Chapter 15 Acids and Bases Homework 13 14

Chapter 15 – Acids and Bases Homework: 13, 14, 15, 16, 17, 18, 20, 21, 22, 23, 24, 25, 27, 29, 30, 31, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 50, 52, 53, 55, 56, 61, 62, 65, 71, 72, 73, 74, 75, 81, 82, 83, 85, 86, 87, 88, 92, 93, 95, 97, 99, 101, 102

15. 3 - Bronsted-Lowry Acids and Bases*** n In chapter 4, introduced the Arrhenius concept of acids and bases n n An Arrhenius acid was something that produced a H+ ion in an aqueous solution An Arrhenius base was something that produced a OHion in an aqueous solution Unfortunately, Arrhenius concept limits acids and bases to aqueous solutions. Bronsted and Lowry expanded and made a more general definition of acids and bases.

The H+ Ion in Water. n Given this equation, we see that hydrogen chloride ionizes in water to form H+(aq). n n n An H+ ion is a proton with no valence electrons This small, positively charged particle interacts strongly with the nonbonding electrons pairs of the water molecules This forms hydrated hydrogen ions

The hydronium ion n When H+ interacts with water, it forms the hydronium ion n n H 3 O+ The hydronium ion will then form bonds with other water molecules, making large clusters of hydrated hydrogen ions n n H 5 O 2+ or H 9 O 4+ Chemists will use H+(aq) and H 3 O+(aq) interchangeably. n It is the hydrated proton that is responsible for the properties of aqueous solutions of acids

Proton-Transfer Reactions n When we closely look at the reaction when HCl dissolves in water, we find the HCl molecule actual transfers an H+ ion (proton) to the water molecule n n So we can represent the reaction between an HCl molecule and a water molecule to form hydronium and chloride ions HCl(g) + H 2 O(l) H 3 O+(aq) + Cl-(aq)

Defining Acids and Bases n Bronsted and Lowry defined acids and bases in terms of their ability to transfer protons n n An acid is a substance (molecule or ion) that can donate a proton to another substance A base in a substance that can accept a proton n So when HCl dissolves in water, HCl acts as a Bronsted. Lowry acid n n It donates a proton to H 2 O And H 2 O would act as a Bronsted-Lowry base because it accepts a proton

n Because the emphasis is on proton transfer the concept also applies to reactions that do not involve solutions n HCl + NH 3 Cl- + NH 4+ n n HCl is the Bronsted-Lowry acid NH 3 is the Bronsted-Lowry base

Another Look n NH 3(aq) + H 2 O(l) ⇄NH 4+(aq) + OH-(aq) n n n Ammonia is an Arrhenius base because adding it to water produces OHIt is a Bronsted-Lowry base because it accepts a proton from H 2 O The H 2 O is the Bronsted-Lowry acid because it donates a proton to the NH 3 molecule

n Acids and bases always work together to transfer a proton n A substance can function as an acid ONLY if there is another substance to simultaneously act like a base To be a Bronsted-Lowry acid, a molecule or ion MUST have a hydrogen atom it can lose as an H+ ion To be a Bronsted-Lowry base, a molecule or ion MUST have a nonbinding pair of electrons to use to bind the H+ ion

Both? n Some substances can act like an acid in one reaction and a base in another n H 2 O is a Bronsted-Lowry base when reacting with HCl n n H 2 O is a Bronsted-Lowry acid when reacting with NH 3 n n HCl(g) + H 2 O(l) H 3 O+(aq) + Cl-(aq) NH 3(aq) + H 2 O(l) ⇄NH 4+(aq) + OH-(aq) A substance that is capable of being both an acid and a base is called amphiprotic n n Acts a base when combined with something more acidic than itself Acts as an acid when combined with something more basic than itself


Conjugate Acid-Base Pairs n n In any acid-base equilibrium both the forward and reverse reaction involve proton transfers Example n n Consider the reaction of an acid (HX) with water HX(aq) + H 2 O(l) ⇆ X-(aq) + H 3 O+(aq) n In the forward reaction HX donates a proton to H 2 O n n So HX is the acid, and H 2 O is the base In the reverse reaction, H 3 O+ donates a proton to the Xion n So H 3 O+ is the acid, and X- is the base

n An acid and a base such as HX and X- that differ only because of the presence of a proton are called a conjugate acid-base pair n Every acid has a conjugate base n Formed from the removal of a proton from the acid n n OH- is the conjugate base of H 2 O X- is the conjugate base of HX

Likewise n Every base has a conjugate acid n Formed by adding a proton to the base n n H 3 O+ is the conjugate acid of H 2 O HX is the conjugate acid of X-

Examples with Equations n n In any acid-base reaction, we can identify two sets of conjugate acid-base pairs. Nitrous acid and water remove H+ n HNO 2(aq) + H 2 O(l) ⇄ NO 2 -(aq) + H 3 O+(aq) Acid Base Conjugate base add H+ Conjugate acid
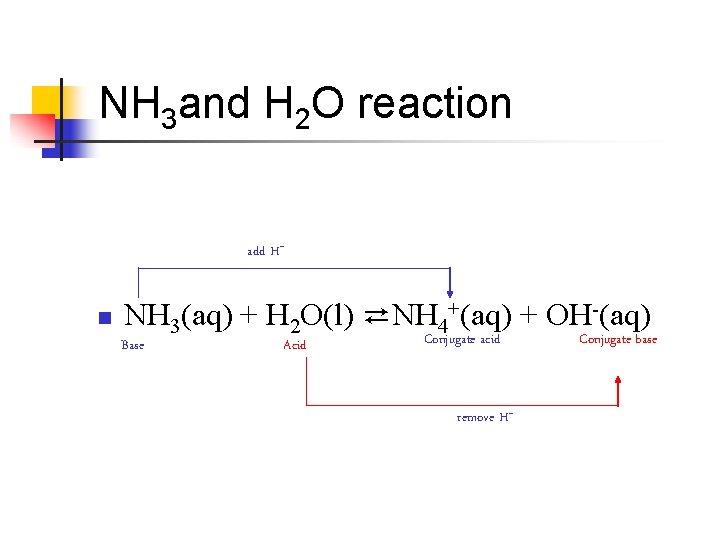
NH 3 and H 2 O reaction add H+ n NH 3(aq) + H 2 O(l) ⇄NH 4+(aq) + OH-(aq) Base Acid Conjugate acid remove H+ Conjugate base

Example n What is the conjugate base of each of the following acids n HCl. O 4 n n H 2 S n n HS- PH 4+ n n Cl. O 4 - PH 3 HCO 3 n CO 3 -2

Example 2 n What is the conjugate acid for each of the following bases? n CNn n SO 42 n n HSO 4 - H 2 O n n HCN H 3 O+ HCO 3 n H 2 CO 3

Example 3 n The hydrogen sulfite ion (HSO 3 -) is amphiprotic n Write an equation for the reaction of HSO 3 with water, in which the ion acts as an acid n n Show the conjugate acid-base pairs Then write an equation for the reaction of HSO 3 - with water, in which the ion acts as an base n Also identify the conjugate acid-base pairs

HSO 3 - as acid then base n HSO 3 -(aq) + H 2 O(l) ⇄SO 32 - (aq) + H 3 O+(aq) n Conjugate pairs n n n HSO 3 -(aq) and SO 32 - (aq) are acid and conjugate base, respectively H 2 O(l) and H 3 O+(aq) are base and conjugate acid, respectively HSO 3 -(aq) + H 2 O(l) ⇄H 2 SO 3(aq) + OH-(aq) n Conjugate pairs n n HSO 3 -(aq) and H 2 SO 3(aq) are base and conjugate acid, respectively H 2 O(l) and OH-(aq) are acid and conjugate base, respectively

15. 4 – Acid Strength and Acid Ionization Constant Ka n n n Some acids are better proton donors than others Likewise, some bases are better proton acceptors than others If we arrange acids in order of their ability to donate a proton. . . n n We find the easier they give up a proton, the less easily its conjugate base accepts a proton Similarly, the more easily a base accepts a proton, the less easily the conjugate acid gives up a proton

Generally n The stronger an acid (more easily it gives up a proton), the weaker it’s conjugate base. n n If we know something about the strength of an acid, we also know something about the strength of its conjugate base. We can break up acids and bases into 3 broad categories, based on their behavior in water
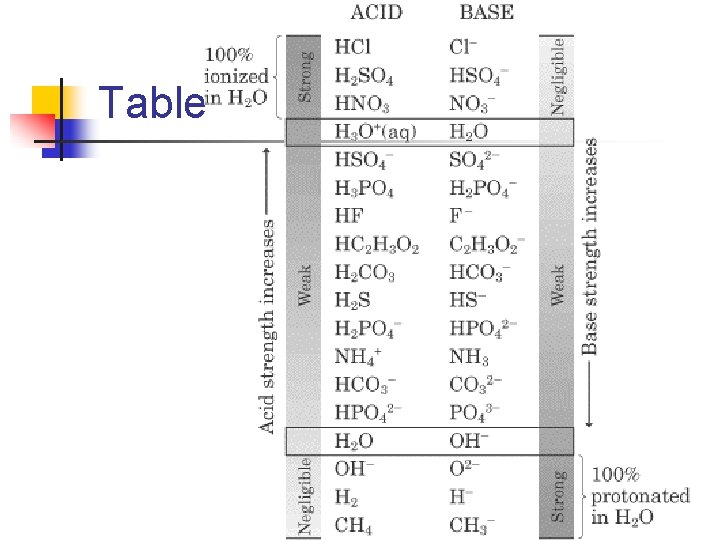
Table
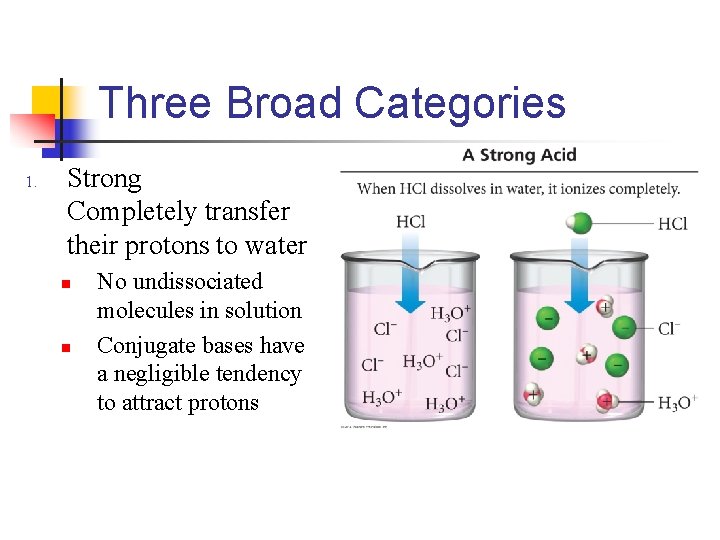
Three Broad Categories 1. Strong Completely transfer their protons to water n n No undissociated molecules in solution Conjugate bases have a negligible tendency to attract protons

Three Broad Categories 2. Weak acids/Weak Base n n n Only partially dissociate in water Exist in solution as a mixture of acid molecules and their constituent ions Conjugate bases show slight ability to attract protons

Three Broad Categories 3. Negligible acids/Strong Base n n Do not act like acids in water Conjugate bases are very strong

Equilibrium? n In every acid-base reaction, the position of the equilibrium favors the transfer of the proton to the stronger base. n Which means the equilibrium mixture has more of the weaker acid and weaker base and less of the stronger acid and stronger base n n Because strong acid has a weak conjugate base (and vice versa) Which really means the equilibrium will favor the side with the weaker base

The Acid Ionization Constant (Ka) n n We quantify the strength of a weak acid with the acid ionization/dissociation constant (Ka) This follows all other rules for equilibrium constants n n Less than one, equilibrium favors reactants Found through equilibrium constant expression

15. 5 - The Autoionization of Water and p. H n One of the most important properties of water is its ability to act as either an acid or base. n n n In the presence of an acid, water acts like a base (proton acceptor) In the presence of a base, water acts like an acid (proton donor) In fact, one water molecule can donate a proton to another water molecule n This is called the autoionization of water
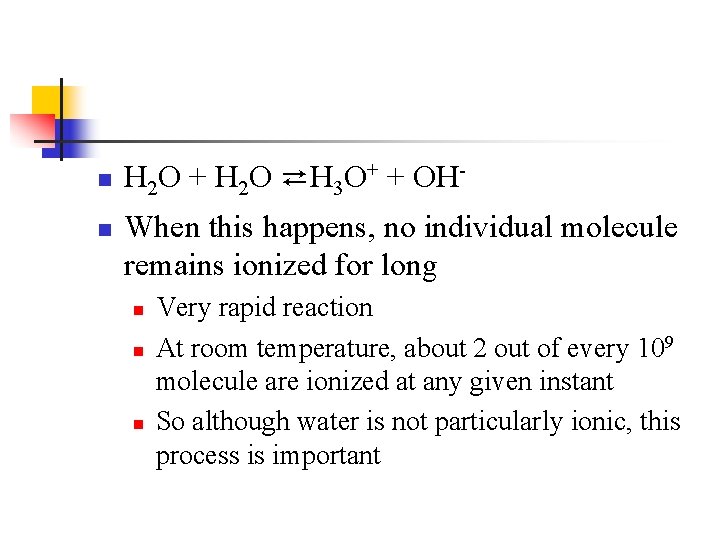
n n H 2 O + H 2 O ⇄H 3 O+ + OHWhen this happens, no individual molecule remains ionized for long n n n Very rapid reaction At room temperature, about 2 out of every 109 molecule are ionized at any given instant So although water is not particularly ionic, this process is important

The Ion Product of Water n Because the ionization of water is an equilibrium process, we can write the following equilibrium-constant expression for it n H 2 O(l) + H 2 O(l) ⇄H 3 O+(aq) + OH-(aq) n Kc = [H 3 O+][OH-] n Where did the H 2 O go? n Not in expression, because is a pure liquid
![Kc = [H 3 O+][OH-] n Because this equilibrium-constant expression refers specifically to the Kc = [H 3 O+][OH-] n Because this equilibrium-constant expression refers specifically to the](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-32.jpg)
Kc = [H 3 O+][OH-] n Because this equilibrium-constant expression refers specifically to the autoionization of water, we use the symbol Kw to represent this n n n Kw is the ion-product constant for water At 25ºC, Kw = 1. 0 x 10 -14 So we can say, that at 25ºC n Kw = [H 3 O+][OH-] = 1. 0 x 10 -14

Playing with the equation n Since H+(aq) and H 3 O+(aq) both mean the hydrated proton, we can also say n n H 2 O(l) ⇄ H+(aq) + OH-(aq) Likewise, Kw can also be expressed as Kw = [H+][OH-] = 1. 0 x 10 -14 This equilibrium-constant expression and Kw are VERY IMPORTANT n Remember these!

Why is this important? n n It is not only applicable to pure water, but to any aqueous solution Although equilibrium between H+ and OH- (along with other ionic equilibria) are affected by the presence of other ions in solution, we tend to ignore those ions except in work requiring a LOT of accuracy n So the equilibrium-constant expression we just made can be used to find either [H+] or [OH-] for a dilute aqueous solution
![n A solution where [H+] = [OH-] is called neutral n n Most solutions n A solution where [H+] = [OH-] is called neutral n n Most solutions](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-35.jpg)
n A solution where [H+] = [OH-] is called neutral n n Most solutions are not neutral. In most solutions, as one of the above increases, the other decreases n n n This keeps the product of their concentrations equal to 1. 0 x 10 -14 When [H+] > [OH-], the solution is acidic When [H+] < [OH-], the solution is basic
![Example n Determine the values of [H+] and [OH-] in a neutral solution at Example n Determine the values of [H+] and [OH-] in a neutral solution at](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-36.jpg)
Example n Determine the values of [H+] and [OH-] in a neutral solution at 25ºC n n Remember, that in a neutral solution [H+] = [OH-] Kw= [H+][OH-]=(x)(x) = 1. 0 x 10 -14 x 2 = 1. 0 x 10 -14 x = 1. 0 x 10 -7 M = [H+] = [OH-]

Example 2 n Calculate the concentration of H+(aq) in n n a solution in which [OH-] is 0. 010 M a solution in which [OH-] is 1. 8 x 10 -9 M n n Unless told otherwise, assume temperature is 25ºC Why is this important?
![A solution in which [OH-] is 0. 010 M n n n [H+][OH-] = A solution in which [OH-] is 0. 010 M n n n [H+][OH-] =](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-38.jpg)
A solution in which [OH-] is 0. 010 M n n n [H+][OH-] = 1. 0 x 10 -14 [H+] = 1. 0 x 10 -14/ [OH-] [H+] = 1. 0 x 10 -14/ 0. 010 [H+] = 1. 0 x 10 -12 Is this solution acidic or basic? n n Basic [OH-] > [H+]
![A solution in which [OH-] is 1. 8 x 10 -9 M n n A solution in which [OH-] is 1. 8 x 10 -9 M n n](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-39.jpg)
A solution in which [OH-] is 1. 8 x 10 -9 M n n n [H+][OH-] = 1. 0 x 10 -14 [H+] = 1. 0 x 10 -14/ [OH-] [H+] = 1. 0 x 10 -14/ 1. 8 x 10 -9 [H+] = 5. 6 x 10 -6 Is this solution acidic or basic? n n Acidic [OH-] < [H+]

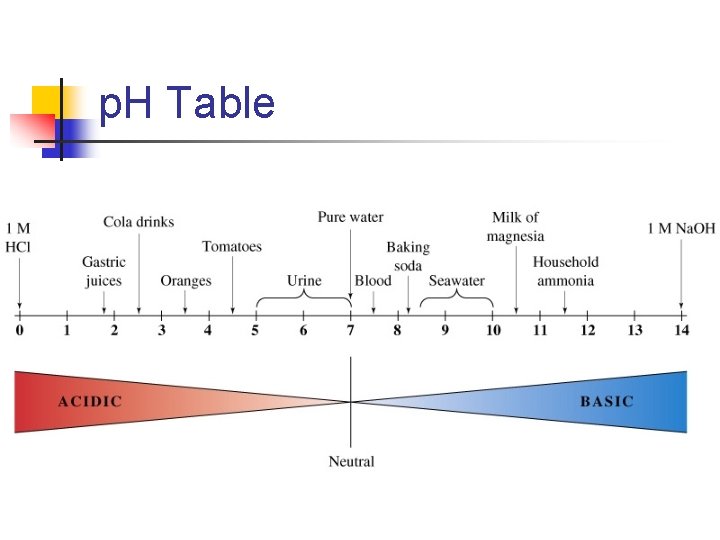
The p. H scale n The concentration of H+(aq) is usually very small n For convenience, we usually express this concentration in terms of p. H n n p. H is the negative logarithm (in base 10) p. H = -log[H+]
![Solution p. H n Neutral solution n n [H+] = [OH-] = 1. 0 Solution p. H n Neutral solution n n [H+] = [OH-] = 1. 0](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-41.jpg)
Solution p. H n Neutral solution n n [H+] = [OH-] = 1. 0 x 10 -7 p. H = -log(1. 0 x 10 -7) p. H = -(-7. 00) = 7. 00 So the p. H of a neutral solution is 7. 00 at 25ºC Acidic solution n n n Acidic solution has [H+] > 1. 0 x 10 -7 Let’s have a p. H of 1. 0 x 10 -3 p. H = -log(1. 0 x 10 -3) p. H = -(-3. 00) = 3. 00 So the p. H for an acidic solution is less than 7. 00 The more acidic the solution, the lower the p. H
![n Basic solution n [H+] < [OH-] Suppose [OH-] = 2 x 10 -3 n Basic solution n [H+] < [OH-] Suppose [OH-] = 2 x 10 -3](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-42.jpg)
n Basic solution n [H+] < [OH-] Suppose [OH-] = 2 x 10 -3 M First we find [H+] (because we need it for p. H) n n n [H+] = Kw / [OH-] = 1. 0 x 10 -14 / 2. 0 x 10 -3 = 5. 0 x 10 -12 M p. H = -log(5. 0 x 10 -12) p. H = 11. 30 So the p. H of a basic solution is greater than 7. 00 The more basic the solution, the higher the p. H
![Notes About p. H n A change in [H+] by a factor of 10 Notes About p. H n A change in [H+] by a factor of 10](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-43.jpg)
Notes About p. H n A change in [H+] by a factor of 10 causes the p. H to change by 1 n n So a solution with a p. H of 6 has 10 x the concentration of H+ than a solution of p. H 7 Also, a solution with a p. H of 1 has 1000 x the concentration of [H+] than a solution with a p. H of 4.
p. H Table

Other “p” Scales n The negative log is also useful for expressing the amounts of other small numbers n n So really, “p” just means -log A common “p” number is p. OH n n n p. OH = -log[OH-] Why is this useful? -log[H+] + (-log[OH-]) = -log. Kw or p. H + p. OH = 14. 00

Measuring p. H n n The p. H of a solution can be measured quickly and accurately with a p. H meter We can also use chemicals called indicators n An indicator is a colored substance that itself can exist in either an acid or a base form n n The two forms have different colors If you know the p. H at which the indicator turns from one color to the other, you can determine whether a solution has a higher or lower p. H value than this value

Indicators

15. 6 – Finding the p. H of a Strong and Weak Acid Solutions n n n The chemistry of aqueous solutions often critically depends on the p. H of the solution Therefore, it is important to look at how the p. H of the solution relates to strong acids and bases Strong acids as bases are called strong electrolytes n This means they exist in aqueous solutions entirely as ions

Strong Acids n The seven most common strong acids include six monoprotic acids and one diprotic acid n n n HCl, HBr, HI, HNO 3, HCl. O 3 and HCl. O 4 H 2 SO 4 For these strong acids, when in solution with these, the solutions exists entirely of H 3 O+ and its conjugate base

Example n n Nitric acid example HNO 3(aq) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq) n n n No equilibrium arrows Reaction goes to completion to products This reaction is also seen as n HNO 3(aq) H+(aq) + NO 3 -(aq)

n In an aqueous solution of a strong acid, the acid is normally the only significant source of H+ ions n n If p. H is more than 6, then we also need to consider the H+ ions from the autoionization of H 2 O Finding the p. H of a strong monoprotic acid is simple n The [H+] equals the original concentration of the acid

Example n If I have a 0. 20 M solution of HNO 3(aq) n n Then the [H+] also equals 0. 20 M Diprotic acids (like H 2 SO 4) are a bit more complex n We will deal with these later

Example 2 n What is the p. H of a 0. 040 M solution of HCl. O 4? n n If HCl. O 4 has a concentration of 0. 040, then [H+] also has a concentration of 0. 040 p. H = -log[H+] = -log(0. 040) = 1. 40

Weak Acids n Most acidic substances are weak acids n n n Will only partially ionize in aqueous solution We use the equilibrium constant for the ionization reaction to find out how much a weak acid ionizes For example purposes, we will represent a weak acid as HA

Therefore n We can write a reaction with a weak acid as such HA (aq) + H 2 O(l) ⇄H 3 O+(aq) + A-(aq) or HA ⇄H+(aq) + A-(aq) n Each of these means the same thing

Finding Ka from p. H n To find one or the other of these things, we treat these problems like an equilibrium problem (from the last chapter)

Example n We have a 0. 10 M solution of formic acid (HCHO 2) and measured the p. H using a p. H meter. n n p. H was measured to be 2. 38 Find the Ka formic acid at this temperature

Step 1 - Write the Equation n n HCHO 2(aq) ⇄H+(aq) + CHO 2 -(aq) Once we have the equation, we can write the equilibrium-constant expression
![Step 2 - Determine concentration of H+ n n n p. H = -log[H+] Step 2 - Determine concentration of H+ n n n p. H = -log[H+]](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-59.jpg)
Step 2 - Determine concentration of H+ n n n p. H = -log[H+] = 2. 38 log[H+] = -2. 38 [H+] = 10 -2. 38 = 4. 2 x 10 -3 M
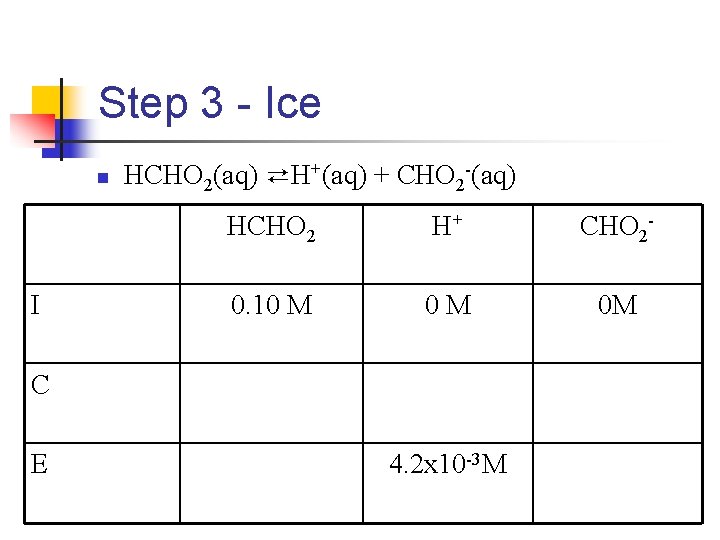
Step 3 - Ice n I HCHO 2(aq) ⇄H+(aq) + CHO 2 -(aq) HCHO 2 H+ CHO 2 - 0. 10 M 0 M 0 M C E 4. 2 x 10 -3 M

Step 4 - Neat Assumption n n Remember, in a weak acid, we will end up with only a small amount of H+ Equilibrium concentration of formic acid n n n (0. 10 - 4. 2 x 10 -3). 10 -. 0042 =. 096 Here’s the deal: 0. 096 is so close to. 10 (only off by 4 thousands of M), that we just assume the concentration of formic acid to be 0. 10

What does this assumption mean for us? n n It means generally we can assume the change in the concentration of the weak acid is so small, its equilibrium concentration will be the same as its initial! We can now plug our values into our equilibrium-constant expression

Step 5 - Plug Everything Into Equilibrium-Constant Expression n HCHO 2(aq) ⇄H+(aq) + CHO 2 -(aq) n This is a reasonable answer n Most weak acids have a Ka between 10 -3 and 10 -10

Using Ka to Calculate p. H n Knowing the Ka AND the initial concentration of the weak acid, we can find [H+] n Which then lets us find the p. H of the weak acid solution

Example n n n 0. 30 M solution of acetic acid, HC 2 H 3 O 2(aq) ⇄H+(aq) + C 2 H 3 O 2+(aq) Ka at 25ºC is 1. 8 x 10 -5 n n According to the formula for acetic acid, the hydrogen that ionizes is the one separate from the rest (it is attached to the oxygen) We write the formula in this way to emphasize that only that one hydrogen is ionized

Step 1 - Write the Equilibrium. Constant Expression n n HC 2 H 3 O 2(aq) ⇄H+(aq) + C 2 H 3 O 2+(aq) Ka at 25ºC is 1. 8 x 10 -5

Step 2 - ICE n HC 2 H 3 O 2(aq) ⇄ H+(aq) + C 2 H 3 O 2 -(aq) Initial Change Equilibrium HC 2 H 3 O 2(aq) H+(aq) C 2 H 3 O 2 -(aq) 0. 30 M 0 0 -x M +x M (0. 30 -x) M x. M

Step 3 - Plug Everything Into Equilibrium-Constant Expression

Simplify n n This will turn into the quadratic But we can simplify this, by remember that Ka is VERY SMALL n n n Because Ka is so small, we recognize equilibrium will be FAR to the left, and that x will be VERY small compared to the initial concentration of acetic acid Thus, we can say that x is negligible compared to 0. 30 Therefore, 0. 30 -x essentially equals 0. 30

New(better) equation! n n n Ka = x 2/0. 30 = 1. 8 x 10 -5 x 2 = (0. 30)(1. 8 x 10 -5) = 5. 4 x 10 -6 x = (5. 4 x 10 -6). 5 = 2. 3 x 10 -3 [H+] = x = 2. 3 x 10 -3 M p. H = -log(2. 3 x 10 -3) = 2. 64

Was Our Assumption Valid? n n We can check to see if our assumption that the initial concentration - x was approximately equal to the initial concentration Find the percent of the acid that ionized n n To do this, divide the [H+] by the initial concentration of the acid, then multiply by 100%. If this value is less than 5%, you were okay. If value was more than 5%, you need to use the quadratic Always check your assumption!
![Assumption Check n n [H+]/[HC 2 H 3 O 2] x 100% 0. 0023 Assumption Check n n [H+]/[HC 2 H 3 O 2] x 100% 0. 0023](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-72.jpg)
Assumption Check n n [H+]/[HC 2 H 3 O 2] x 100% 0. 0023 M/ 0. 30 M x 100% =0. 77% Our assumption was valid

Weak Acid Properties n We find the concentration of H+ is only a very small percentage of the concentration of the acid n n Which means only a small amount of the acid actually donates a proton Because there is such a low concentration of H+, properties relying on that concentration tend to be less evident, compared to a strong acid

n Properties include n n Electrical conductivity Rate of reaction with an active metal

Mixtures of Acids n Consider mixing 0. 10 M HCl with 0. 10 M of HCHO 2 n There are 3 possible sources of H+ ions in this solution n From the dissociation of HCl (strong acid) n n From the partial dissociation of HCHO 2 (weak acid) n n Ka = STRONG Ka = 1. 8 x 10 -4 From the autoionization of water n Ka = 1. 0 x 10 -14

This looks more intimidating that it is n n Because HCl is strong, it completely ionizes But, the H+ formed from the HCl actually suppresses (we’ll learn more about this next chapter) n Think of this in terms of Le. Chatelier’s principle for the weak acid + n HCHO 2(aq) ⇄ H (aq) + CHO 2(aq) So really, we neglect the small contribution of anything but the strongest! This even works with two (or more) weak acids, as long as one is much weaker than the other

15. 7 Base Solutions n Bases are handled very much like acids n There are strong ones and weak ones n n The strong ones dissociate completely The weak ones dissociate or react with water partially

Strong Bases n n There are relatively few common strong bases The most common soluble strong bases are n ionic hydroxides of the alkali metals and the alkaline earth metals n n Na. OH, Li. OH, Ca(OH)2, Mg(OH)2, etc. These compounds completely dissociate into ions in an aqueous solution n n So 0. 30 M Na. OH is really 0. 30 M Na+(aq) and 0. 30 M OH-(aq) In other words, we do not find undissociated Na. OH in a solution
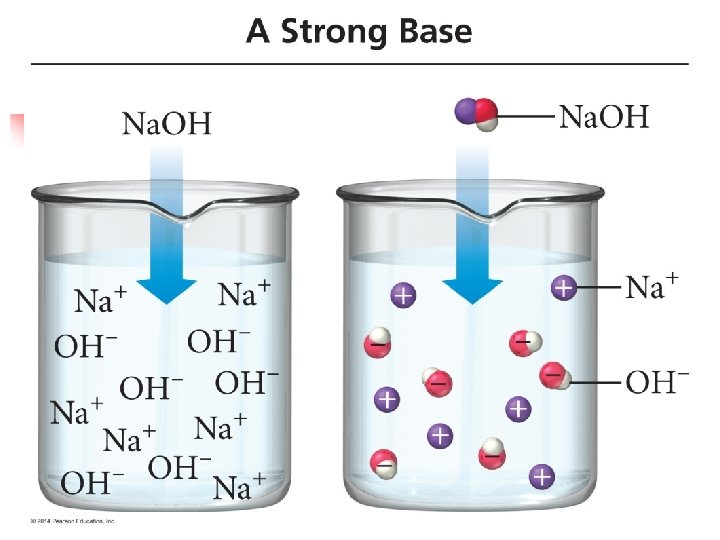

n Because strong bases dissociate entirely (in an aqueous solution), finding their p. H is also relatively straightforward.

Example What is the p. H of a 0. 028 M solution of Na. OH? n n Note: Two ways to do this 1. 2. n Find the [H+] Find p. OH, then convert to p. H I will do both
![Find the [H+] n n n [OH-][H+] = 1. 0 x 10 -14 / Find the [H+] n n n [OH-][H+] = 1. 0 x 10 -14 /](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-82.jpg)
Find the [H+] n n n [OH-][H+] = 1. 0 x 10 -14 / 0. 028 = 3. 57 x 10 -13 M p. H = -log(3. 57 x 10 -13) = 12. 45

Find p. OH, then convert to p. H n n p. OH = -log(0. 028) = 1. 55 p. H + p. OH = 14. 00 - p. OH = p. H 14. 00 -1. 55 = 12. 45

Example 2 n Ca(OH)2 is a strong base that dissociates in water to give TWO OH- ions per formula unit n n This means that the concentration of OH- will be twice that of the concentration of Ca(OH)2 What is the p. H of a 0. 0011 M solution of Ca(OH)2?
![n n n [OH-] = 2 x 0. 0011 M = 0. 0022 M n n n [OH-] = 2 x 0. 0011 M = 0. 0022 M](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-85.jpg)
n n n [OH-] = 2 x 0. 0011 M = 0. 0022 M p. OH = -log(0. 0022) = 2. 66 p. H = 14. 00 - p. OH = 14. 00 - 2. 66 = 11. 34

Other Strong Bases n If a substance reacts with water to form OH- (aq), it will be strongly basic n n Most commonly these will be compounds with the oxide (O 2 -) ion Ionic metal oxides (Na 2 O and Ca. O) are often used in these cases n n With the metal oxides, each mole of the O 2 - reacts to form 2 moles of OH- Ionic hydrides (H-) and nitrides (N 3 -) also will act as bases in water

Weak bases n Many substances behave as weak bases in water n n They react with water, and pull protons from H 2 O Forming the conjugate acid of the base and OHions n B(aq) + H 2 O ⇄ HB+ + OH-(aq)


n The most commonly encountered weak base is ammonia n n n NH 3(aq) + H 2 O(l) ⇄NH 4+(aq) + OH-(aq) So it would have the equilibrium-constant expression of Kb is the special equilibrium constant used in the ionization of a weak base n Called the base-dissociation constant
![Using Kb to calculate [OH-] n Frankly, use the same procedure you would have Using Kb to calculate [OH-] n Frankly, use the same procedure you would have](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-90.jpg)
Using Kb to calculate [OH-] n Frankly, use the same procedure you would have used to find [H+] using Ka n n Use ICE Treat the equilibrium concentration of the weak base as the same as the initial concentration of the weak base

Types of Weak Bases n n It can be a little tricky to determine a weak base from a chemical formula However, weak bases fall into two general categories

Category 1 n The first category includes neutral substances with an atom with a nonbonding pair of electrons n n n These nonbonding pairs of electrons are where the base accepts the proton Many of these substances contain nitrogen Lewis structure necessary to identify

Category 2 n This category includes the anions of weak acids n In other words: The conjugate base of a weak acid

Relationship Between Kaand Kb n n Ka x Kb = K w or p. Ka + p. Kb = p. Kw= 14. 00 What does this mean? n If we know the K value for a conjugate acid/base of what we are looking for, we can find the K value of our target

Example n Find the base-dissociation constant for the fluoride ion. n n Kb for F- is not listed in any tables However, Ka for HF is given Ka for HF is 6. 8 x 10 -4 Kb= Kw / Ka= 1. 0 x 10 -14 / 6. 8 x 10 -4 = 1. 6 x 10 -11

Using p. H to Determine the Concentration of a Salt n n A solution is made by adding solid sodium hypochlorite (Na. Cl. O) to enough water to make 2. 00 L of solution has a p. H of 10. 50. How many moles of Na. Cl. O were added to the water? Cl. O-(aq) ⇄HCl. O(aq) + OHn Na. Cl. O is water soluble, so the Cl. O- is what we are really concerned with

Basic Necessary Data n n p. H of solution is 10. 50 Ka for HCl. O is 2. 9 x 10 -8
![Step 1: Find [OH-] n n n p. OH = 14. 00 - p. Step 1: Find [OH-] n n n p. OH = 14. 00 - p.](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-98.jpg)
Step 1: Find [OH-] n n n p. OH = 14. 00 - p. H = 14. 00 - 10. 50 = 3. 50 -log[OH-] = 3. 50 [OH-] = 10 -3. 50 = 3. 16 x 10 -4 M n So this is the equilibrium concentration of OH-
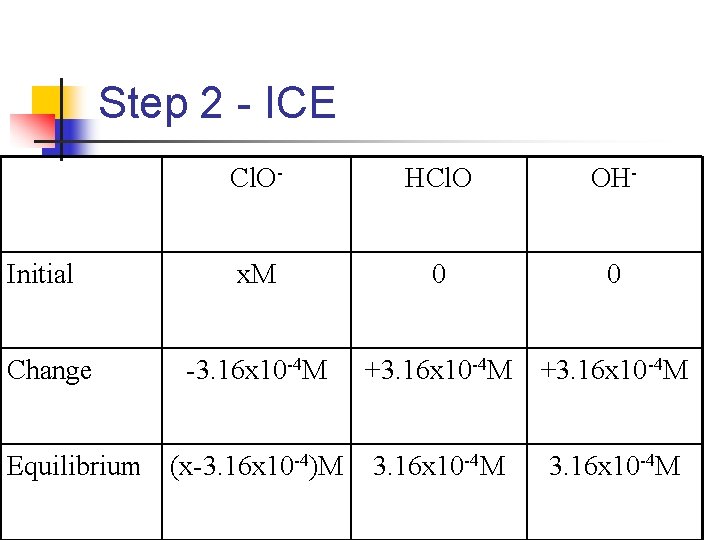
Step 2 - ICE Initial Change Cl. O- HCl. O OH- x. M 0 0 -3. 16 x 10 -4 M +3. 16 x 10 -4 M Equilibrium (x-3. 16 x 10 -4)M
![Step 3 - Find [Cl. O-] Mathmathmath Step 3 - Find [Cl. O-] Mathmathmath](http://slidetodoc.com/presentation_image_h/769f24d644a494a9ce9a315825e843c4/image-100.jpg)
Step 3 - Find [Cl. O-] Mathmathmath

Step 4: Find moles n Molarity of Na. Cl. O is 0. 30 M

15. 8 - Acid-Base Properties of Salt Solutions* n We can assume that when salts dissolve in water, they are completely dissociated n n Most salts are strong electrolytes Consequently, the acid-base properties of salt solutions are due to the behavior of their cations and anions. n n n Many ions will react with water to form H+ or OHThis type of reaction is often called hydrolysis The p. H of an aqueous solution can be predicted by considering the ions of which the salt is composed.

Anions as Weak Bases n In general, an anion (X-) in solution can be considered the conjugate base of an acid n n Cl- is the conjugate base of HCl and C 2 H 3 O 2 - is the conjugate base of HC 2 H 3 O 2 Whether an anion reacts with water to produce hydroxide depends upon the strength of the acid to which it is a conjugate n Remember, the stronger the acid, the weaker the conjugate base

Identifying Original Acid from Conjugate Base n To identify the acid and assess its strength, we simply add a proton to the anion s formula n n X- plus a proton (H+) gives HX If the acid is a strong acid, then the anion in question will be a negligible base n n Therefore, the anion will not affect the p. H of the solution Such as Cl- in an aqueous solution does not affect the p. H, and is treated as a spectator ion in acid-base chemistry

n However, if the HX is NOT one of the seven strong acids, then it is a weak acid n n In this case, the anon is a weak base Which means the anion will react (to a small extent) with water to produce the weak acid and hydroxide ions n n X-(aq) + H 2 O(l) ⇄HX(aq) + OH-(aq) Because OH- is generated, it will increase the p. H of the solution, making it basic

n Anions that have ionizable protons (such as HSO 3 -) are amphiprotic n n n They will act as either an acid or a base Behavior depends on the relative sizes of Ka and Kb for the ion If Ka > Kb, the solution will be acidic

Cations as Weak Acids n Polyatomic cations whose formulas contain one or more protons can be considered the conjugate acid of weak basis n n n NH 4+ is the conjugate acid of the weak base NH 3 So NH 4+ will react with water to lower the p. H However, ions of alkali and some of the heavier alkaline earth metals do not react with water n n n Heavier alkaline earth metals = all but Be Therefore do not affect p. H These exceptions are the cations found in the strong bases

Combined Effect of Cation and Anion in Solution n If an aqueous salt solution contains n n an anion that does NOT react with water a cation that does NOT react with water we expect the p. H to be neutral If the solution contains n n n an anion that reacts to produce hydroxide a cation that does not react with water we expect p. H to be basic

n If the solution contains n n A cation that reacts with water to form a hydronium ion An anion that does not react with water We expect the p. H to be acidic If the solution contains n n n A cation that reacts with water to form a hydronium ion An anion that reacts with water to produce a hydroxide ion p. H becomes more difficult to determine n Each ion must be then considered separately

In Summary 1. 2. 3. 4. An anion that is the conjugate base of a strong acid will not affect the p. H of a solution An anion that is the conjugate base of a weak acid will cause an increase in p. H A cation that is the conjugate acid of a weak base will cause a decrease in p. H The cations of group 1 and heavier members of group 2 (Ca 2+, Sr 2+ and Ba 2+) will not affect p. H

5. 6. Other metal ions will cause a decrease in p. H When a solution contains both the conjugate base of a weak acid and the conjugate acid of a weak base, the ion with the larger equilibrium constant (Ka or Kb) will have the greater influence on p. H

Example List the following in order of increasing p. H n i. iii. iv. 0. 1 M Ba(C 2 H 3 O 2)2 0. 1 M NH 4 Cl 0. 1 M NH 3 CH 3 Br 0. 1 M KNO 3

Analysis 0. 1 M Ba(C 2 H 3 O 2)2 i. n n Ba 2+ is the ion of one of the heavy group 2 elements, will not affect p. H C 2 H 3 O 2 - is the conjugate base of a weak acid. Will make solution basic 0. 1 M NH 4 Cl ii. n n Cl- is the conjugate base of a strong acid. Will not affect p. H NH 4+ is the conjugate acid of a weak base. Will make solution acidic

0. 1 M NH 3 CH 3 Br iii. n n n NH 3 CH 3+ is the conjugate acid of a weak base. Will make solution acid Br- is the conjugate base of a strong acid. Will not affect p. H Kb of NH 3 is 1. 8 x 10 -5 Kb of NH 2 CH 3 is 4. 4 x 10 -4 Since NH 3 has smaller Kb, and is weaker base, NH 4+ is the stronger acid, so ii. will be the lower p. H

0. 1 M KNO 3 iv. n n n K+ ion is the of the strong base KOH NO 3 - is the conjugate base of the strong acid HNO 3 Neither will react with water, making solution neutron Overall increasing p. H n n 0. 1 M NH 4 Cl < 0. 1 M NH 3 CH 3 Br < 0. 1 M KNO 3 < 0. 1 M Ba(C 2 H 3 O 2)2

Example 2 n Predict whether the salt Na 2 HPO 4 will form an acidic or basic solution on dissolving with water n n Na 2 HPO 4 is amphiprotic 2 Possible reactions n n n HPO 42 -(aq) ⇄H+ (aq) + PO 43 -(aq) HPO 42 -(aq) + H 2 O ⇄H 2 PO 4 -(aq) + OH-(aq) Whichever reaction has the larger equilibrium constant will determine whether the solution is acidic or basic

n HPO 42 -(aq) ⇄H+ (aq) + PO 43 -(aq) n n n HPO 42 -(aq) + H 2 O ⇄H 2 PO 4 -(aq) + OH-(aq) n n n This is one of the given equations within the chapter. Ka = 4. 2 x 10 -13 We can find the Kb for this by its conjugate acid, H 2 PO 4 Ka = 6. 2 x 10 -8 Ka x Kb = K w Therefore, Kb= 1. 6 x 10 -7 So this solution is basic

15. 9 - Polyprotic Acids n Many acids have more than one ionizable H atom n n n These acids are called polyprotic acids These tend to ionize in steps Sulfurous Acid example (H 2 SO 3) n H 2 SO 3(aq) ⇄H+(aq) + HSO 3 -(aq) n n Ka 1= 1. 7 x 10 -2 HSO 3 -(aq) ⇄H+(aq) + SO 32 -(aq) n Ka 2= 6. 4 x 10 -8

Ka 1 and Ka 2 ? n The numbers on the constants refer to the particular proton that is ionizing n n n So ka 1 refers to the reaction that ionizes the first proton And ka 2 refers to the reaction that ionizes the second proton Ka 1 > Ka 2 n n n After the first proton left, we have a negatively charged compound. Protons less easily lost from a negatively charged particle Therefore, it is always easier to remove the first proton from a polyprotic acid than it is to remove the second. n And if an acid had 3 ionizable protons, easier to remove the second than the 3 rd.

n n Usually the Ka values for the first and second disassociation constant differs by at least a factor of 103 Note: Sulfuric acid (H 2 SO 4) has a Ka 1 as “large” n n Because with the first proton removed, sulfuric acid acts like a strong acid (no equilibrium) Though with the removal of the second proton, it acts like a weak acid.

Making Life Easy n Because Ka 1 is so much larger than any of the other K values for a given acid, almost all of the [H+] comes from the first ionization n As long as the other Ka values differ by a factor of 103 or more, we can estimate the p. H of a polyprotic acid by looking only at Ka 1

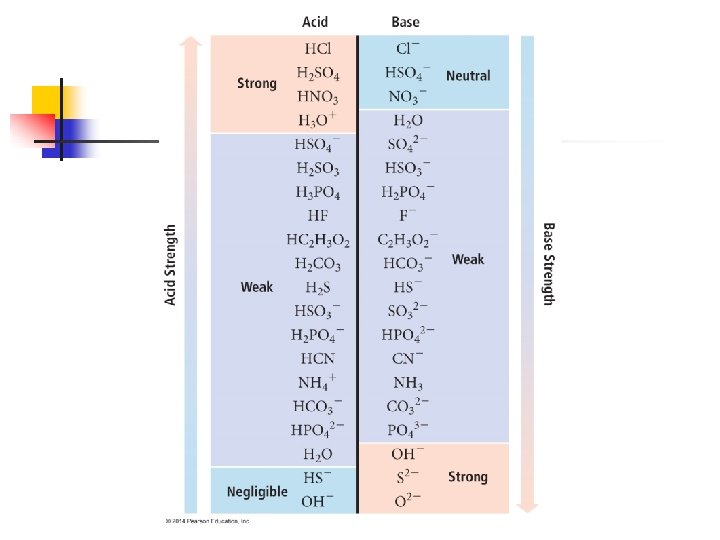
Example n The solubility of CO 2 in pure water at 25ºC at 0. 1 atm is 0. 0037 M. The common practice is to assume that all of the dissolved CO 2 is in the form of the carbonic acid (H 2 CO 3), which is produced by the reaction between the CO 2 and H 2 O. CO 2(aq) + H 2 O(l) ⇄H 2 CO 3(aq)

n n What is the p. H of a 0. 0037 M solution of H 2 CO 3? Ka 1 = 4. 3 x 10 -7 Ka 2 = 5. 6 x 10 -11 Note: Since Ka 2 differs from Ka 1 by more than 103, we treat the acid like a monoprotic acid (we ignore Ka 2)
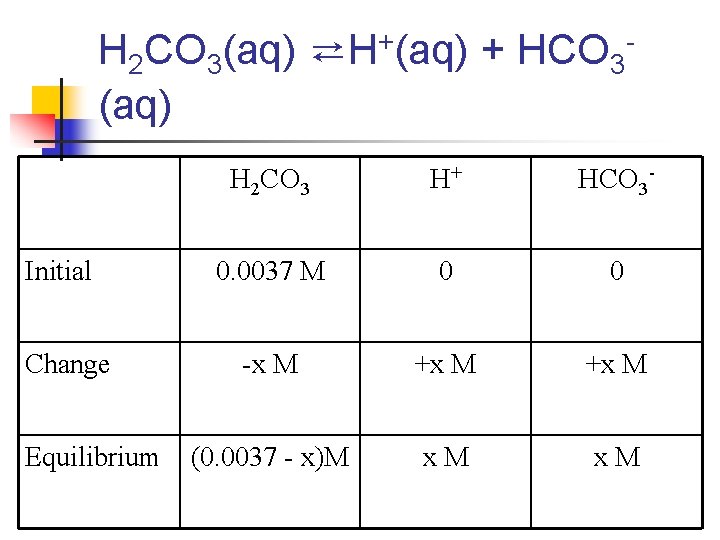
H 2 CO 3(aq) ⇄H+(aq) + HCO 3(aq) Initial Change Equilibrium H 2 CO 3 H+ HCO 3 - 0. 0037 M 0 0 -x M +x M (0. 0037 - x)M x. M

Use the equilibrium constantexpression
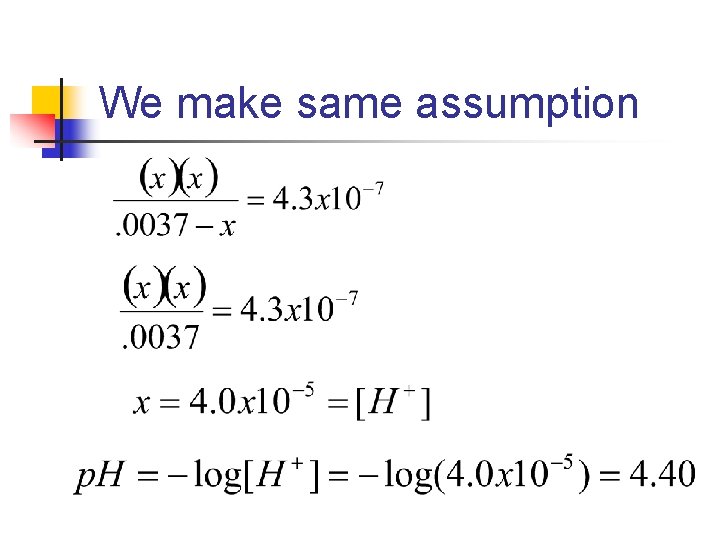
We make same assumption

15. 10 – Acid Strength and Molecular Structure n n When a substance is dissolved in water, it may behave as an acid, base, or be neither. We will look at how the chemical structure of a substance determines these properties

Factors that Affect Acid Strength n A molecule containing H will transfer a proton ONLY if the H-X bond is polarized n n n Meaning that the X has a relatively large electronegativity Which is why HCl is so acidic (Cl has a large electronegativity) Why Na. H is basic n n Because the H atom possesses a negative charge and will attract protons And also why the H-C bond in CH 4 is neither an acid nor a base n Because the H-C bonds are not polar at all

But that is not all! n A second factor that helps determine whether a molecule that has an H-X bond will donate a proton is the strength of the bond n n Very strong bonds are less easily dissociated than weaker ones H-F is the most polar of bonds, but HF is a weak acid n Because the H-F bond is relatively strong compared to other hydrogen halides

And still not all! n A third factor is the stability of the conjugate base, Xn n In general, the greater the stability of the conjugate base, the stronger the acid So the overall strength of an acid is dependent upon these three factors n n n Polarity of the H-X bond Strength of the H-X bond Stability of the conjugate base, X-

Binary Acids n In general, the H-X bond strength is the most important factor in finding the acid strength among binary acids in which X is in the same group in the periodic table n n A binary acid is one containing just hydrogen and one other element The strength of an bond tends to decrease as the element X increases in size n n n Bond strength decreases and the acidity increases down a group HF < HCl H 2 O < H 2 S

Across the Periodic Table n Bond strength change is less moving across a row in the periodic table than down the table n n Bond polarity becomes the major factor determining the acidity of a binary acids in the same row. So acidity generally increases as the electronegativity of X increases n n So acids get stronger from left to right CH 4 < NH 3 << H 2 O < HF

Oxyacids n n Many common acids, such as H 2 SO 4 contain one or more O-H bonds Acids in which OH groups and possibly additional oxygen atoms are bound to a central atom are called oxyacids. n We will look at what factors determine whether an OH group will behave as an acid or a base.

n Consider an OH group bound to some atom, Y, which might in turn have other groups attached to it n n -Y-O-H At one extreme, Y might be a metal, such as Na, K or Mg n n n Because of low electronegativity, electrons are pulled to the oxygen, and ionic compound containing OH- is formed These would be sources of OH-, and would be a base When Y is a nonmetal n n The bond to O is covalent, and substance does not easily lose the OHSo substance would either be neutral or acidic n n Which is which depends on the electronegativity of Y More electronegative Y = more acidic

Y as a nonmetal continued Again, as Y becomes more electronegative, compound becomes more acidic n n Reason for this: 1. As electron density is drawn toward Y, the O-H bond becomes weaker and more polar, favoring the loss of H+

Even More Oxygen! n Many oxyacids contain more oxygen atoms bonded to the central atom Y n n The additional oxygens (all electronegative) pull electron density from the O-H bond This further increases its polarity Also helps stabilize the conjugate base So strength of an acid will increase as additional electromagnetic atoms bond to the central atom, Y.

Summary n For oxyacids that have the same number of OH groups and the same number of O atoms n n Acid strength increases with increasing electronegativity of the central atom For oxyacids that have the same central atom Y n Acid strength increases as the number of oxygen atoms attached to Y increases

Additionally n Because oxidation number of the central atom increases as the number of attached O atoms increases n In a series of oxyacids, the acidity increase as the oxidation number of the central atom increases

Example n Arrange the compounds in the following series in order of increasing acidity n As. H 3, HI, Na. H, H 2 O n n All binary acids So we look at electronegativity n n n More electronegative = more acidic Na. H has lowest electronegativity, so most basic As less electronegative than O, so As more basic than O I most electronegative, so HI most acidic Na. H < As. H 3 < H 2 O < HI

Example 2 n Arrange the compounds in the following series in order of increasing acidity n n H 2 Se. O 3, H 2 Se. O 4, H 2 O Number of oxygens bonded to central atom is the biggest determiner here n Acidity of oxyacids increases with # of oxygen atoms bonded n n So H 2 Se. O 4 is a stronger acid than H 2 Se. O 3 Both H 2 Se. O 4 and H 2 Se. O 3 are similar to H 2 SO 4 and H 2 SO 3 respectively. Both SHOULD be strong acids. Making them stronger acid than water H 2 O < H 2 Se. O 3 < H 2 Se. O 4
- Slides: 141