Chapter 15 Acids Bases Acids Sour taste Corrosive

Chapter 15 Acids & Bases

Acids • Sour taste • Corrosive • React with certain metals to produce hydrogen gas. • React with carbonates and bicarbonates to produce carbon dioxide gas Bases • Bitter taste • Feel slippery • Corrosive

Arrhenius acid is a substance that produces H+ (H 3 O+) in water Arrhenius base is a substance that produces OH- in water
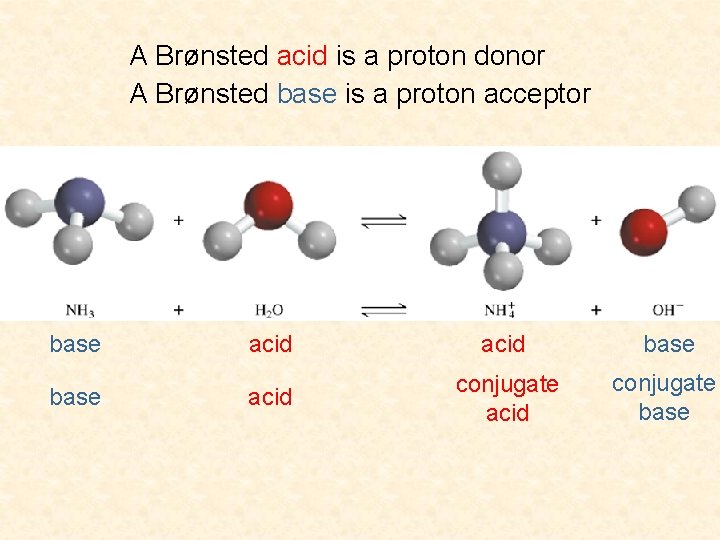
A Brønsted acid is a proton donor A Brønsted base is a proton acceptor base acid conjugate base

A Brønsted acid must contain at least one ionizable proton! • Monoprotic acids = yield one H+ ion • Diprotic acids = 2 H+ ions in 2 separate steps • Triprotic acids = 3 H+ ions; rather rare
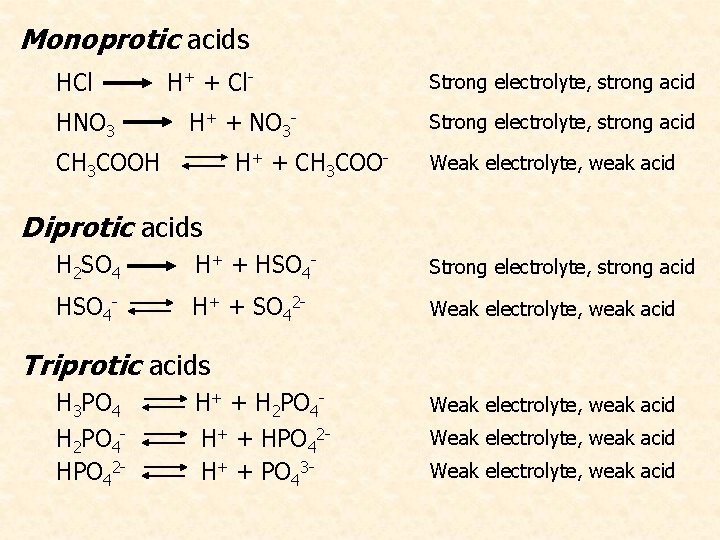
Monoprotic acids HCl HNO 3 H+ + Cl. H+ + NO 3 - CH 3 COOH H+ + CH 3 COO- Strong electrolyte, strong acid Weak electrolyte, weak acid Diprotic acids H 2 SO 4 H+ + HSO 4 - Strong electrolyte, strong acid HSO 4 - H+ + SO 42 - Weak electrolyte, weak acid Triprotic acids H 3 PO 4 H 2 PO 4 HPO 42 - H+ + H 2 PO 4 H+ + HPO 42 H+ + PO 43 - Weak electrolyte, weak acid

Definition of An Acid Arrhenius acid is a substance that produces H+ (H 3 O+) in water A Brønsted acid is a proton donor
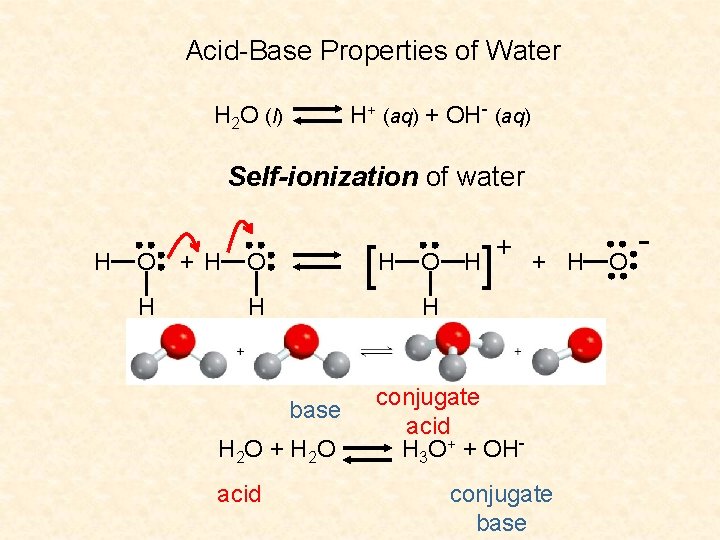
Acid-Base Properties of Water H+ (aq) + OH- (aq) H 2 O (l) Self-ionization of water H O H + H [H O H ] H + H H base H 2 O + H 2 O acid O + conjugate acid H 3 O+ + OHconjugate base O -
![p. H – A Measure of Acidity p. H = -log [H+] Solution Is p. H – A Measure of Acidity p. H = -log [H+] Solution Is](http://slidetodoc.com/presentation_image_h2/6237af348c3ce426d8981047fffd649b/image-9.jpg)
p. H – A Measure of Acidity p. H = -log [H+] Solution Is neutral [H+] = [OH-] At 250 C [H+] = 1 x 10 -7 p. H = 7 acidic [H+] > [OH-] [H+] > 1 x 10 -7 p. H < 7 basic [H+] < [OH-] [H+] < 1 x 10 -7 p. H > 7 p. H [H+]
![Ion-Product Constant: [H+][OH-] = Kw = 1. 0 x 10 -14 -log [H+] – Ion-Product Constant: [H+][OH-] = Kw = 1. 0 x 10 -14 -log [H+] –](http://slidetodoc.com/presentation_image_h2/6237af348c3ce426d8981047fffd649b/image-10.jpg)
Ion-Product Constant: [H+][OH-] = Kw = 1. 0 x 10 -14 -log [H+] – -log [OH-] = 14. 00 p. H + p. OH = 14. 00 p. OH = -log [OH-]

The p. H of rainwater collected in a certain region of the northeastern United States on a particular day was 4. 82. What is the H+ ion concentration of the rainwater? p. H = -log [H+] = 10 -p. H = 10 -4. 82 = 1. 5 x 10 -5 M The OH- ion concentration of a blood sample is 2. 5 x 10 -7 M. What is the p. H of the blood? p. H + p. OH = 14. 00 p. OH = -log [OH-] = -log (2. 5 x 10 -7) = 6. 60 p. H = 14. 00 – p. OH = 14. 00 – 6. 60 = 7. 40

What is the p. H of a 2 x 10 -3 M HNO 3 solution? HNO 3 is a strong acid – 100% dissociation. Start 0. 002 M HNO 3 (aq) + H 2 O (l) End 0. 0 M H 3 O+ (aq) + NO 3 - (aq) 0. 002 M p. H = -log [H+] = -log [H 3 O+] = -log(0. 002) = 2. 7 What is the p. H of a 1. 8 x 10 -2 M Ba(OH)2 solution? Ba(OH)2 is a strong base – 100% dissociation. Start 0. 018 M Ba(OH)2 (s) End 0. 0 M Ba 2+ (aq) + 2 OH- (aq) 0. 018 M 0. 036 M p. H = 14. 00 – p. OH = 14. 00 + log(0. 036) = 12. 6

Antacids and the Stomach p. H Balance Na. HCO 3 (aq) + HCl (aq) Na. Cl (aq) + H 2 O (l) + CO 2 (g) Mg(OH)2 (s) + 2 HCl (aq) Mg. Cl 2 (aq) + 2 H 2 O (l)
![Acid-Base Indicators HIn (aq) H+ (aq) + In- (aq) [HIn] 10 Color of acid Acid-Base Indicators HIn (aq) H+ (aq) + In- (aq) [HIn] 10 Color of acid](http://slidetodoc.com/presentation_image_h2/6237af348c3ce426d8981047fffd649b/image-14.jpg)
Acid-Base Indicators HIn (aq) H+ (aq) + In- (aq) [HIn] 10 Color of acid (HIn) predominates [In ] [HIn] -) predominates Color of conjugate base (In 10 [In-]

p. H
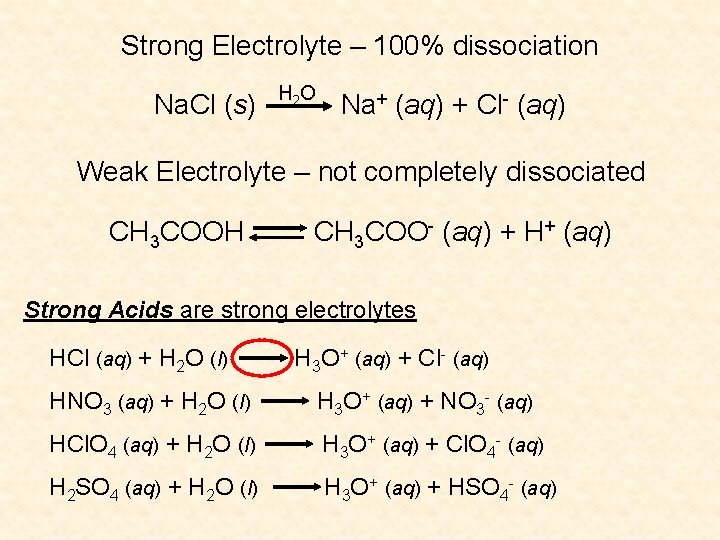
Strong Electrolyte – 100% dissociation Na. Cl (s) H 2 O Na+ (aq) + Cl- (aq) Weak Electrolyte – not completely dissociated CH 3 COOH CH 3 COO- (aq) + H+ (aq) Strong Acids are strong electrolytes HCl (aq) + H 2 O (l) H 3 O+ (aq) + Cl- (aq) HNO 3 (aq) + H 2 O (l) H 3 O+ (aq) + NO 3 - (aq) HCl. O 4 (aq) + H 2 O (l) H 3 O+ (aq) + Cl. O 4 - (aq) H 2 SO 4 (aq) + H 2 O (l) H 3 O+ (aq) + HSO 4 - (aq)
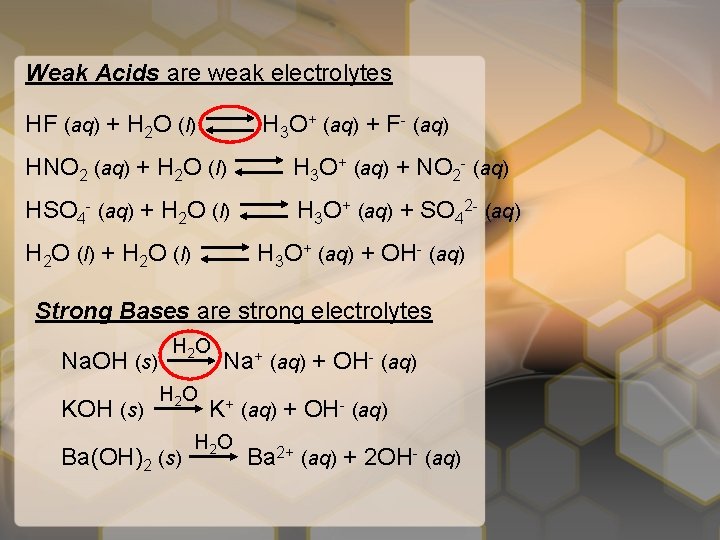
Weak Acids are weak electrolytes HF (aq) + H 2 O (l) H 3 O+ (aq) + F- (aq) HNO 2 (aq) + H 2 O (l) H 3 O+ (aq) + NO 2 - (aq) HSO 4 - (aq) + H 2 O (l) H 3 O+ (aq) + SO 42 - (aq) H 2 O (l) + H 2 O (l) H 3 O+ (aq) + OH- (aq) Strong Bases are strong electrolytes Na. OH (s) KOH (s) H 2 O Ba(OH)2 (s) Na+ (aq) + OH- (aq) K+ (aq) + OH- (aq) H 2 O Ba 2+ (aq) + 2 OH- (aq)

Weak Bases are weak electrolytes F- (aq) + H 2 O (l) NO 2 - (aq) + H 2 O (l) OH- (aq) + HF (aq) OH- (aq) + HNO 2 (aq) Conjugate acid-base pairs: • The conjugate base of a strong acid has no measurable strength. • H 3 O+ is the strongest acid that can exist in aqueous solution. • The OH- ion is the strongest base that can exist in aqueous solution.

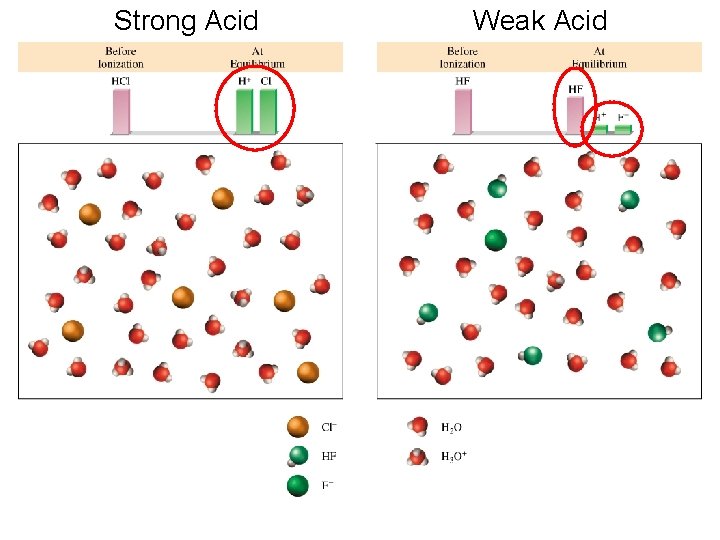
Strong Acid Weak Acid
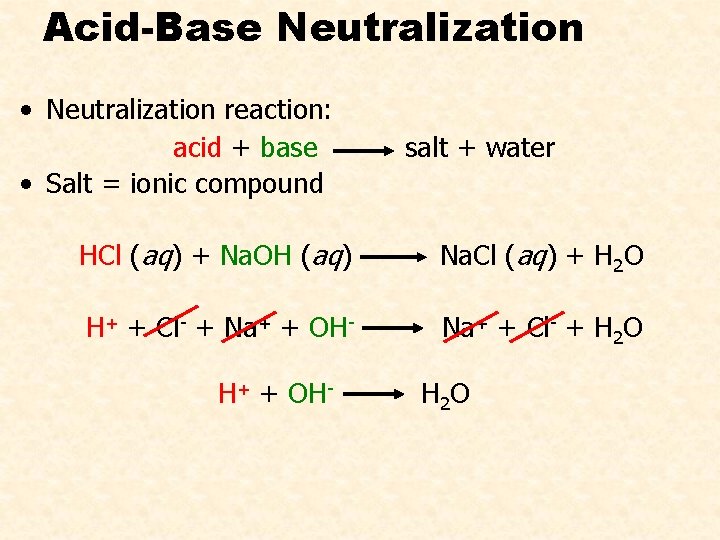
Acid-Base Neutralization • Neutralization reaction: acid + base • Salt = ionic compound salt + water HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O H+ + Cl- + Na+ + OH- Na+ + Cl- + H 2 O H+ + OH- H 2 O

Titrations Titration: a solution of known concentration is added gradually to a solution of unknown concentration

Equivalence point – the point at which the reaction is complete Indicator – substance that changes color at (or near) the equivalence point Slowly add base to unknown acid UNTIL The indicator changes color (pink)
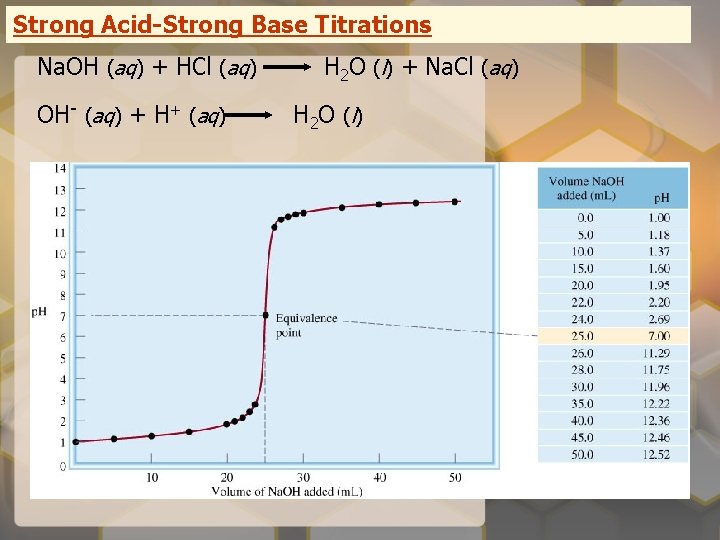
Strong Acid-Strong Base Titrations Na. OH (aq) + HCl (aq) OH- (aq) + H+ (aq) H 2 O (l) + Na. Cl (aq) H 2 O ( l)

Acid-Base Properties of Salts Neutral Solutions: Salts resulting from strong acid-strong base reactions. Na. Cl (s) H 2 O Na+ (aq) + Cl- (aq) Basic Solutions: Salts derived from a strong base and a weak acid. Na. CH 3 COOH (s) H 2 O CH 3 COO- (aq) + H 2 O (l) Na+ (aq) + CH 3 COO- (aq) CH 3 COOH (aq) + OH- (aq)

Acid-Base Properties of Salts Acid Solutions: Salts derived from a strong acid and a weak base.

Acid-Base Properties of Salts Acid Solutions: Salts with small, highly charged metal cations (e. g. Al 3+, Cr 3+, and Be 2+) and the conjugate base of a strong acid. 3+ Al(H 2 O)6 (aq) 2+ Al(OH)(H 2 O)5 (aq) + H+ (aq)

Buffers 1. A weak acid or a weak base and 2. The salt of the weak acid or weak base Both must be present! A buffer solution has the ability to resist changes in p. H upon the addition of small amounts of either acid or base.
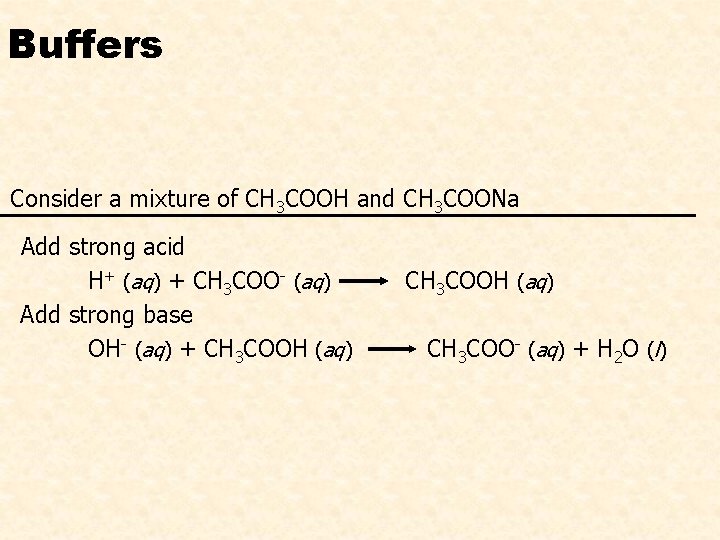
Buffers Consider a mixture of CH 3 COOH and CH 3 COONa Add strong acid H+ (aq) + CH 3 COO- (aq) Add strong base OH- (aq) + CH 3 COOH (aq) CH 3 COO- (aq) + H 2 O (l)

Which of the following are buffer systems? (a) KF/HF (b) KBr/HBr (a) HF is a weak acid and F- is its conjugate base buffer solution (b) HBr is a strong acid not a buffer solution
- Slides: 30