing OH− • some metal hydroxides also become more soluble in basic solution ü acting as a Lewis base forming a complex ion • substances that behave as both an acid and base are said • to be amphoteric some cations that form amphoteric hydroxides include Al 3+, Cr 3+, Zn 2+, Pb 2+, and Sb 2+

3+ Al • Al 3+ is hydrated in water to form an acidic solution Al(H 2 O)63+(aq) + H 2 O(l) Al(H 2 O)5(OH)2+(aq) + H 3 O+(aq) • addition of OH− drives the equilibrium to the right and continues to remove H from the molecules Al(H 2 O)5(OH)2+(aq) + OH−(aq) Al(H 2 O)4(OH)2+(aq) + H 2 O (l) Al(H 2 O)4(OH)2+(aq) + OH−(aq) Al(H 2 O)3(OH)3(s) + H 2 O (l)
- Slides: 128