Chapter 15 AcidBase Equilibria Section 15 4 Titrations

Chapter 15 Acid-Base Equilibria

Section 15. 4 Titrations and p. H Curves Titration Curve § Plotting the p. H of the solution being analyzed as a function of the amount of titrant added. § Equivalence (Stoichiometric) Point – point in the titration when enough titrant has been added to react exactly with the substance in solution being titrated. Copyright © Cengage Learning. All rights reserved 2

Section 15. 4 Titrations and p. H Curves Neutralization of a Strong Acid with a Strong Base To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 3
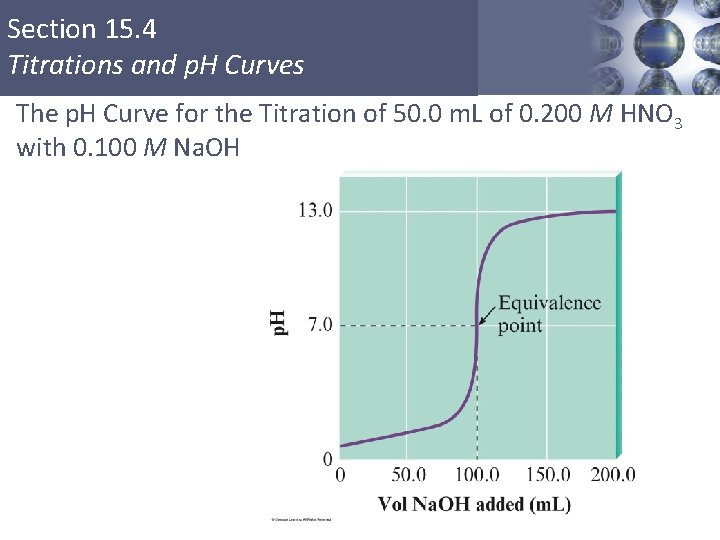
Section 15. 4 Titrations and p. H Curves The p. H Curve for the Titration of 50. 0 m. L of 0. 200 M HNO 3 with 0. 100 M Na. OH Copyright © Cengage Learning. All rights reserved 4
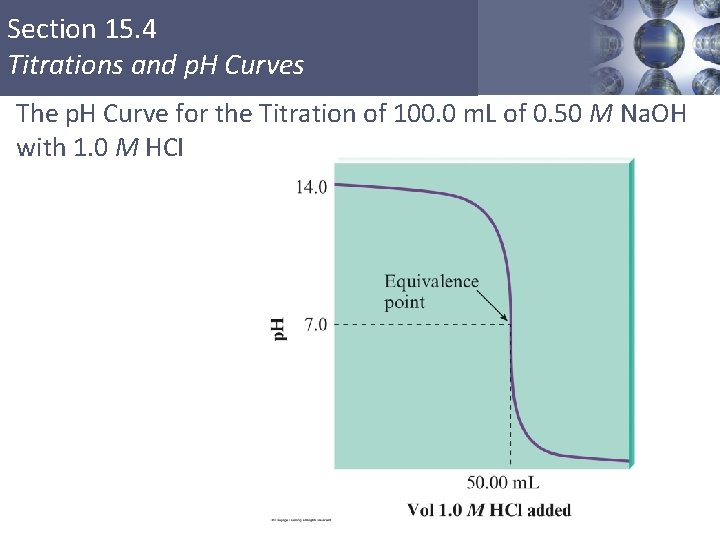
Section 15. 4 Titrations and p. H Curves The p. H Curve for the Titration of 100. 0 m. L of 0. 50 M Na. OH with 1. 0 M HCI Copyright © Cengage Learning. All rights reserved 5

Section 15. 4 Titrations and p. H Curves Weak Acid–Strong Base Titration Step 1: Step 2: A stoichiometry problem (reaction is assumed to run to completion) then determine concentration of acid remaining and conjugate base formed. An equilibrium problem (determine position of weak acid equilibrium and calculate p. H). Copyright © Cengage Learning. All rights reserved 6

Section 15. 4 Titrations and p. H Curves CONCEPT CHECK! Consider a solution made by mixing 0. 10 mol of HCN (Ka = 6. 2 × 10– 10) with 0. 040 mol Na. OH in 1. 0 L of aqueous solution. What are the major species immediately upon mixing (that is, before a reaction)? Copyright © Cengage Learning. All rights reserved 7

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § Why isn’t Na. OH a major species? § Why aren’t H+ and CN– major species? § List all possibilities for the dominant reaction. Copyright © Cengage Learning. All rights reserved 8

Section 15. 4 Titrations and p. H Curves Let’s Think About It… The possibilities for the dominant reaction are: 1. 2. 3. 4. 5. H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH–(aq) HCN(aq) + H 2 O(l) H 3 O+(aq) + CN–(aq) HCN(aq) + OH–(aq) CN–(aq) + H 2 O(l) Na+(aq) + OH–(aq) Na. OH Na+(aq) + H 2 O(l) Na. OH + H+(aq) Copyright © Cengage Learning. All rights reserved 9

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § How do we decide which reaction controls the p. H? H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH–(aq) HCN(aq) + H 2 O(l) H 3 O+(aq) + CN–(aq) HCN(aq) + OH–(aq) CN–(aq) + H 2 O(l)

Section 15. 4 Titrations and p. H Curves Let’s Think About It… HCN(aq) + OH–(aq) CN–(aq) + H 2 O(l) § What are the major species after this reaction occurs? Copyright © Cengage Learning. All rights reserved 11

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § Now you can treat this situation as before. § List the possibilities for the dominant reaction. § Determine which controls the p. H. Copyright © Cengage Learning. All rights reserved 12

Section 15. 4 Titrations and p. H Curves CONCEPT CHECK! Calculate the p. H of a solution made by mixing 0. 20 mol HC 2 H 3 O 2 (Ka = 1. 8 × 10– 5) with 0. 030 mol Na. OH in 1. 0 L of aqueous solution. Copyright © Cengage Learning. All rights reserved 13

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § What are the major species in solution? § Why isn’t Na. OH a major species? § Why aren’t H+ and C 2 H 3 O 2– major species? Copyright © Cengage Learning. All rights reserved 14

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § What are the possibilities for the dominant reaction? 1. 2. 3. 4. 5. H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH–(aq) HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2–(aq) HC 2 H 3 O 2(aq) + OH–(aq) C 2 H 3 O 2–(aq) + H 2 O(l) Na+(aq) + OH–(aq) Na. OH(aq) Na+(aq) + H 2 O(l) Na. OH + H+(aq) § Which of these reactions really occur? Copyright © Cengage Learning. All rights reserved 15

Section 15. 4 Titrations and p. H Curves Let’s Think About It… § Which reaction controls the p. H? H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH–(aq) HC 2 H 3 O 2(aq) + H 2 O(l) H 3 O+(aq) + C 2 H 3 O 2–(aq) HC 2 H 3 O 2(aq) + OH–(aq) C 2 H 3 O 2–(aq) + H 2 O(l) § How do you know? Copyright © Cengage Learning. All rights reserved 16

Section 15. 4 Titrations and p. H Curves Let’s Think About It… HC 2 H 3 O 2(aq) + OH– Before Change 0. 20 mol C 2 H 3 O 2–(aq) + H 2 O 0. 030 mol – 0. 030 mol After 0. 17 mol 0 0 +0. 030 mol K = 1. 8 × 109 Copyright © Cengage Learning. All rights reserved 17

Section 15. 4 Titrations and p. H Curves Steps Toward Solving for p. H HC 2 H 3 O 2(aq) + H 2 O Initial Change Equilibrium H 3 O+ + C 2 H 3 O 2 -(aq) 0. 170 M ~0 0. 030 M –x +x +x 0. 170 – x x 0. 030 + x Ka = 1. 8 × 10– 5 Copyright © Cengage Learning. All rights reserved 18

Section 15. 4 Titrations and p. H Curves EXERCISE! Calculate the p. H of a 100. 0 m. L solution of 0. 100 M acetic acid (HC 2 H 3 O 2), which has a Ka value of 1. 8 × 10– 5. p. H = 2. 87 Copyright © Cengage Learning. All rights reserved 19

Section 15. 4 Titrations and p. H Curves CONCEPT CHECK! Calculate the p. H of a solution made by mixing 100. 0 m. L of a 0. 100 M solution of acetic acid (HC 2 H 3 O 2), which has a Ka value of 1. 8 × 10– 5, and 50. 0 m. L of a 0. 10 M Na. OH solution. Copyright © Cengage Learning. All rights reserved 20

Section 15. 4 Titrations and p. H Curves CONCEPT CHECK! Calculate the p. H of a solution at the equivalence point when 100. 0 m. L of a 0. 100 M solution of acetic acid (HC 2 H 3 O 2), which has a Ka value of 1. 8 × 10– 5, is titrated with a 0. 10 M Na. OH solution. Copyright © Cengage Learning. All rights reserved 21
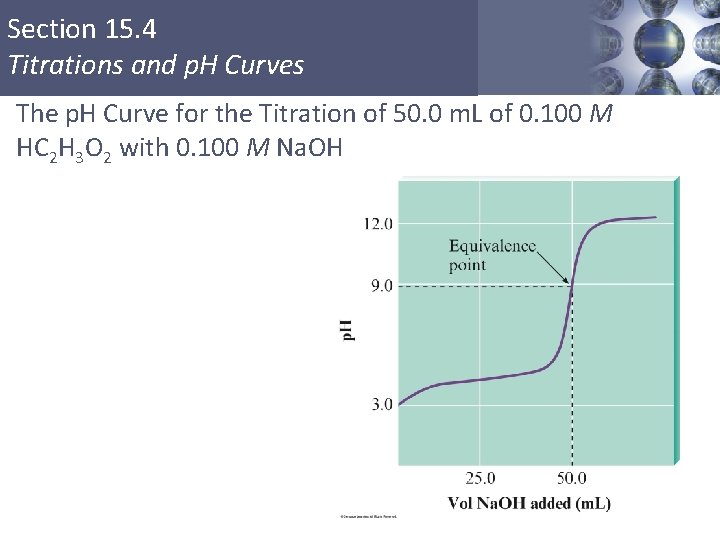
Section 15. 4 Titrations and p. H Curves The p. H Curve for the Titration of 50. 0 m. L of 0. 100 M HC 2 H 3 O 2 with 0. 100 M Na. OH Copyright © Cengage Learning. All rights reserved 22
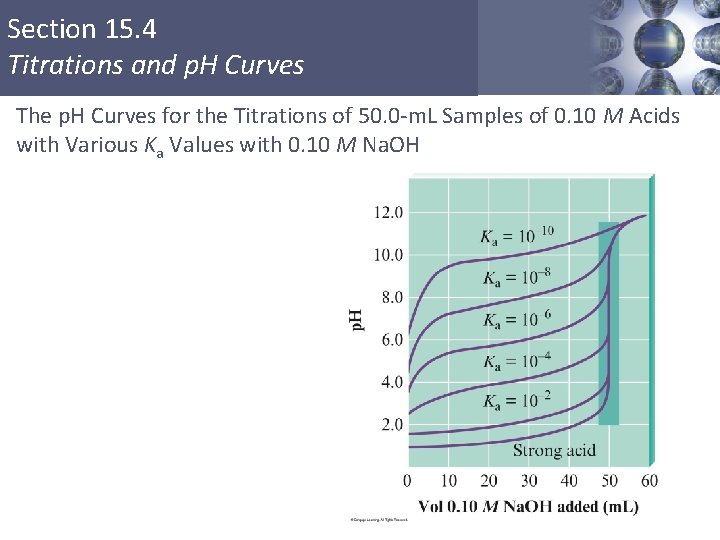
Section 15. 4 Titrations and p. H Curves The p. H Curves for the Titrations of 50. 0 -m. L Samples of 0. 10 M Acids with Various Ka Values with 0. 10 M Na. OH Copyright © Cengage Learning. All rights reserved 23
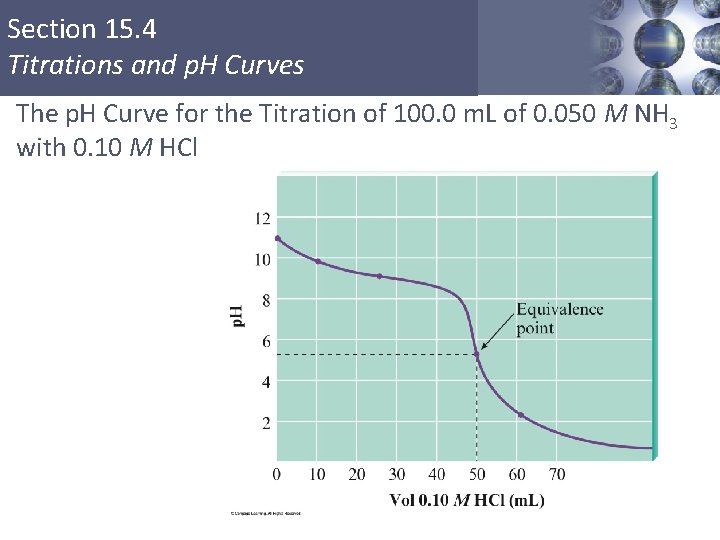
Section 15. 4 Titrations and p. H Curves The p. H Curve for the Titration of 100. 0 m. L of 0. 050 M NH 3 with 0. 10 M HCl Copyright © Cengage Learning. All rights reserved 24

Section 15. 5 Acid-Base Indicators § Marks the end point of a titration by changing color. § The equivalence point is not necessarily the same as the end point (but they are ideally as close as possible). Copyright © Cengage Learning. All rights reserved 25

Section 15. 5 Acid-Base Indicators The Acid and Base Forms of the Indicator Phenolphthalein Copyright © Cengage Learning. All rights reserved 26

Section 15. 5 Acid-Base Indicators The Methyl Orange Indicator is Yellow in Basic Solution and Red in Acidic Solution Copyright © Cengage Learning. All rights reserved 27
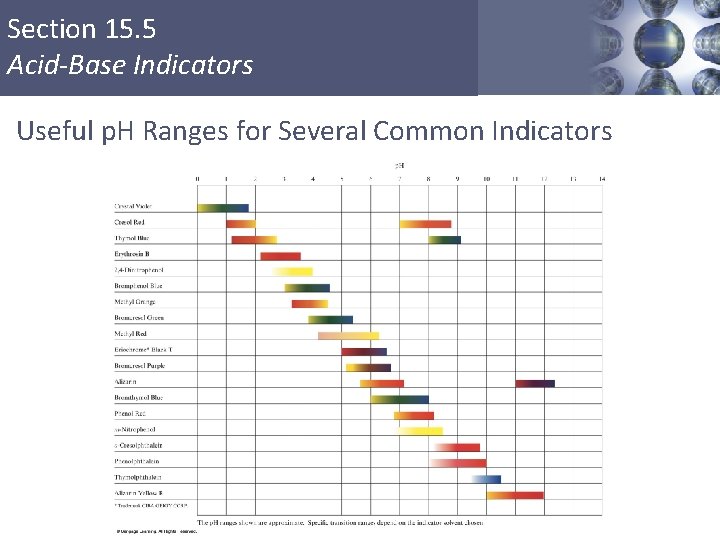
Section 15. 5 Acid-Base Indicators Useful p. H Ranges for Several Common Indicators Copyright © Cengage Learning. All rights reserved 28
- Slides: 28